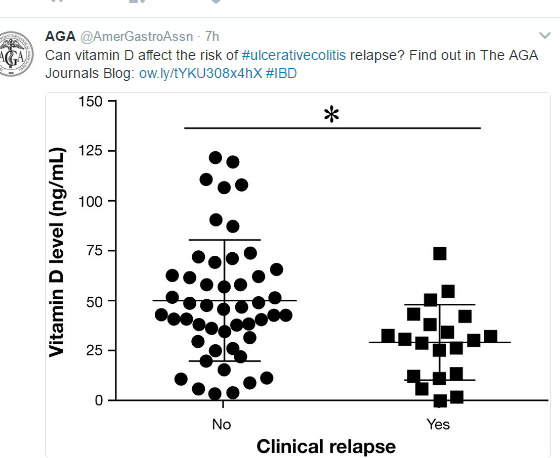
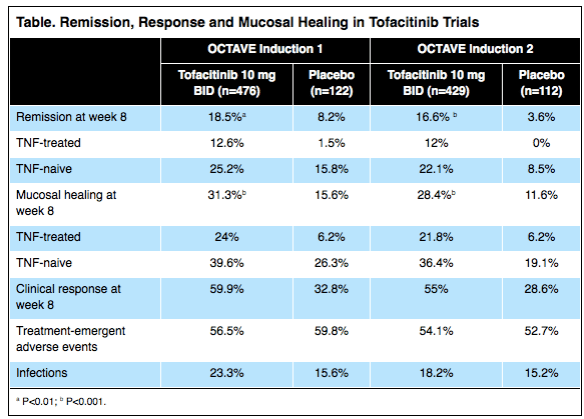
A recent study (S Naviglio et al. Inflamm Bowel Dis 2017; 23: 986-90) confirms that there is a low rate of ocular disease in pediatric inflammatory bowel disease (IBD); in this cohort, half had Crohn’s disease (CD) and half had ulcerative colitis.
In this single center study, 94 children with a median age of 13.4 yrs were offered ophthalmologic examination (2014-2016). None of these patients reported ocular symptoms. The authors assert that 70% had intestinal remission, though 64% had elevated fecal calprotectin levels (>100 mg/kg). Key finding: One patient (1.06%) had ocular finding of uveitis (previously diagnosed prior to study)
The authors indicate that hepatobiliary manifestations, present in 9, were the most common extraintestinal IBD manifestation (EIM). Arthropathy occurred in 8, cutaneous manifestations occurred in 6 and ‘metastatic’ CD occurred in 4.
My take: Ocular disease is an infrequent EIM in pediatric patients with IBD.
Related article: K Hata et al. Inflamm Bowel Dis 2017; 23: 1019-24. This article found that patients with EIMs were more likely to have chronic pouchitis after colectomy for ulcerative colitis. Overall, chronic pouchitis developed in 3.3%, 7.6% and 16.6% at 2, 5, and 10 years respectively. Key finding: preoperative EIM yielded a HR of 4.52.