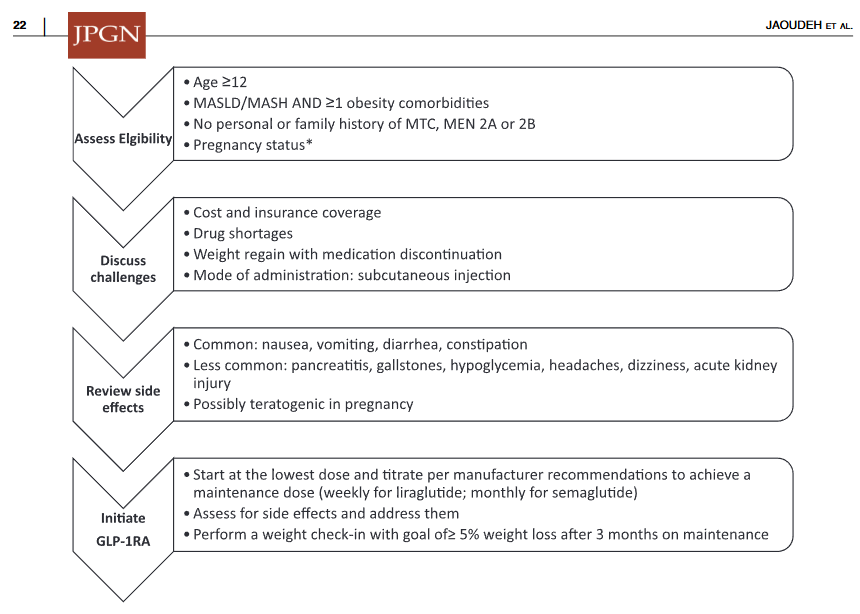
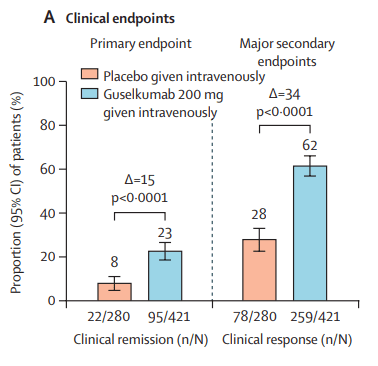
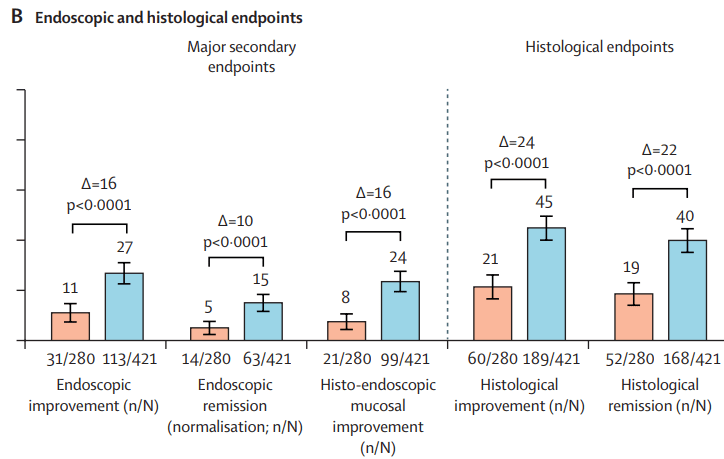
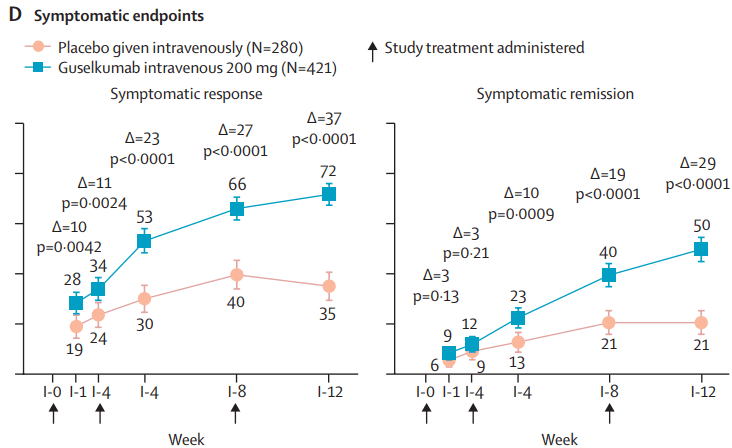
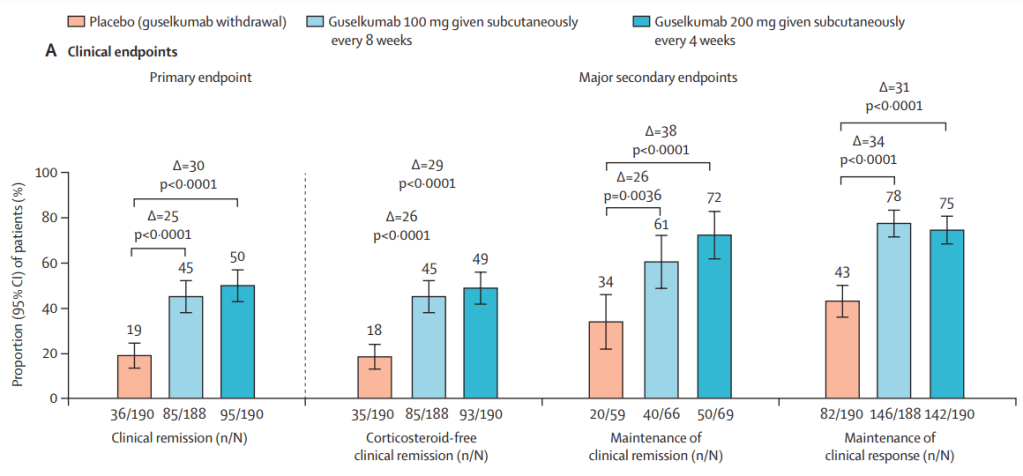
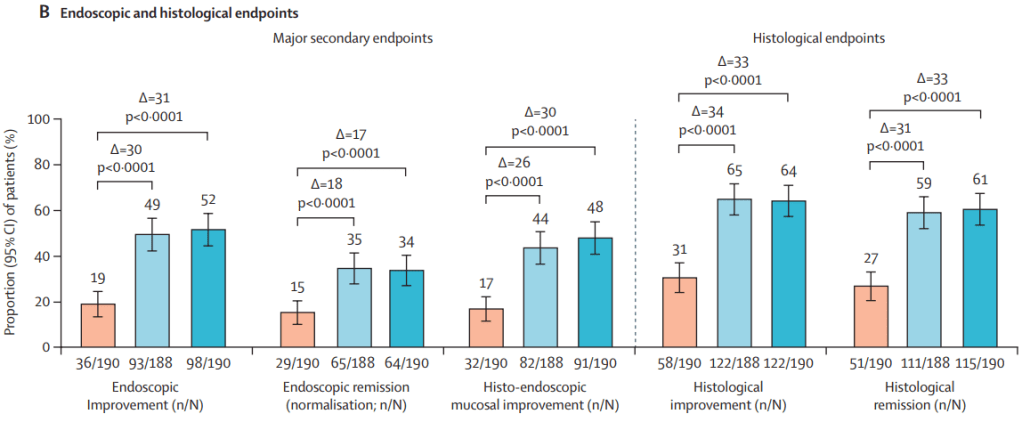
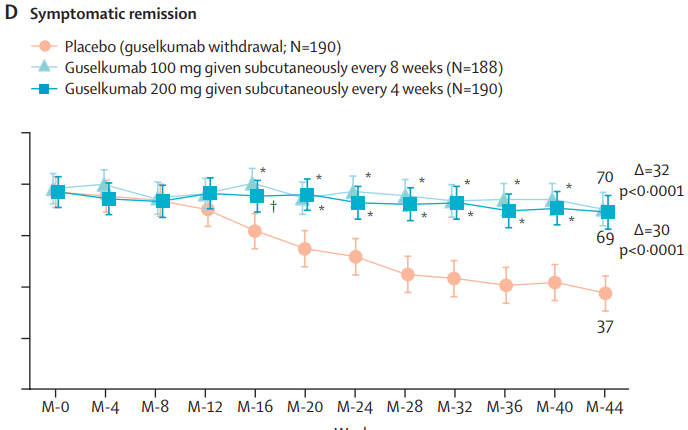
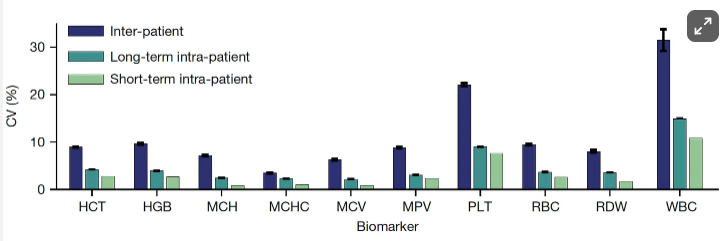
Two recent commentaries on “hot button” issues:
- ER Menzin: The Pediatrician’s Lament. NEJM 2025; 392: 320-321
- PMG Santos et al. Texas Executive Order GA-46 and the Erosion of Trust in Health Care. NEJM 2025; 392: 108-109
An excerpt from the first commentary:
“Throughout my career, I have seen new vaccines approved: pneumococcus, rotavirus, meningococcus, and human papillomavirus. In each case, I have studied the data, reviewed published recommendations, and adjusted my language to encourage vaccination. I consider the high immunization rate in my patient panel to be one of my greatest professional accomplishments — a quantitative metric of the benefit I provide…
Every so often, parents will look at me over a smiling infant and tell me they want their child to have only one or two recommended vaccines. Can I choose the most important? I tell them the question is akin to asking me to pick my favorite child — an impossible task…
Some will ask, “Can you recommend a good pediatrician who does not believe in vaccines?” No, I say, no more than I can recommend a good physicist who does not believe in gravity…
Even if patients are skeptical of the alphabet soup of institutions designed to protect and safeguard their health, they still have confidence in the long-standing relationships with their clinicians. To deserve that trust, we are obligated to raise our collective voice in defense of science, health, and vaccines.”
From the 2nd Commentary:
“On Halloween morning, 2024, Texas physicians received disturbing news about hospital policies set in compliance with Governor Greg Abbott’s Executive Order GA-46 — a rule that mandates the collection and reporting of information on patient citizenship status during intake…
Throughout medical training, physicians learn that trust is a cornerstone of patient care: we ask patients to share deeply personal information about themselves and their loved ones, with the assurance that legal and ethical safeguards protect against the misuse of such information. Chief among these safeguards is the Health Insurance Portability and Accountability Act (HIPAA), which restricts physicians from disclosing protected health information (PHI) without a person’s consent. Citizenship status, though not traditionally considered PHI, may be treated as such when it is paired with medical information obtained during patient encounters…
GA-46 … will deter immigrants, both documented and undocumented, from seeking help for serious medical concerns… Under the Emergency Medical Treatment and Labor Act (EMTALA), hospitals are required to provide emergency care to all patients, regardless of citizenship status. GA-46 indirectly conflicts with EMTALA’s intent to guarantee access to emergency services for all people by discouraging undocumented people from seeking care…
From an economic standpoint, Texas officials have stated that the goal of GA-46 is to protect the financial solvency of public hospitals; however, federal financial support covers most uncompensated care costs. Moreover, contributions from immigrants help sustain the viability of public health insurance programs. In 2017, immigrants helped offset a $67 billion deficit in health care costs for U.S.-born citizens by paying $58 billion more in taxes and premiums than was spent on their health care; 89% of this surplus was attributed to contributions made by undocumented immigrants…
Physicians in Texas and Florida must continue to inform patients of their right to refuse disclosure of their citizenship status.”
Related blog posts:
- “The Staggering Success of Vaccines”
- Vaccine Safety -Put into Perspective
- Flu Shots & Other Vaccines Linked to Lower Rates of Dementia
- Good News Story: The Remarkable Hepatitis B Vaccine Story
- Why Rich Kids Get Measles More Often in the U.S. …
- Parental Immunity (to Education) and Vaccine Decision …
- Measles, Seizures and Sometimes Death due to Vaccine …
- Why Doctors Don’t Want Unvaccinated Children in Their …
- Rotavirus Vaccine: 4-year Savings One Billion Dollars and …
- The Paradox of Vaccine Resistance | gutsandgrowth
- “Too many vaccines and autism” is debunked | gutsandgrowth