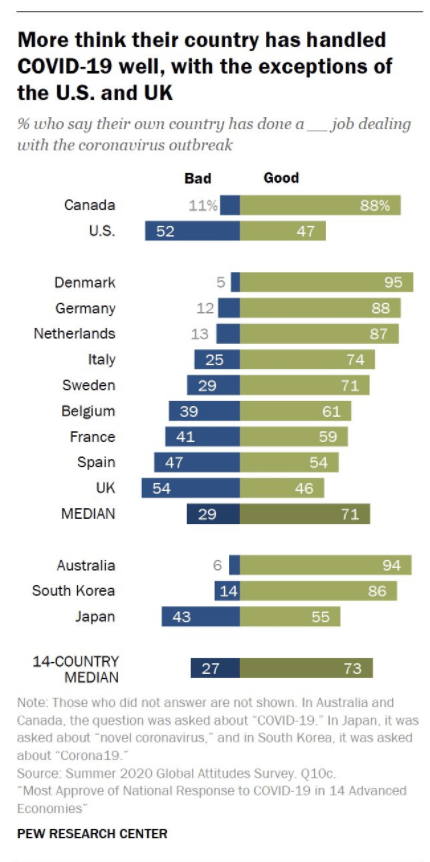
From Annals of Internal Medicine 2020 https://doi.org/10.7326/M20-5352: J Blackburn et al. Full Text: Infection Fatality Ratios for COVID-19 Among Noninstitutionalized Persons 12 and Older: Results of a Random-Sample Prevalence Study
Background: Mortality rates have been calculated from confirmed cases, which overestimates the infection fatality ratio (IFR). To calculate a true IFR, population prevalence data are needed from large geographic areas where reliable death data also exist.
Results: The Table below suggests IFR of 0.01% for those <40, 0.12% for those 40-59, and 1.71% for those ≥60 in noninstitutionalized persons. The Table indicates nearly a 3-fold increase risk in Non-White persons. Whites had an IFR of 0.18%; non-Whites had an IFR of 0.59%. Also, I think the Table incorrectly suggests that Females have a higher IFR than Males (but the numbers suggest that they are equivalent).
From The New Yorker, Atul Gawande: We Can Solve the Coronavirus-Test Mess Now—If We Want To
This is a lengthy article which describes some of the mistakes that we’ve made with testing, some of the technical details with various tests, pooled testing, at-home testing, wastewater testing, and how to fix testing (including assurance testing) to gain control of this pandemic.
An excerpt:
We could have the testing capacity we need within weeks. The reason we don’t is not simply that our national leadership is unfit but also that our health-care system is dysfunctional….
In the United States, getting a test is anything but easy…[And] through early August, results routinely took four days or more, making the tests essentially useless.
“Assurance testing” has been required by countries such as Iceland, France, and Germany for travellers from abroad in order to avoid a mandatory two-week quarantine