J Holmgren et al. Inflamm Bowel Dis 2023; 29: 339-348. Open Access! The Risk of Serious Infections Before and After Anti-TNF Therapy in Inflammatory Bowel Disease: A Retrospective Cohort Study
Methods: Retrospective study with 980 patients at 5 centers participating in the Swedish IBD Quality Register. Serious infections, defined as infections requiring in-patient care, the year before and after the start of anti-TNF treatment were evaluated.
Key findings:
- A 72.0% reduction in the incidence rate of perianal abscesses and intra-abdominal abscesses during treatment with anti-TNF was found compared with before treatment.
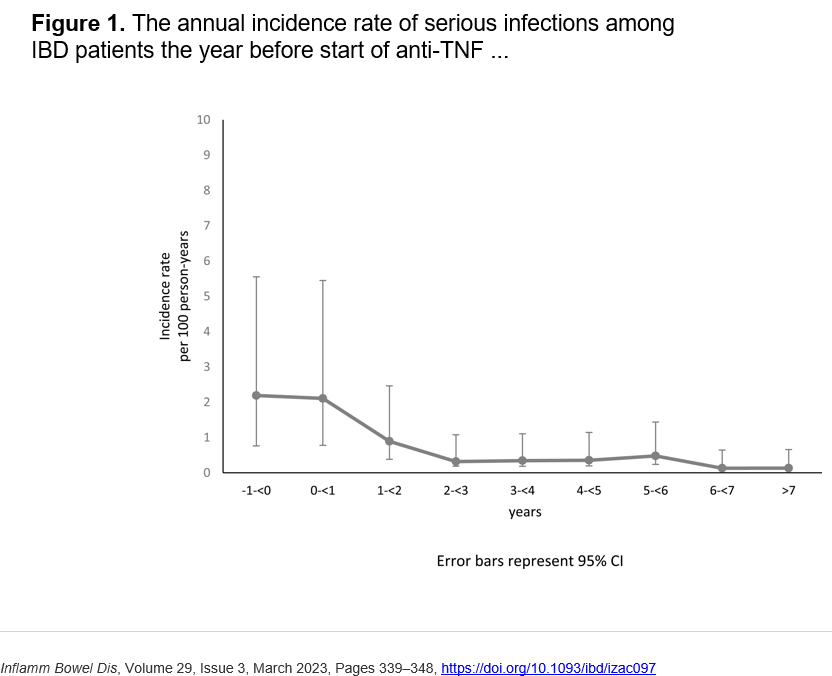
- Figures 2 & 3 show than most infection rates decreased with treatment. CMV infection did not change significantly with 0.10 per 100 person-years prior to treatment and 0.14 per 100 person-years after starting anti-TNF therapy
- ” In the current study, patients younger than 20 years old experienced a substantial decrease of infection incidence rate ratio (0.11) with the introduction of anti-TNF treatment. The results could be explained by the fact that young patients have a more active disease with increased risk of infection before treatment with anti-TNF.”
- “The most common type of infection after anti-TNF treatment was pneumonia. The high incidence of pneumonia confirms earlier data.9,36,37” However, the authors show that the rate of pneumonia dropped from 0.51 to 0.27 per 100 person-years after starting anti-TNF therapy.
The authors note that a prior study by “Zabana et al showed that patients with IBD had an increased risk for serious infection after starting immunosuppressive treatment compared with before treatment (median follow-up 3 years before and 5 years after)… the discrepancy in the result may be explained by selection bias. We included all patients starting anti-TNF treatment. However, Zabana et al included only patients who suffered from infections during immunosuppressive treatment and retrospectively examined the risk of infection before start of treatment.24“
Limitations of study: several other important factors affecting infections were not captured in this study including steroid exposure and nutritional status.
My take (from authors): “The incidence rate of serious infection among IBD patients did not increase with anti-TNF therapy. Instead, serious infections seemed to decrease more than 1 year after initiation of anti-TNF treatment.”
Related blog posts: