RE St Pierre-Hetz et al. JPGN Reports. 2026;1–6. A novel central line securement vest reduces line trauma and improves quality of life in patients with intestinal failure
This single center study compared a novel central line securement vest (n=12) to traditional securing methods (n=11).
Key findings:
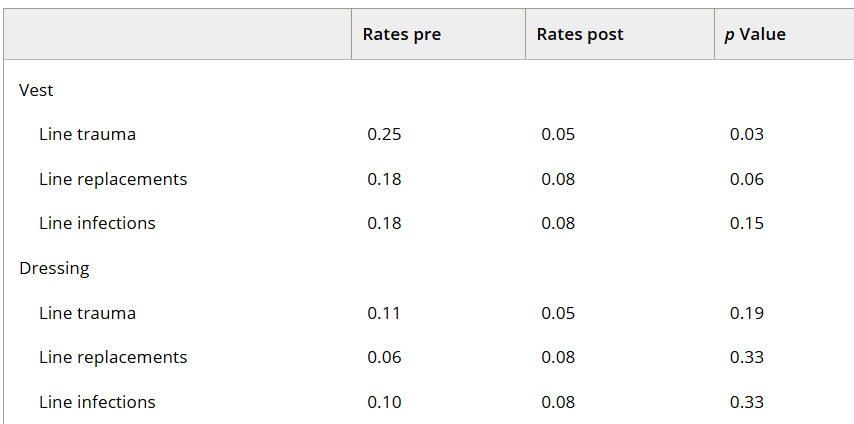
- The vest group had lower rates of line trauma during the period of vest use versus prior to its use. The rates of infections and replacements were a little lower during vest use (not statistically-significant) compared to prior to use. The control group had similar rates of trauma, infections, and replacements before and during the study period.
- The vest group had much higher rates of line issues prior to vest use than the control group
- 5 of the 12 vest patients discontinued use before the planned 12-month evaluation
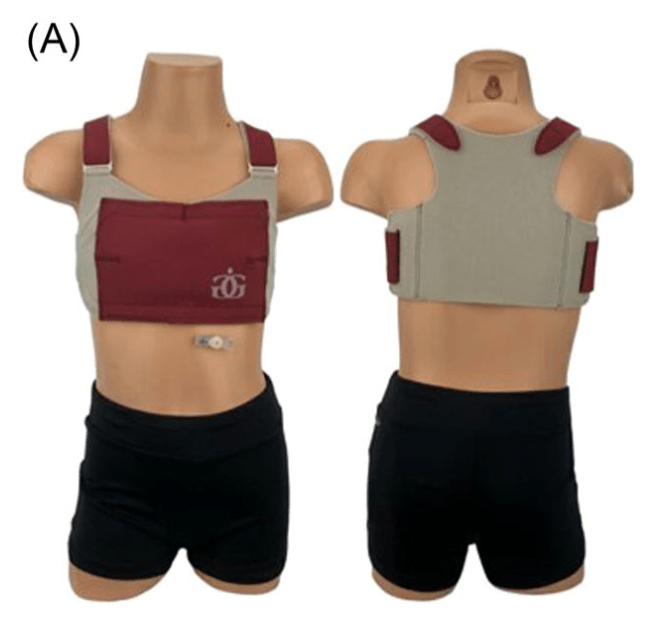

The story behind the inventor is fascinating as well. “Gus Gear” was “founded by Sarah Palya over a decade ago after her son Gus was diagnosed with intestinal failure.” Due to this, he required long-term parenteral nutrition. “Gus also has autism, and his constant movement and activity put him at risk for accidental line trauma and infection. Sarah looked for ways to secure his central line while also allowing him to live as normally as possible, without severely restricting his activity. Finding no solutions, she sat down at her sewing machine and created the very first Gus Gear Central Line Vest to secure his catheter.” The company now makes a similar product to secure GTubes, LOCK 3000 Belt.
My take: This small study supports the use of this product, especially in those prone to line trauma: Gus Gear Central Line Vest® (Dr. Hochman has no commercial ties to this company)
Origin of the expression: “Necessity is the mother of invention“
Related blog posts: