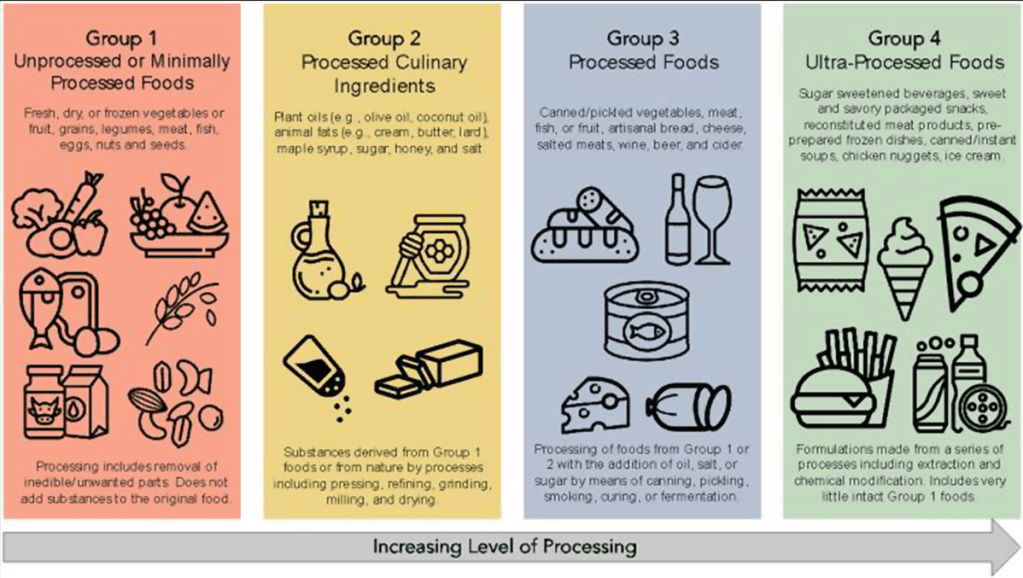
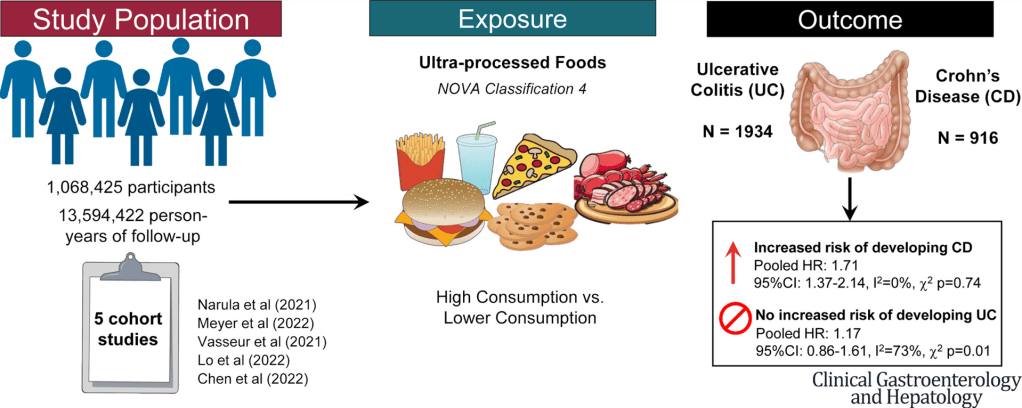
BN Limketkai et al. Clin Gastroenterol Hepatol 2023; 21: 2508-2525. Open Access! Dietary Interventions for the Treatment of Inflammatory Bowel Diseases: An Updated Systematic Review and Meta-analysis
This was a systematic review of prospective controlled trials (n=27) of solid food diets for the induction or maintenance of remission in IBD.
Key findings:
- For induction of remission in Crohn’s disease (CD), the Mediterranean diet was similar to the Specific Carbohydrate Diet (low certainty of evidence), and partial enteral nutrition (PEN) was similar to exclusive enteral nutrition (very low certainty of evidence).
- PEN reduced risk of relapse (very low certainty of evidence), whereas reduction of red meat or refined carbohydrates did not (low certainty of evidence).
- For ulcerative colitis, diets were similar to controls (very low and low certainty of evidence).
My take: Most of the dietary treatments for IBD have low to very low certainty of evidence regarding their effectiveness. Dietary changes are very likely to be helpful but more studies with rigorous endpoints are still needed.
Related blog posts:
- Outcomes with Enteral Nutrition
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- No Solid Conclusions” for Alternative/Complementary Therapies for Inflammatory Bowel
- More Evidence That A Proinflammatory Diet May Increase the Risk of Crohn’s Disease | gutsandgrowthDisease | gutsandgrowth
- Trial by Diet Approach for Crohn’s Disease in Children (this blog post has link to 16 other dietary therapy posts)
- Can IBD Be Treated with Diet Alone?
- Dietary Therapy for Inflammatory Bowel Disease –Useful Update
- Top Lecture: Enteral Nutrition for Crohn’s Disease
- Head-to-Head: Nutritional Therapy versus Biological Therapy in Pediatric Crohn’s Disease | gutsandgrowth
- Ultraprocessed Food and the Risk of Inflammatory Bowel Disease
- Risk Factors for Inflammatory Bowel Disease: Ultra-Processed Food (Part 1)