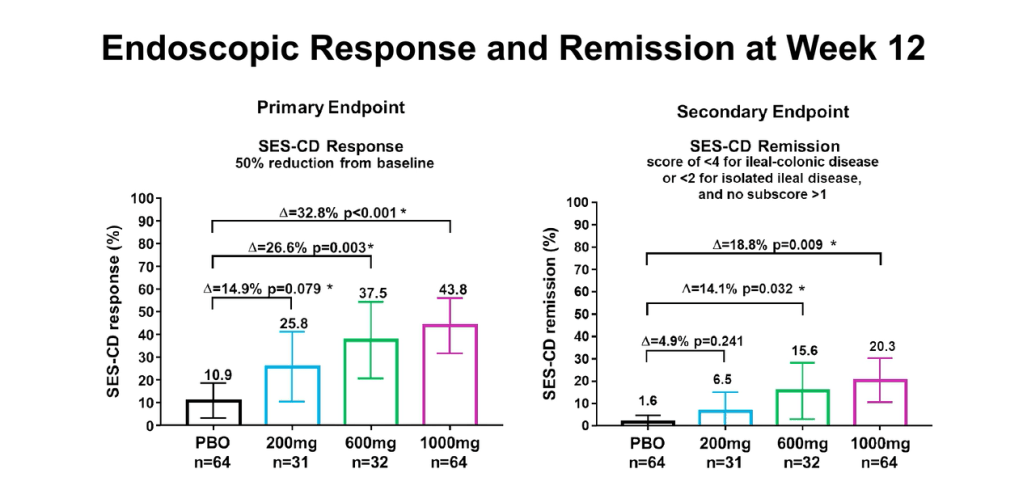
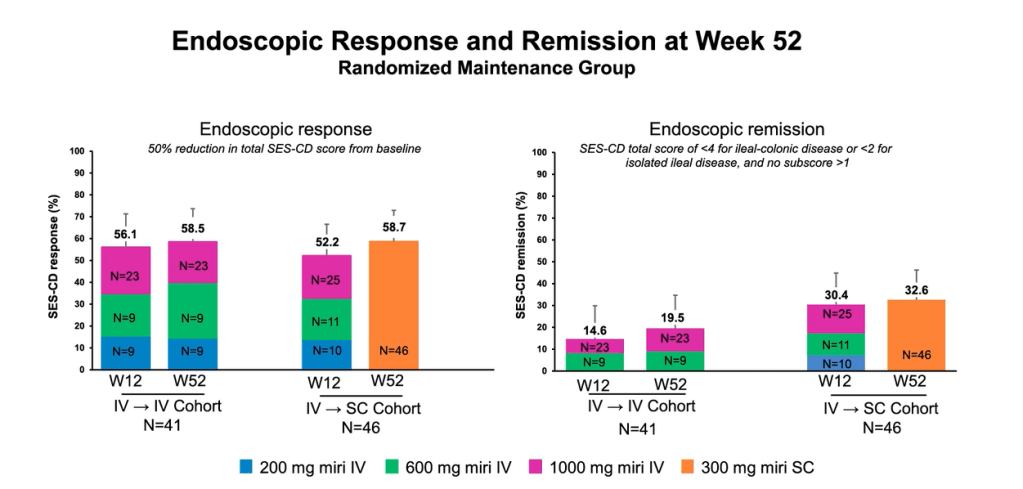
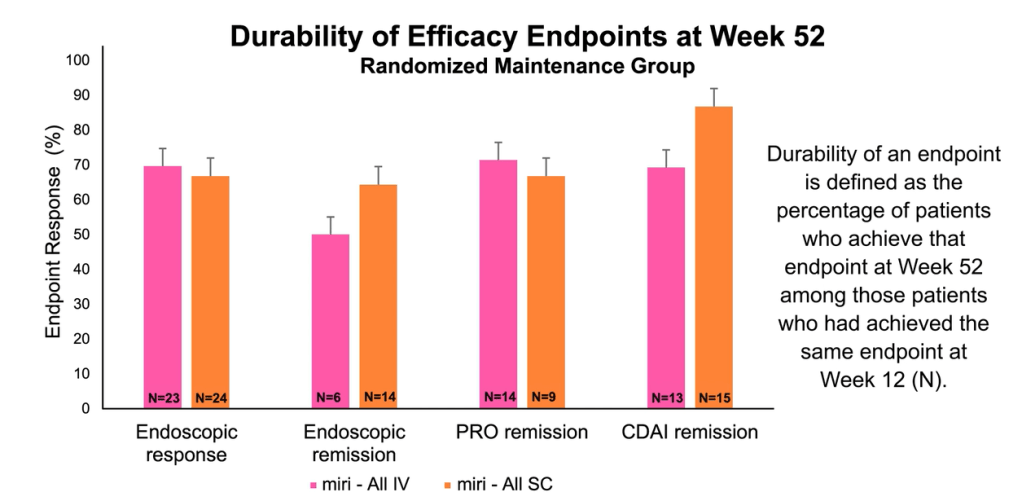
MJ Dushnicky et al. JPGN 2021; 73: 626-629. Pediatric Patients with a Dual Diagnosis of Inflammatory Bowel Disease and Chronic Recurrent Multifocal Osteomyelitis
This article describes a retrospective review of seven patients with a dual diagnosis of inflammatory bowel disease (IBD) and chronic recurrent multifocal osteomyelitis (CRMO). In their cohort 4 of 6 were receiving anti-TNF therapy at the time of CRMO diagnosis. Misleading statements from this article:
- The triad of IBD, CRMO and psoriasis has not been reported previously to their knowledge
- “It seems unlikely that anti-TNF-alpha therapy would promote its [CRMO] development”
In JPGN Reports (not available on pubmed), Cordesse et al (JPGN Reports; November 2020 – Volume 1 – Issue 2 – p e007) identified the association of IBD, CRMO and psoriasis; in addition, they identified a paradoxical reaction to anti-TNF-alpha therapy; in this case series of three patients, anti-TNF-alpha therapy triggered CRMO and stopping anti-TNF-alpha therapy led to resolution of CRMO in two of the cases.
In a response to a letter to the editor (Hochman JA. JPGN 2022; DOI: 10.1097/MPG.0000000000003407. Faulty Information Regarding CRMO and IBD), Dushnicky et al (DOI: 10.1097/MPG.0000000000003433) note that JPGN Reports is not available on Pubmed; however, the articles that have described this association are near the top of a google search if one looks for “IBD, CRMO and Psoriasis.” Interestingly, in their response to the letter to the editor, the authors did not amend their claim that anti-TNF therapy is unlikely to promote CRMO despite being furnished with information showing that it can. In my view, the situation with CRMO is similar to psoriasis which can be treated with anti-TNF therapy and can paradoxically be caused by anti-TNF agents as well.
My take:
- CRMO is important to recognize due to its association with IBD and to realize that antibiotics are not an effective treatment.
- Anti-TNF-alpha agents can cause CRMO in some patients.
- In 2022, a web browser search (eg Google), in addition to Pubmed, is probably worthwhile when claiming that this is the first case of xyz ‘to our knowledge.’