DLR Sun et al. J Pediatr 2024; 266: 113878. Hemoglobin Threshold for Blood Transfusion in Young Children Hospitalized with Iron Deficiency Anemia
Background: This retrospective single-center study examined the transfusion threshold in children 6 months to 36 months (mean 18.5 months, n=125) in light of current society recommendations which advise against blood transfusion in hemodynamically stable children with iron deficiency anemia.
Key findings:
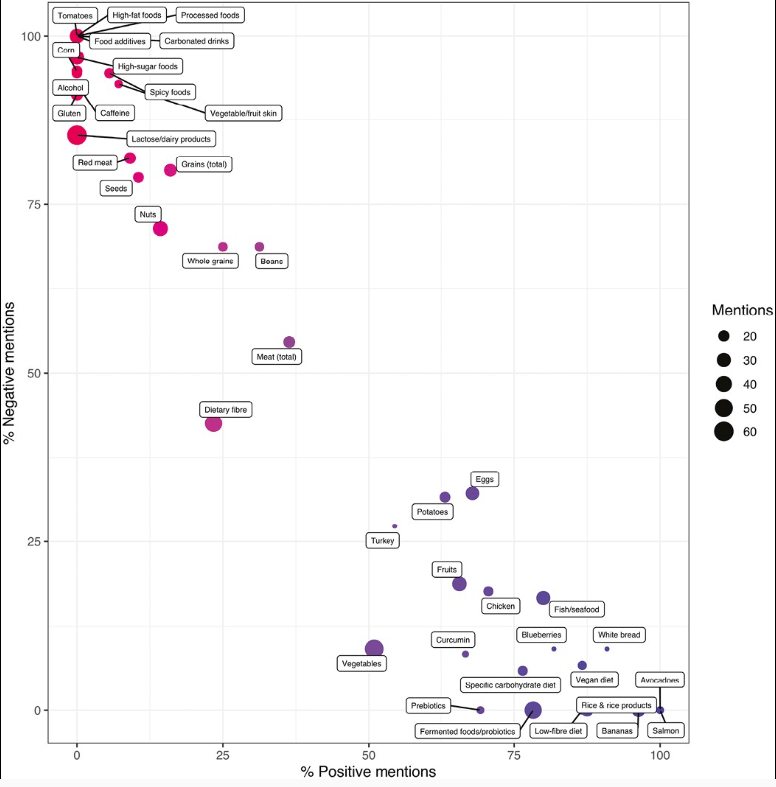
- “A hemoglobin of 39 g/L had sensitivity 92% and specificity 72% for transfusion.”
- In this study, there were 38 children with a hemoglobin <50 g/L who were NOT transfused
Discussion points:
- “There is a paucity of evidence to support a hemoglobin threshold for transfusion in the management of iron deficiency anemia (IDA)….McEvoy et al recently developed an algorithm for the management of young children with IDA in the ED with a consensus of surveyed hematologists recommending a hemoglobin of <50 g/L be used for transfusion.”
- In one study, “the median time to increased hemoglobin by at least 20 g/L…was 7 days for children receiving iron sucrose and 44 days for children receiving oral iron alone.”
My take: In children without active bleeding who are hemodynamically stable, more restrictive use of transfusion is now standard practice. In clinical practice, the exact threshold for transfusion is not clear. This study suggests that it is somewhere between 3.9 g/dL and 5 g/dL.
Related blog posts (regarding anemia and active bleeding):
- All Bleeding Stops (part 2) “A randomized trial showed lower rates of death (the primary outcome), rebleeding, and adverse events with a transfusion threshold of 7 g per deciliter than with a transfusion threshold of 9 g per deciliter.”
- Update on Upper GI Bleeding Recommendations
- All bleeding stops | gutsandgrowth
- Transfusion strategy in acute GI bleeding | gutsandgrowth

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.