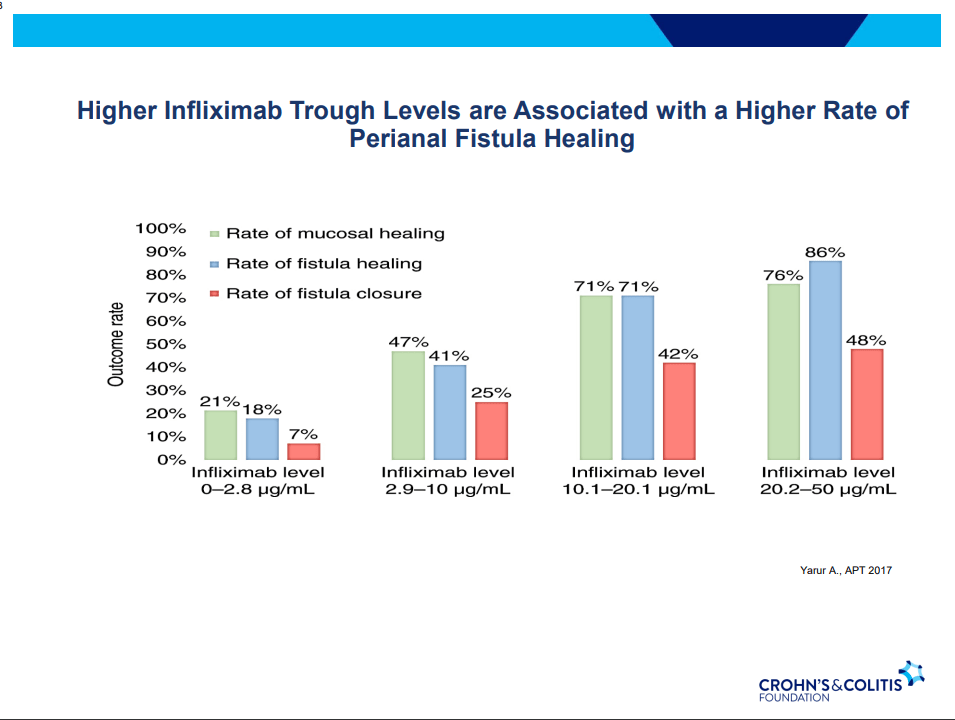
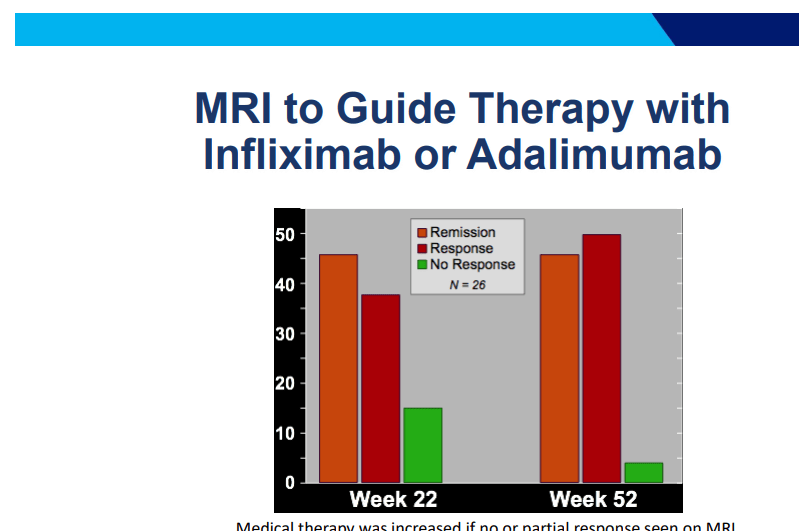
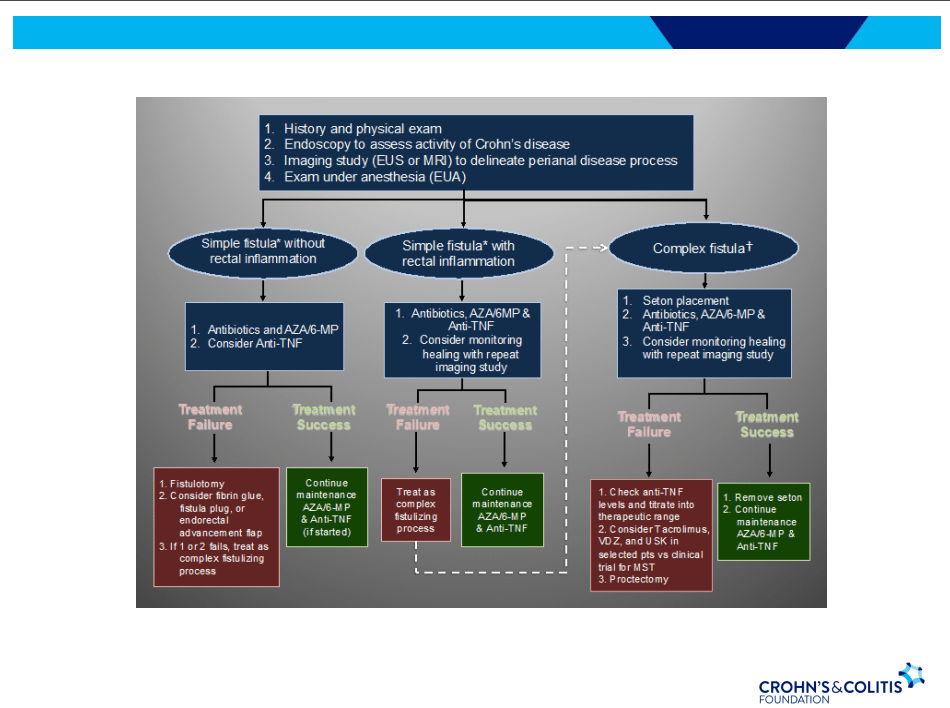
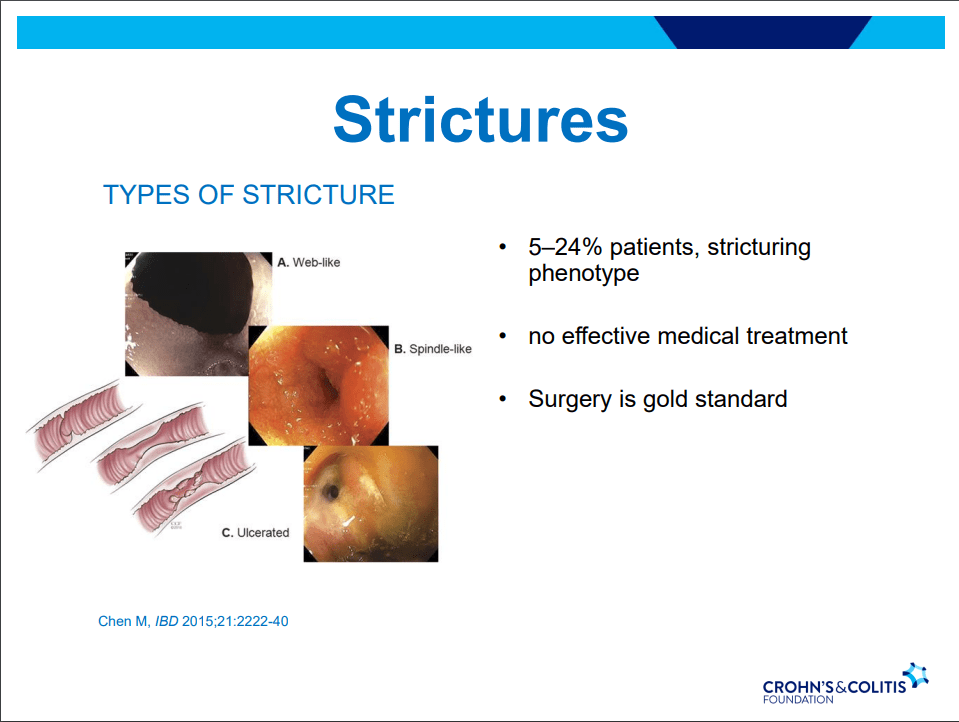
C Gamble et al. NEJM 2023; 389:795-807. Timing of Primary Surgery for Cleft Palate
Conclusion from study authors: Medically fit infants who underwent primary surgery for isolated cleft palate in adequately resourced settings at 6 months of age were less likely to have velopharyngeal insufficiency at the age of 5 years than those who had surgery at 12 months of age.
The associated editorial (R Tse, O Jackson, N Engl J Med 2023; 389:857-858. Mind the Gap) notes that “over two thirds of infants who were screened were excluded because their cleft was part of a syndrome, they were deemed to be medically unfit for early surgery, or they had a cleft that was too wide.” Also, “the incidence of additional surgery to treat velopharyngeal insufficiency was greater in the 6-month group than in the 12-month group (30 procedures in 27 children vs. 17 procedures in 16 children), and speech assessments performed after the secondary procedures were used in the analysis. Thus, the trial evaluated the mixed effects of primary surgery and secondary surgery in some patients.” Finally, “the considerations with regard to early surgery include the greater technical complexity of the procedure and the greater risks associated with anesthesia (airway complications and potential neurodevelopmental sequelae). Early surgery may also contribute to midfacial growth restriction, which becomes apparent only later, in adolescence, and may require complex corrective jaw surgery.”
My take: Early cleft palate surgery is not best for all infants with this defect.
Related blog post: Skinny Babies with Cleft Lips and/or Cleft Palates