The Minnesota Health Department Stats are easy to review -here is a link: COVID-19 Vaccine Breakthrough Weekly Update
Here are some recent stats -showing that vaccines reduce the risk of hospitalization and death in all age groups:

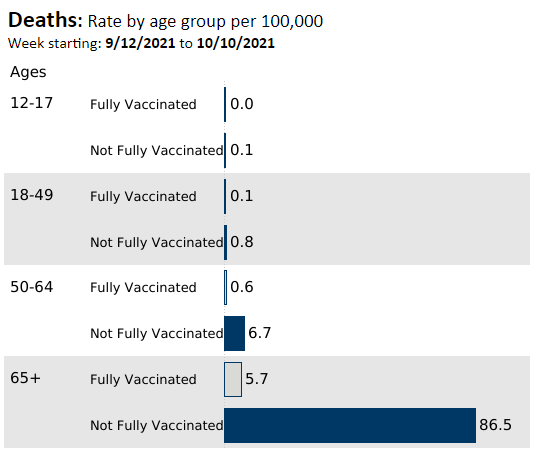



The Minnesota Health Department Stats are easy to review -here is a link: COVID-19 Vaccine Breakthrough Weekly Update
Here are some recent stats -showing that vaccines reduce the risk of hospitalization and death in all age groups:

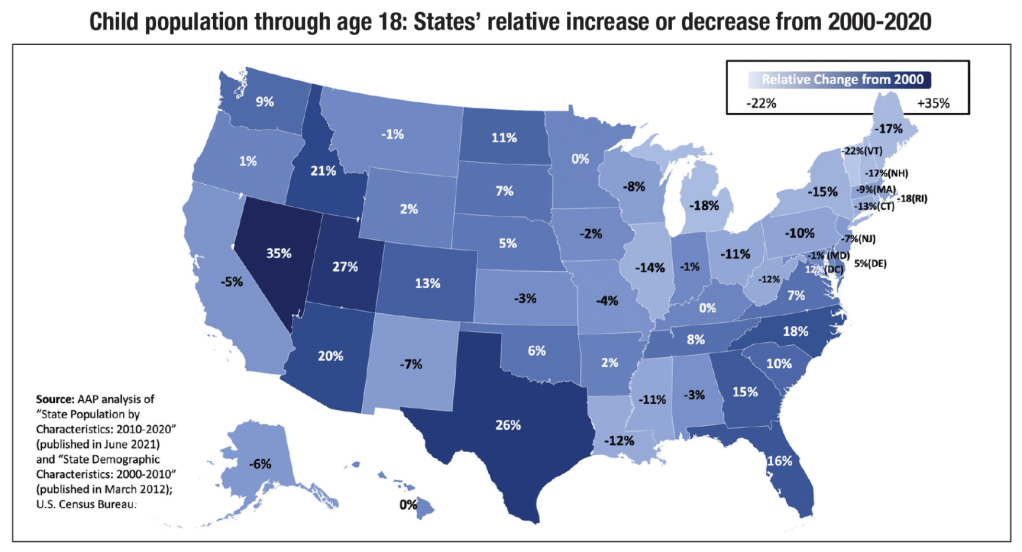
From AAP News (11/1/21): U.S. child population decreasing, becoming more diverse
An excerpt:
E Owen et al. JPGN 2021; 73: e120. Chronic Iron Deficiency Anaemia Caused by Blue Rubber Bleb Syndrome
Related blog post: Something new for blue (BRBNS)

FS Kim et al. JPGN 2021; 73: 610-614. Open Access (PDF): Experience Using Ustekinumab in Pediatric Patients With Medically Refractory Crohn Disease
In this retrospective study with 38 pediatric patients with Crohn’s disease, 34% had stricturing or penetrating disease. Key findings:
My take: Ustekinumab had good efficacy in this group of refractory pediatric patients.
Related blog posts:
E Story et al. JPGN 2021; 73: 604-609. Safety of Venous Thromboprophylaxis With Low-molecular-weight Heparin in Children With Ulcerative Colitis
In this retrospective study with 218 inpatient pediatric patients with active ulcerative colitis, the key findings:
My take: The absolute risk of VTE is low in the pediatric population. This study shows that enoxaparin prophylaxis is NOT associated with increased issues with blood loss. In those with active disease, the presence of CVC and use of steroids are known risk factors and require consideration of, at minimum, nonpharmacologic interventions.
Related blog posts:

ME McSweeney et al. JPGN 2021; 73: 586-591. A Retrospective Review of Primary Percutaneous Endoscopic Gastrostomy and Laparoscopic Gastrostomy Tube Placement
This article is a large retrospective single-center (Boston Children’s) review of laparoscopic gastrostomy (LAP, n=545) compared to percutaneous endoscopic gastrostomy (PEG, n=545) (2010-2015). The authors note a shift in attitude in their institution in favor LAP due to perceived safety advantages (citing references 14-16 -reviewed below).
Key findings:
In the discussion, the authors acknowledge that in their institution, “no differences in total complication rates within the first 6 months were seen.” My view is that the difference in cellulitis needs to interpreted cautiously in a retrospective study. The swelling and erythema around a PEG site in the first days postoperatively can be difficult to determine conclusively if there is cellulitis. With regard to potential cost advantages of LAP, this is affected in the PEG group by whether changing to a skin level device is done at the time of another procedure and whether the case is done in an outpatient surgery center where costs are lower.
My take: There is likely a lower rate of visceral perforation among children who have LAP placement over PEG placement. However, overall, it is uncertain if the complication rate from LAP placement is lower than PEG placement.
Review of some of the cited studies from this article:
S Miyata et al. J Pediatr Surg 2017; 52: 1416-1420. Comparison of operative outcomes between surgical gastrostomy and percutaneous endoscopic gastrostomy in infants (N=1456, Retrospective Study) “Conclusions: When G-tube and PEG were performed as the only procedure throughout a hospitalization in infants and neonates, the two techniques had comparable risks of postoperative complications and mortalities.”
F Sandberg et al. Pediatr Surg Int. 2018; 34 (12):1321-1327. Comparison of major complications in children after laparoscopy-assisted gastrostomy and percutaneous endoscopic gastrostomy placement: a meta-analysis While this meta-analysis with eight studies (n=1500 patients) found a lower risk of complications with LAP compared to PEG, the authors note that “there were no randomized-controlled trials. Overall, the quality of the included studies was determined to be unsatisfactory.” In addition, the results were heavily influenced by one study which reported 20 major complications (death or reoperation within 30 days) among 134 PEG patients.
L Baker et al. J Pediatr Surg 2015; 50: 718-725. A systematic review and meta-analysis of gastrostomy insertion techniques in children In this study, the authors examined 22 studies with 5438 patients that met inclusion criteria. No differences in major complications were noted in studies comparing open versus laparoscopic approaches or open versus PEG. Then, in a subset 10 studies (n=1599 laparoscopic, n=1161 PEG), the authors state that laparoscopic gastrostomy and PEG revealed a significantly increased risk in major complications with PEG (OR 0.29, 95% CI: 0.17–0.51, p < 0.0001). In this study, the most striking difference were 20 cases of visceral perforation with PEG and one case with LAP. Again, the data are limited by the retrospective nature of all but one study. This “suggest that individual-specific and/or institutional-specific factors likely influence outcomes and diminish the generalizability of the presented results.” Cited advantages of LAP: better visualization of site, ability to transfix stomach to abdominal wall, and ability to place primary button.
Related blog posts:

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
AAP News (Nov 2021): First oral pan-genotypic HCV drugs approved for children as young as 3 years
“The Food and Drug Administration (FDA) has approved Epclusa (sofosbuvir and velpatasvir) and Mavyret (glecaprevir and pibrentasvir) for treatment of chronic hepatitis C virus (HCV) infection in pediatric patients as young as 3 years. These products are the first all-oral, pan-genotypic (genotypes 1-6) HCV treatment regimens for pediatric patients 3 years and older…Both products are available as oral tablets and as newly approved oral pellets in dosing strengths suitable to accommodate the recommended weight-based dosing in pediatric patients.”
From HCVGuidelines.org (updated September 2021):

The following images are from Abbvie patient-provider handouts. I do not receive any funding from any pharmaceutical company but think these instructions are useful.



Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
J David et al. JPGN 2021; 73: 599-603. Unexpected Poor Growth in Pediatric Patients on Food-based Enteral Therapy: Case Series and Suggested Practice Changes
In this chart review with five patients, weight loss occurred when patients received only a fraction of their feeds due to feeding pump inaccuracy.
Related blog posts:

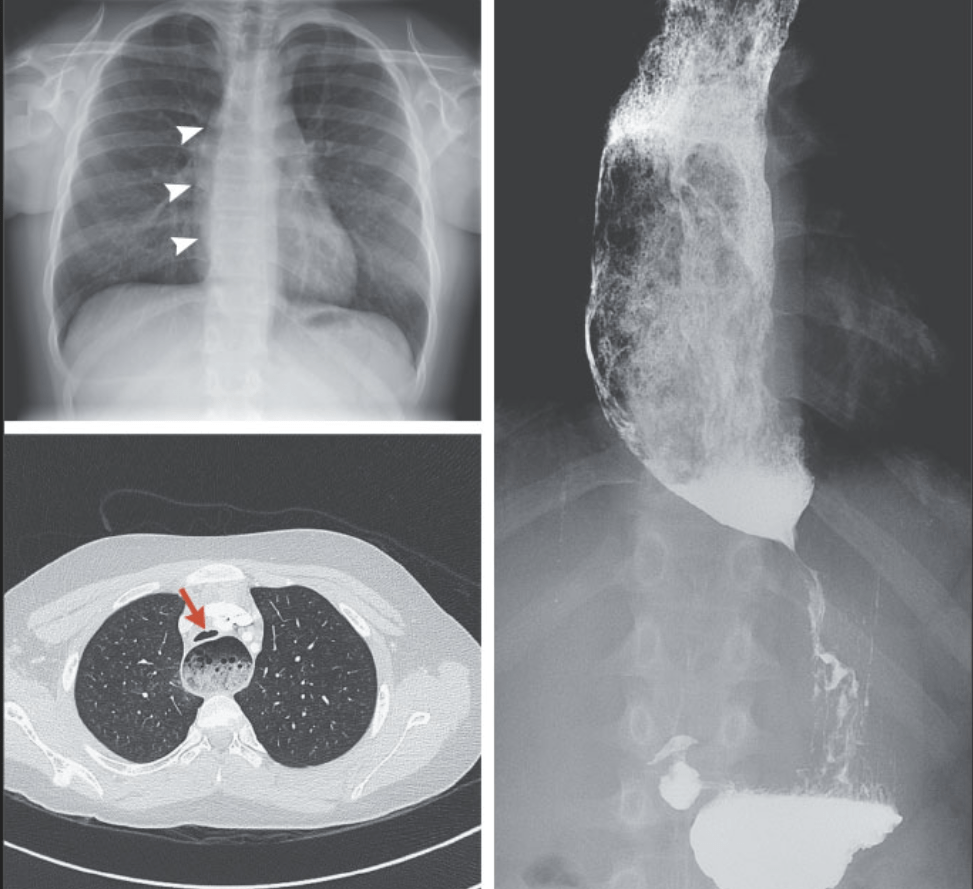

S Woodhull, DR Segar N Engl J Med 2021; 385:e64. DOI: 10.1056/NEJMicm2108193
Related blog posts:
H Yanai et al. The Lancet 2021; The Crohn’s disease exclusion diet for induction and maintenance of remission in adults with mild-to-moderate Crohn’s disease (CDED-AD): an open-label, pilot, randomised trial https://doi.org/10.1016/S2468-1253(21)00299-5
In this open-label trial of adults with mild-to-moderate biologic naive Crohn’s disease, key findings:
My take: Dietary therapy may be effective option for motivated adult patients with Crohn’s disease.
Related blog posts:

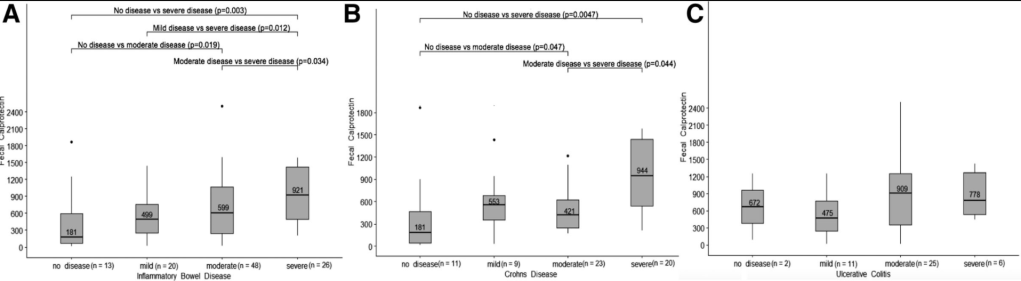
E Crawford et al. JPGN Reports 2021; 22 – Issue 4 – p e129. Open Access: Association of Fecal Calprotectin With Endoscopic and Histologic Activity in Pediatric Inflammatory Bowel Disease
This retrospective study used data from 331 patients (n=107 with IBD). Fecal calprotectin (FC) was done between 30 days and 1 day before colonoscopy.
Key findings:
My take: Calprotectin levels are particularly helpful as a screen for IBD (probably using threshold of at least 120) and its use to monitor clinical response. This study shows it has some utility in predicting disease severity.