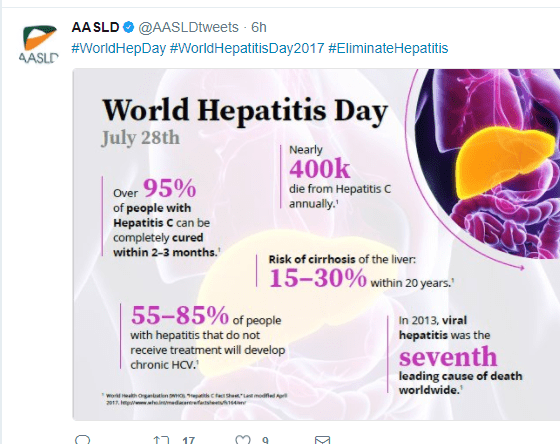In case you missed this yesterday –World Hepatitis Day July 28, 2017
Predicting Future Liver Disease with GGT Levels in Biliary Atresia Patients
A recent study (AJ Freeman, VL Ng, S Harpavat, A Hrycko, Z Apted, P Bulut, T Leong, SJ Karpen. Clin Gastroenterol Hepatol 2017; 15: 1133-35) describes the predictive value of γ-glutamyltransferase (GGT) in predicting thrombocytopenia/portal hypertension among biliary atresia patients.
In this retrospective study from three centers who had followup for at least 4 years, GGT values at 2 years of age were examined among biliary atresia patients (n=46) who continued with their native liver.
Key findings:
- GGT ≥100 U/L had a predictive positive relationship with thrombocytopenia at 4, 5, and 6 years of age. Patients with elevated GGT had lower platelet count (160 vs. 211) and their values continued to decline. GGT ≥100 U/L at 2 yrs predicted thrombocytopenia (<150) at age 4 with a sensitivity of 0.88, specificity of 0.57.
- Patients with normal GGT values had “essentially stable platelet counts over the next 4 years.” GGT <100 U/L at 2 yrs predicted a low risk of thrombocytopenia with negative predictive value of 0.89, 0.92, and 0.93 at age 4, 5, and 6 respectively.
My take: This study quantitates a useful point –patients with biliary atresia and elevated GGT values are likely to develop evidence of portal hypertension.
Hazardous Toys: Jarts and Magnets
I had completely forgotten about Jarts until reading a recent editorial by Athos Bousvaros (J Pediatr 2017; 186: 6-7). He succinctly describes how these lawn darts were ultimately removed from the market primarily due to the advocacy of a father who became a strong advocate after the death of his daughter.
A more complete description of the effort to remove Jarts -from Mental Floss website: How One Grieving Father Got Lawn Darts Banned
Dr. Bousvaros, in commentary on a study on high-powered (neodymium) magnets (Rosenfeld D et al. J Pediatr 2017; 186: 78-81) describes the similarities between these magnets and the jarts. Both have caused catastrophic injuries and death. However, the recent removal of these magnets from the market was overturned. There is no national tracking system for magnet ingestions in U.S. or Canada. However, the referenced study demonstrated a dramatic reduction in medical/surgical procedures in 2014-2015 (n=10) when a ban was placed compared to 2011-2012 (n=29).
For U.S physicians, all we can do currently is to report to the CPSC (Consumer Product Safety Commission) all magnet-related injuries and to publicize the dangers of these hazardous products. To report: go to CPSC website (link: CPSC website) and “report an unsafe product” on the right side of the page.
Related blog posts:
Big Study of Primary Sclerosing Cholangitis
This blog has reviewed multiple publications on primary sclerosing cholangitis (see blog posts below). Now, a study from 37 centers with 7121 patients with PSC has been published: TJ Weismuller et al. Gastroenterol 2017; 152: 1975-84. Given the relative infrequency of PSC, this retrospective report offers more insight into the predictors of the clinical course of PSC.
Key points:
- Most of the patients in the study had large duct PSC (89.8%); 3.6% had small duct disease and 6.6% had overlapping PSC/autoimmune hepatitis.
- Mean age of cohort at diagnosis was 38.5 yrs.
- 70% of PSC patients developed IBD with ulcerative colitis (UC) about 5-times more common than Crohn’s disease.
- 37% of patients met the primary endpoint of either liver transplantation or death
- Individuals with small duct PSC had a favorable outcome; only one of 254 (0.4%) developed cholangiocarcinoma (CCA). Risk of primary endpoint was much lower in small duct PSC compared with classical PSC with an adjusted hazard ratio of 0.23.
- Individuals with PSC/AIH variant also had a reduced risk of primary endpoint compared with classical PSC with an adjusted hazard ratio of 0.73.
- Overall, CCA occurred in 594 patients (8.3%); the incidence of CCA changed markedly with the age of the patient. In the youngest group (<20 years), the rate was 1.2 per 100 patient-years, it was 6.0 in 21-30 yr-olds, 9.0 for 31-40 yr-olds, 14.0 fr 41-50 year olds, 15.2 for 51-60 yr-olds, and 21.0 per 100 patient-years in those older than 60 years.
- The absence of IBD, particularly UC, was associated with a lower risk PSC clinical course. Patients with UC had increased liver disease progression compared with patient’s with Crohn’s disease, with a HR of 1.56.
- The median transplant-free survival time was 14.5 years; the estimated survival was approximately 21 years in the entire cohort
It is noted that an important limitation is that the cohort is from specialist centers and may not reflect a more typical population-based cohort; that is, this patient population is likely to be severely affected.
My take: Patients with small-duct PSC have a much lower risk of disease progression.
Related blog posts:
- Should we care about subclinical PSC? (This post has links to others related to PSC)
- PSC -Natural History Study (pediatric)
Is there a link between Eosinophilic Esophagitis and Celiac Disease?
Probably most pediatric gastroenterologists have seen patients who underwent endoscopy for celiac disease and found out that the patient had both celiac disease and esophageal eosinophilia. Whether the esophageal eosinophilia should be classified as eosinophilic esophagitis (EoE) is based in part on whether one concludes that the EoE is a separate disorder and unrelated to the celiac disease.
One useful retrospective study on this topic (S Hommeida et al. JPGN 2017; 65: 58-63) examines the association between celiac disease and EoE. Key findings:
- Among a cohort of 10,201 children seen at the Mayo clinic, 595 were considered to have EoE and 546 had celiac disease.
- Only 10 patients had both celiac disease and EoE.
- The risk of EoE was not increased in children with celiac disease compared to those without celiac disease (odds ratio 0.29). The prevalence of EoE in children with celiac disease was 1.8% whereas the prevalence among all children undergoing endoscopy was 5.8%.
- 4 of 10 children treated only with GFD clinically improved (no followup histology)
Limitations:
- The diagnosis of EoE was not clear in this study. As noted in the associated editorial (pg 1-2), “the use of a high-dose proton pump inhibitor at the time of initial diagnosis is not mentioned.”
- Overall, the number of patients with both EoE and celiac disease was small. Thus, a much larger study could be necessary to prove the lack of an association.
My take: This study suggests that there is not an association between EoE and celiac disease. Some patients with both disorders will respond to a gluten free diet, whereas some will require additional treatment directed at EoE.
Related study: T Wallach et al. JPGN 2017; 65: 64-8. This retrospective study showed poor adherence to biopsy guidelines in EoE and celiac disease. Among 9171 children, 8% were biopsied in accordance with 2007 AGA EoE consensus recommendations and 35% in accordance with 2006 AGA celiac guidelines. Higher detection rates were observed among patients who had higher adherence to diagnostic guidelines. With both diseases, obtaining sufficient number of biopsies is key; and with celiac disease, obtaining biopsies from duodenal bulb as well as distal duodenum is recommended.
AGA Blog: What are the complications of PPI Therapy?
AGA Journals Blog: What are the complications of proton pump inhibitor (PPI) Therapy?
The blog post reviews a recent article on PPIs and potential complications.
An excerpt:
A review article from Michael F. Vaezi et al discusses potential adverse consequences of proton-pump inhibitor (PPI) therapy in the July issue of Gastroenterology…(2017; 153: 35-48). The authors discuss overzealous conclusions based on weak associations that have caused widespread alarm, leading to inappropriate discontinuation of a medicine that is needed for an established disease process. They present absolute and relative risks for adverse effects associated with long-term use of PPIs…
Vaezi et al review the consistency of proposed associations with PPI use and the time period between the PPI exposure and outcome, and the effects of different doses. They provide guidance for methodologies of future studies.
The review article concludes that PPIs have revolutionized the management of patients with GERD and patients at risk of upper gastrointestinal ulceration and bleeding from aspirin or NSAIDs. However, many patients receive PPIs unnecessarily for conditions or symptoms for which they would not have been expected to provide benefit… Vaezi et al state that, as always, PPIs should be given in the lowest effective dose, for the shortest possible time.
They add that much of the evidence linking PPI use to serious long-term adverse consequences is weak and insubstantial. It should not deter prescribers from using appropriate doses of PPIs for appropriate indications.
Full text of original article: Complications of Proton Pump Inhibitor Therapy
Table 6 lists the strengths of the findings along with other Hill Criteria to assess all of the proposed complications. The vast majority of potential complications have “weak” proof; the exceptions include bacterieal enteric infections/Clostridium difficile infection which have moderate strength of evidence and and fundic gland polyps which have high strength of evidence.
My take: This study and the associated AGA Journals blog post indicate that most of the reports of complications associated with PPI remain unproven and are based on weak evidence.
Image Only: Put Only Food in Your Mouth
In addition to being a choking hazard, some fidget spinners have button batteries to light up. Button batteries are particularly dangerous if lodged in the esophagus.
Related blog post: Foreign bodies in children -expert guidance
Briefly Noted: Outpatient Liver Biopsy
A small retrospective study (R Bolia et al. JPGN 2017; 65: 86-88) with 497 patients (626 biopsies) found that all complications were identified within 8 hours. Thirty (48%) had complications, with a subcapsular hematoma being most common (n=14). Less common adverse events included fever (n=5), skin site ooze (n=3), intraperitoneal bleeding (n=3), hemobilia (n=2), anaphylaxis to gelfoam (n=2), and sepsis (n=1). In this study, the majority of biopsies were performed by interventional radiology (n=492); though, the complication rate was similar in both groups.
The authors conclude that their data support the outpatient liver biopsies in children.
My take: I disagree with the authors’ conclusion to some extent. Their population is too small to detect rare but severe complications. Our empiric practice is watch children older than 6 years of age for 6 hours and watch younger children (or others deemed at increased risk) for 24 hours.
Related blog posts:
Slim Pickings: Data for 2nd-Line Autoimmune Hepatitis Pediatric Therapy
A recent study (AN Zizzo et al. JPGN 2017; 65: 6-15) performed a systematic review and meta-analysis of pediatric autoimmune hepatitis (AIH) studies.
The most remarkable finding was that there were only 76 patients from 15 qualifying studies.
Other findings:
- Response to mycophenolate mofetil (MMF) with 34 patients was 36% (according to abstract) at 6 months (discrepancy in article –results state 38% response)
- Response to cyclosporine with 15 patients was 83% (discrepancy in article –results state 86% response)
- Response to tacrolimus with 4 patients was 50%
- Adverse effects were very common, particularly with cyclosporine (64% noted at least 1 adverse effect)
The article has an associated editorial (N Kerkar, pg 2-3). “The adverse event profile of cyclosporine with gingival hyperplasia, hypertrichosis, nephrotoxicity, and neurotoxicity made it challenging for long-term use in children.” Besides the small number of patients, “the studies that were included were largely “observational”‘ which limits their findings as well. The study authors recommend MMF as the preferred option for 2nd-line therapy.
My take: Fortunately, most patients with autoimmune hepatitis respond to first line therapy with azathioprine/steroids. It is unclear what is the optimal 2nd-line treatment for refractory patients.
Related blog entries:
Will Emerging Therapies for Fatty Liver Disease Be Affordable?
With non-alcoholic steatohepatitis (NASH), there are currently no established medical therapies. However, several candidate medications look promising. However in recent years, many new medications have come with an impressive price tag and this has led to questions about whether emerging therapies for NASH will be affordable.
A recent article looked at the medication Obeticholic Acid, which was approved for treating primary biliary cholangitis. It is possible that it will be helpful for NASH. Yet, its cost , currently, is about $70,000 per year
GIHepNews: Despite clinical promise, obeticholic acid may be too expensive for treating NASH
Here’s an excerpt:
In the 72-week Phase II trial, called FLINT, 273 men and women with NASH were randomly assigned to receive OCA or placebo (Lancet 2015;385:956-965). Liver histology improved in 45% of those receiving OCA versus 21% in those receiving sham therapy (P=0.002). An increased risk for pruritus was the most notable adverse event among patients taking OCA (23% vs. 6% for placebo), according to the researchers. Based on the favorable benefit–risk results of the Phase II study, a Phase III trial is ongoing…
The expected benefit of OCA over lifestyle modifications for all the major long-term outcomes, such as decompensated cirrhosis (10% vs. 9.4%), liver-related mortality (9% vs. 8.1%) and transplant-free survival (72.2% vs. 71.5%), were relatively modest, the researchers reported. Those differences resulted in a cost per quality-adjusted life-year saved of $5.2 million with the assumption that 16% of patients would relapse…
“If the efficacy compared to placebo is of the same order found in the FLINT trial, the current cost of the drug would be prohibitive in a population-based context,” said Dr. Lavine, who was a co-investigator on the trial.
My take: Given the growing burden of NASH, new effective treatments are needed. In my view, though, cost-effectiveness has to be a consideration.