Yesterday’s post highlighted immune-mediated disorders likely caused by anti-TNF therapy; this includes rheumatoid arthritis, psoriasis, hidradenitis suppurativa, and chronic recurrent multifocal osteomyelitis. Anti-TNF inhibitors can be the reason for drug-induced liver disease (DILI) including autoimmune hepatitis (AIH) as well.
- In one study, 8% of children receiving anti-TNF therapy developed a new elevation in ALT.
- Most often liver enzyme elevation is mild and transient
- Differential diagnosis for persistent elevation can be due to DILI, autoimmune liver disease (eg. PSC, AIH), or rarely due to a combination (autoimmune drug-induced liver disease). The latter can improve with drug cessation and with corticosteroid treatment.
Some slides on this topic (courtesy of William. Balistreri):


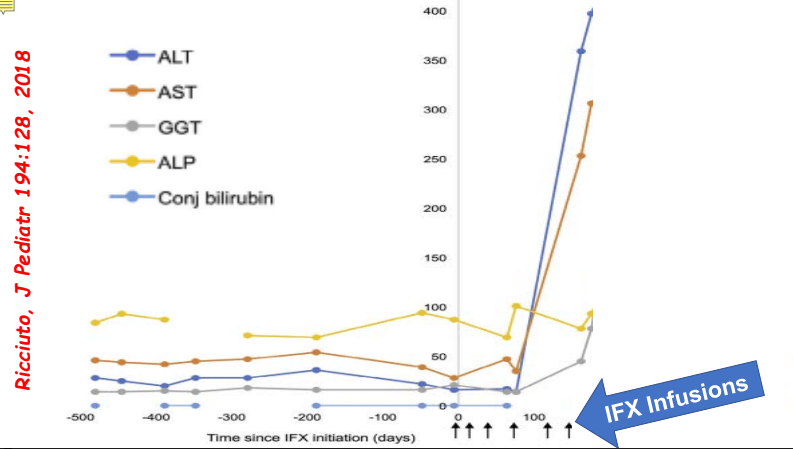
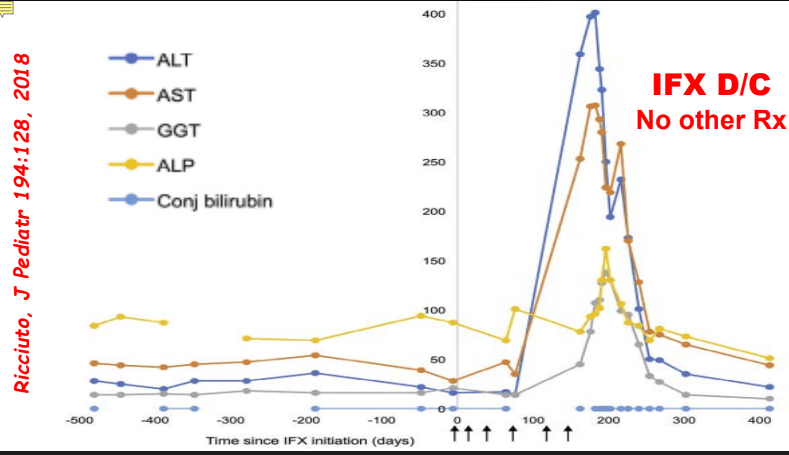
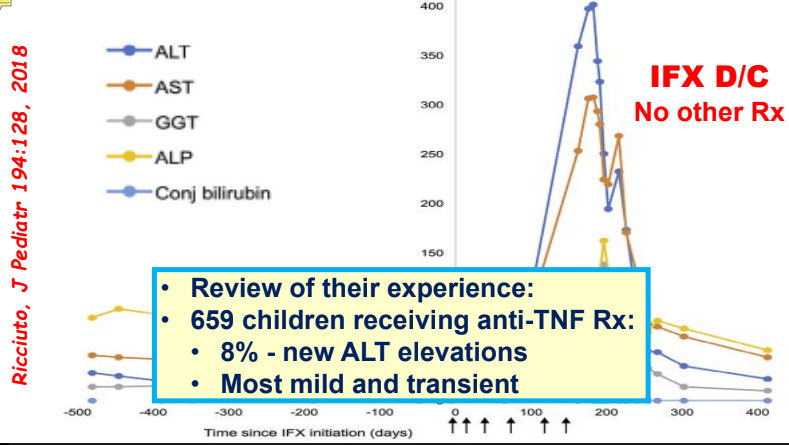


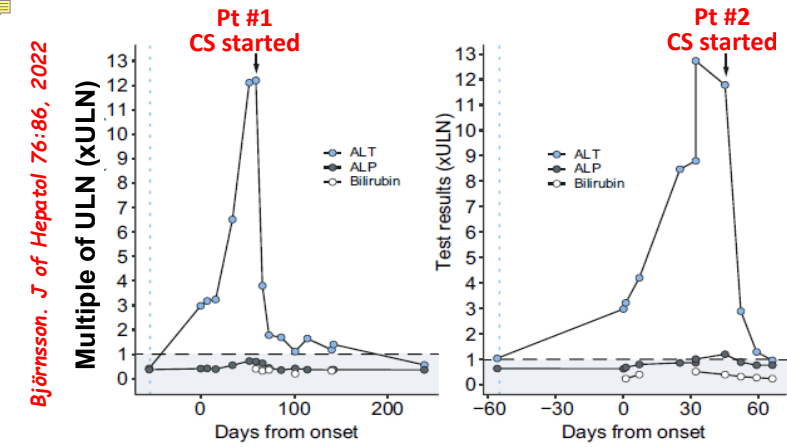
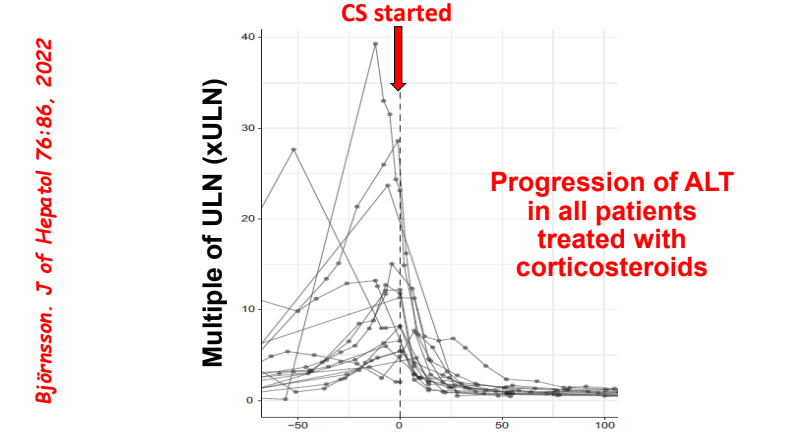
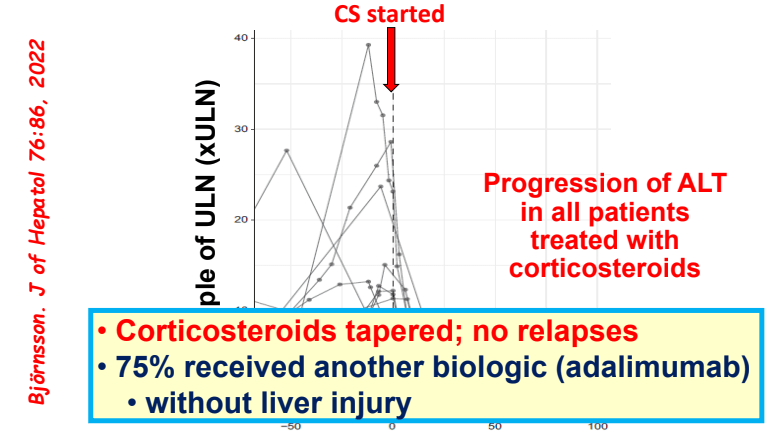

My take: Serious liver injury related to anti-TNF therapy is rare. When liver enzymes are persistently elevated, consider DILI including anti-TNF agents.
Related blog posts:
- Autoimmune Hepatitis Associated with Anti-TNF Therapy. Serious liver injury related to anti-TNF therapy is rare. A great place to understand the spectrum of liver problems potentially related to infliximab is the livertox website
- Liver Injury from Anti-TNF Agents
- Understanding the Reasons for Abnormal Liver Enzymes in Pediatric Inflammatory Bowel Disease | gutsandgrowth
- Liver Problems with Inflammatory Bowel Disease
- Advice on drug-induced liver injury (DILI) | gutsandgrowth
- Liver toxicity -where to look online | gutsandgrowth









