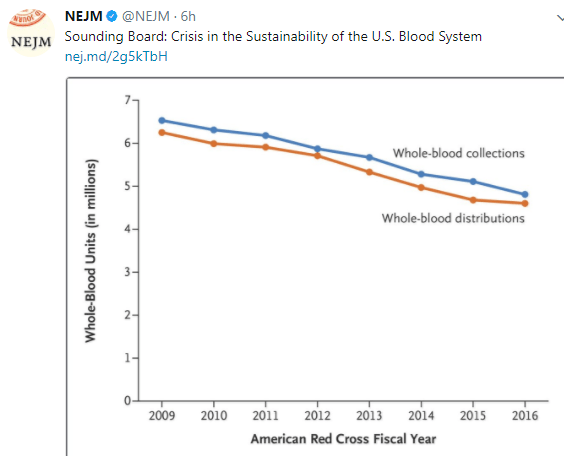
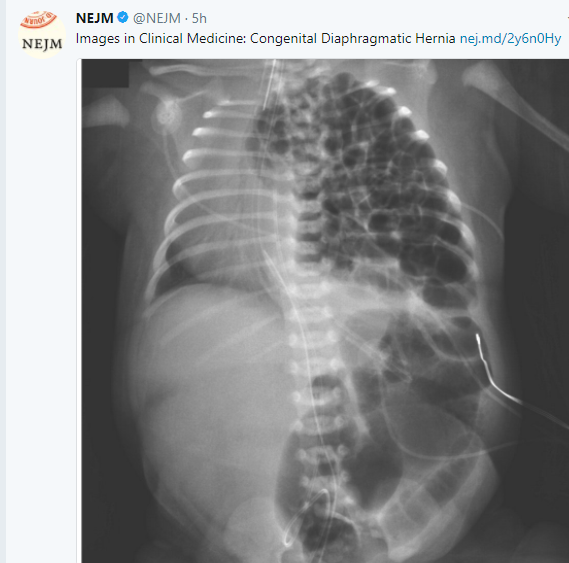
A recent study (TD Adams et al. NEJM 2017; 377: 1143-55) examines outcomes of bariatric surgery after 12 years. The ‘skinny’ on this study is that the weight loss/improved metabolic measures associated with bariatric surgery were very durable but there was a small increased risk of suicide among those undergoing bariatric surgery.
In this study, there were three cohorts:
- Surgery group: 418 patients
- Nonsurgery group 1: 417 patients. This group had sought surgery but did not receive surgery (often due to insurance coverage) (147 underwent subsequent surgery)
- Nonsurgery group 2: 321 patients. This group had not sought surgery (39 underwent subsequent surgery)
Key findings:
- At 12 yrs, mean change from baseline body weight was -35 kg in surgery group, compared with -2.9 kg in nonsurgery group 1 and 0 kg in nonsurgery group 2
- Of those with type 2 diabetes in the surgery group, type 2 diabetes remitted in 75% at 2 yrs and remained remitted in 51% at 12 yrs.
- The surgery group had higher remission rates of hypertension and dyslipidemia as well.
- 7 deaths by suicide were noted -5 in the surgery group, and 2 in the nonsurgery 1 group but only after the patients had undergone subsequent bariatric surgery
My take: Weight loss and improved metabolic changes at 6 yrs were maintained over the following 6 yrs. It is troubling that the surgery and/or weight loss is associated with suicide in a small number of patients.
Related blog entries:
- Six year outcomes with Bariatric Surgery
- Reaching Consensus on Bariatric Intervention in Children and Adolescents | gutsandgrowth
- Bariatric Surgery and Reversal of NASH | gutsandgrowth
- Childhood Obesity and Consensus Recommendations | gutsandgrowth
- Trends in Adolescent Bariatric Surgery | gutsandgrowth
- Eliminating sweetened beverages to help obesity | gutsandgrowth
- Weight of the Nation | gutsandgrowth
- Cardiovascular disease for the entire family | gutsandgrowth
- Psychology of obesity and food addiction | gutsandgrowth
- Staggering cost of obesity | gutsandgrowth