Author Archives: gutsandgrowth
TARGET Study: Does Energy-Dense Nutrition Improve Outcomes in the Critically Ill
A recent double-blind randomized study (NEJM 2018; 379: 1823-44) examined the outcomes of 3957 adult patients undergoing mechanical ventilation who received either a 1.5 kcal formula or 1.0 kcal formula for provision of enteral nutrition.
Key Findings:
- While the volume of formula was similar, the 1.5 kcal group received a mean of 1863 kcal/day compared to 1262 kcal/day for the 1.0 kcal group.
- Yet, this did not translate into a survival benefit. By day 90, 26.8% of the 1.5 kcal group had died compared with 25.7% of the 1.0 kcal group (RR 1.05, 95% CI 0.94-1.16, P=0.41)
- Higher caloric delivery did not affect survival, receipt of organ support, duration of hospital stay, the incidence of infective complications or adverse events.
- Regurgitation was more common in the 1.5 kcal group: 18.9% vs 15.7%, RR 1.20, 95% CI 1.05-1.38)
- The 1.5 kcal group were more likely to receive promotility medications (47.4% vs 39.6%, RR 1.20)
- The 1.5 kcal group were more likely to receive insulin (55.8% vs 49.0%, RR 1.14)
In their discussion, the authors note that only 2% of patients had a BMI less than 18.5; thus, their cohort is unable to determine whether these patients could benefit from increased calories.
My take (borrowed in part from authors): “Increasing energy intake with the administration of energy-dense enteral nutrition did not affect survival among critically ill adults.” These types of studies are important in challenging assumptions that meeting calorie needs (with enteral or parenteral nutrition) will improve outcomes in hospitalized patients–though, this may be true in some populations.
Related blog posts:
- Detrimental Effect of Early Parenteral Nutrition in Critically-Ill Children The concept of providing early aggressive nutrition is NOT supported by this study
- Early versus late parenteral nutrition in critically ill adults
Does Screening for Hepatocellular Carcinoma Improve Outcomes in Patients with Cirrhosis?
Despite widespread recommendations to screen patients with cirrhosis for hepatocellular carcinoma (HCC), a recent study (AM Moon et al. Gastroenterol 2018; 16: 1777-85) found “No Association Between Screening for Hepatocellular Carcinoma and Reduced Cancer-Related Mortality in Patients with Cirrhosis.” The title of the study did not make sense to me based on previous publications that have noted increased risk of HCC in patients with cirrhosis and the presumption that screening would allow effective interventions to prevent death due to HCC. So I looked at the study a little closer:
Background/Methods: The authors utilized a matched case-control study within the U.S. Veterans Affairs health care system to determine whether ultrasonography (US) or alpha-fetoprotein (AFP) screening was associated with decreased cancer-related mortality.
They identified 238 patients with cirrhosis who died of HCC between 2013-2015 –all of whom had a diagnosis of cirrhosis at least 4 years before the diagnosis of HCC. Then, they matched them with a control patient with cirrhosis who did not have HCC and had been identified at least 4 years prior to matched case’s HCC.
Key findings:
- There was no significant difference between the cases and the controls in the proportions who underwent screening:
- For U/S screening: 52.9% cases and 54.2% for controls.
- For AFP (serum) screening, 74.8% vs 73.5% respectively.
- For either U/S or AFP screening, 81.1% vs 79.4%.
- For both U/S and AFP screening, 46.6% vs 48.3% respectively.
- Table 4 provides odds ratios and adjusted odds ratios for the cases compared to controls. The Adjusted Odds ratios for U/S 0-4 years before index case was 0.95, for AFP 1.08, and for either U/S or AFP 1.11.
The authors found that HCC screening with U/S and/or AFP was not associated with decreased risk of HCC-related mortality.
In their study, the authors note that most studies on HCC screening have been observational which have numerous limitations including lead-time biases (which can overestimate the benefits of screening) and patient selection. Two randomized controlled trials reached conflicting conclusions; these trials were conducted in China where HCC is mainly associated with hepatitis B infection.
The authors point out that liver societies like AASLD and EASL have recommended U/S every 6 months with or without AFP measurements for HCC surveillance in patients with cirrhosis. However, non-liver societies have NOT “endorsed HCC screening because of the lack of high-quality data.” Neither the US Preventive Services Task Force nor the American Cancer Society make recommendations for HCC screening. And, “the National Cancer Institute found no evidence that screening decreases mortality from HCC but did find evidence that screening could result in harm.”
Strengths of this study:
- All VA patients have access to medical care; this limits bias due to access to HCC screening
- The matched-case control design with random controls across a system that delivers care to 8 million veterans across the country indicates that the findings are likely “typical of community-based settings” and likely to yield “estimates of the impact of screening …[that] approximates the results that would be expected from a randomized controlled trial”
Why Have Previous Studies Indicated that HCC Screening is Worthwhile?
- According to the authors, even though HCC detected by screening is on average detected at an earlier stage than those detected due to symptoms, “this does not prove that screening leads to earlier detection. Another explanation is that screening is more likely to identify slow-growing tumors, which have a lower stage, and more likely to miss the fast-growing tumors, which are identified at a higher stage by symptoms.”
- “It is possible that the HCCs most likely to lead to death are the HCCs least likely to be identified by current screening modalities at an early stage.”
- In addition, “whether early treatment for HCC in patients with cirrhosis leads to a decrease in case fatality is questionable.” Patients who receive surgical resection or locoregional treatments remain at risk for recurrent HCC, new HCC and progressive liver dysfunction. While liver transplantation can cure HCC and cirrhosis, only a “small minority of patients with HCC undergo liver transplantation.” In 2012, only 1,733 patients received liver transplantation for HCC out of a reported 24,696 incident cases.
My take: This study offers a lot of insight regarding HCC screening and questions its usefulness, though I doubt this study will change how most hepatologists practice.
Related blog posts:
- HBV-related Pediatric HCC: 8 needles in 4 haystacks
- How strong is the case for HCC screening?
- Looking for trouble
- Increasing Incidence of Hepatocellular Carcinoma in the U.S. | gutsandgrowth
PEG Placement in Cystic Fibrosis
A small retrospective study (RT Khalaf et al.NCP 2018; LINK: doi.org/10.1002/ncp.10219) showed that PEG placement was associated with a trend (not statistically significant) towards improved lung function in children with cystic fibrosis (CF). Reference from Kipp Ellsworth twitter feed. There were 20 patients who had PEG placed compared to 40 patients who did not.
Findings:
- BMI percentile increased per month for those with PEG (0.51, 95% confidence interval (CI) = −0.05–1.08, P = .08), but decreased for those without PEG (−0.03, 95% CI = −0.33–0.28, P = .86); however, the difference (0.54; 95% CI = −0.10–1.18, P = .10) was not statistically significant.
- FEV1 change with time showed a decrease for patients with PEG (−0.04; 95% CI = −0.30–0.22, P = .74) and those without PEG (−.22; 95% CI = −0.45–0.01, P = .06). Although the FEV1 decrease for those without PEG was higher than those with PEG, the difference between the groups was not statistically significant (0.18; 95% CI = −0.17–0.52, P = .32)
My take: While the differences are not statistically-significant, this study indicates that PEG placement is NOT detrimental to lung function in CF and may be beneficial.
Related blog posts:
- Why Do Canadians with Cystic Fibrosis Live Longer?
- Gastrostomy Tubes for Children with Cystic Fibrosis
- Lumacaftor-Ivacaftor for Cystic Fibrosis
- Ivacaftor for Cystic Fibrosis | gutsandgrowth
- CFTR Modulators for Cystic Fibrosis
- FDA Expands Use of Cystic Fibrosis Medication | gutsandgrowth
- “Origins of Cystic Fibrosis Lung Disease”
Fish Oil for Ulcerative Colitis?
A small randomized, double-blind, placebo-controlled study (E Scaioli et al. Clin Gastroenterol Hepatol 2018; 16: 1268-75) examined the use of Eicosapentaenoic acid-Free Fatty Acid Form (EPA-FFA) a component of n-3 fish oil for patients with ulcerative colitis UC).
From 2014-2016, the investigators enrolled 60 patients who had partial Mayo score <2 and fecal calprotectin >150 mcg/g who had been receiving stable therapy for at least 3 months. Then they were randomized 1:1 to receive EPA 1000 mg BID or placebo for 6 months.
Key findings:
- 19 of 30 (63%) EPA-FFA group compared with 4 of 30 (13.3%) of placebo-treated group had achieved the primary endpoint of a 100-point reduction in fecal calprotectin at 6 months. OR 12.0, P<.001
- The secondary endpoint of clinical remission was noted in 23 of 30 (77%) in the EPA-FFA group compared with 15 of 30 (50%), OR 3.29, P=.035)
- No serious adverse effects were reported.
Limitations:
- Small number of patients from a single center
- Short follow-up
- In those without clinical relapse, a followup colonoscopy was not performed
My take: In this study EPA-FFA was associated with lower calprotectin and higher rates of remaining in remission. More data are needed.
Related blog posts:
- Spice it up? Curcurmin for ulcerative colitis
- Herbal Medicines for IBD and IBS | gutsandgrowth
- The Search for a Dietary Culprit in IBD | gutsandgrowth
Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
How to Rectify the Problem of Missed Impactions
Blog Note: This blog site was recently upgraded such that blog followers should not see wordpress.com ads and the domain name is officially gutsandgrowth.com rather than gutsandgrowth.wordpress.com.
A recent study (S Pradhan, B Jagadisan. JPGN 2018; 67: 570-5) describes the yield of digital rectal exam (DRE) for detecting impaction in the setting of functional constipation.
Methods: In this prospective, observational study with an initial cohort of 233 (from a tertiary center) with a history of suggestive of constipation, the authors identified 169 who satisfied Rome III criteria for functional constipation. After excluding 48 who had impactions detected without DRE and another 24 who had DRE not performed (24 due to fissures, 2 due to recent enema, and 2 due to parental preference), there were 93 who had DRE.
Key finding: “Children satisfying Rome III criteria without requiring digital rectal examination …have impaction detectable only by DRE in 30.1%” (28 of 93).
My take: Since treatment is modified in those with an impaction, DRE remains important part of the examination in children with constipation or symptoms attributed to constipation.
Related posts:
- Don’t Let the Chief of Staff Review this Study on Constipation
- Stimulants for constipation
- Diagnostic tests hardly ever help patients poop
- What’s Wrong with Ordering an AXR for Constipation in the ER …
- Miralax Safety | gutsandgrowth
- Updated Pediatric Expert Constipation Guidelines | gutsandgrowth
- AGA Constipation Guidelines | gutsandgrowth
- “Simple Remedies for Constipation” | gutsandgrowth
- Reliability of colonic manometry | gutsandgrowth
- …ER Performance for Suspected Constipation | gutsandgrowth
Avatrombopag Reduces Need for Platelet Transfusion in Patients with Chronic Liver Disease
Briefly noted: N Terrault et al. Gastroenterol 2018; 155: 705-18. In two trials, ADAPT-1 and ADAPT-2, the use of avatrombopag, a thrombopoietin receptor agonist, was superior to placebo in increasing platelet counts and reducing need for platelet transfusion for bleeding related to procedures.
Romaine Lettuce and E coli Outbreak
According to the CDC update (Nov 26, 2018) –Outbreak of E. coli Infections Linked to Romaine Lettuce: “43 people infected with the outbreak strain of E. coli O157:H7 have been reported from 12 states. A list of the states and the number of cases in each can be found on the Map of Reported Cases page.
Illnesses started on dates ranging from October 8, 2018 to October 31, 2018. Ill people range in age from 1 to 84 years, with a median age of 25. Sixty-nine percent of ill people are female. Of 38 people with information available, 16 (42%) have been hospitalized, including one person who developed hemolytic uremic syndrome, a type of kidney failure. No deaths have been reported.”
“CDC is advising that U.S. consumers not eat and retailers and restaurants not serve or sell any romaine lettuce harvested from the Central Coastal growing regions of northern and central California. If you do not know where the romaine is from, do not eat it.”
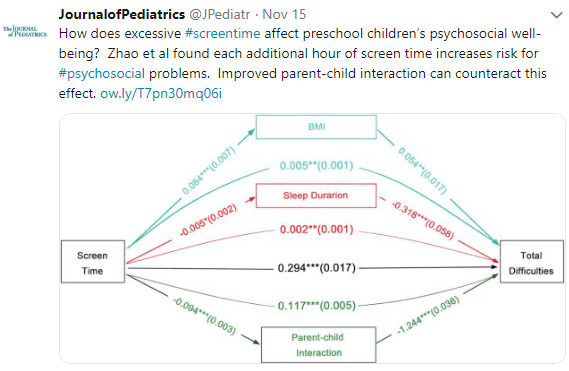
Interesting Study -Detrimental Dose-Response of Screen Time
In 1995, there was a Batman movie, “Batman Forever,” in which one of the central villains, the Riddler, places these brainwave devices over the TVs to gain control of Gotham. The sad part, according to a recent study (J Zhao et al. J Pediatr 2018; 202; 157-62) is there is no need to add a brainwave device to a TV set. Excessive screen time alone is quite detrimental.
In this cross-sectional survey in Shanghai with more than 20,000 children, the authors found the following:
- Mean screen time for preschool children was 2.8 hrs per day. 78.6% exceeded 1 hour per day and 53% exceeded 2 hrs per day.
- Every additional hour of screen time was associated with increased risk for poor psychosocial well-being; this effect on well-being had a number of mediators including reducing parent-child interaction as well as increased body mass index and reduced sleep duration.
My take: This study reinforces the consequences of excessive screen time –now, the hard part — how to translate these findings into reduction in screen time.
Related blog posts:
- Skinnier TVs and Heavier Children
- Sleep Duration and Subsequent Obesity | gutsandgrowth
- 7 Ways Parents Can Influence Risk of Obesity | gutsandgrowth
- Staggering cost of obesity | gutsandgrowth
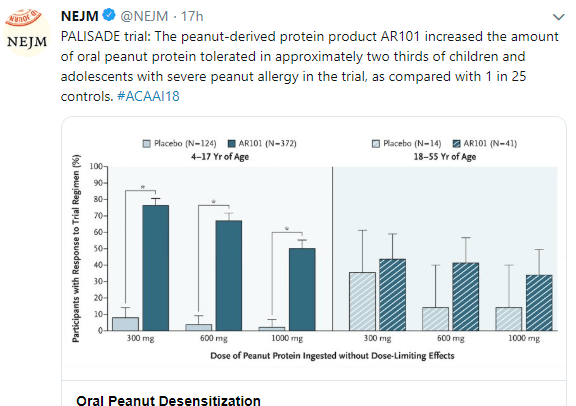
New Strategy to Overcome Severe Reactions to Peanuts
A recent study (N Engl J Med 2018; 379:1991-2001) showed that
Link to abstract: AR101 Oral Immunotherapy for Peanut Allergy
Methods: Participants with an allergic response were randomly assigned, in a 3:1 ratio, to receive AR101 (a peanut-derived investigational biologic oral immunotherapy drug) or placebo in an escalating-dose program.
Conclusions: In this phase 3 trial of oral immunotherapy in children and adolescents who were highly allergic to peanut, treatment with AR101 resulted in higher doses of peanut protein that could be ingested without dose-limiting symptoms and in lower symptom severity during peanut exposure at the exit food challenge than placebo.
Related blog posts: