This blog entry has abbreviated/summarized these presentations. Though not intentional, some important material is likely to have been omitted; in addition, transcription errors are possible as well.
Here is a link to postgraduate course syllabus: NASPGHAN PG Syllabus – 2017
Therapeutic Drug Monitoring
Andrew Grossman Children’s Hospital of Philadelphia
The topic of therapeutic drug monitoring, both reactive and proactive, has been discussed numerous times on this blog. This talk provided a good review of this topic.
Key points:
- Greatest predictor of infliximab treatment failure was a low infliximab (<0.9 mcg/mL at anytime or <2.2 mcg/mL at 14 weeks) (Castelle et al Am J Gastro 2013; 108: 962-71)
- Low level antibodies to infliximab may be transient in ~28% and may be overcome with escalation of therapy
- Tissue levels of infliximab (and other agents) may be inadequate despite good serum levels
What if anti-TNF fails
Maria Oliva-Hemker Johns Hopkins University School of Medicine
Key points:
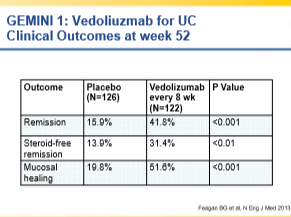
- Discussed prevalence of problem with anti-TNF failures and main options: vedolizumab, ustekinumab, and surgery
- Vedolizumab can take a while to work, particularly for Crohn’s disease
- Limited data in pediatrics for these newer agents
- Ustekinumab has some preliminary data indicating benefit with anti-TNF induced psoriaform rashes
- Newer agents also likely to need therapeutic drug monitoring
- Overall, ustekinumab and vedolizumab have good safety profiles at this point
Prevention of postoperative Crohn’s disease
Miguel Regueiro University of Pittsburgh
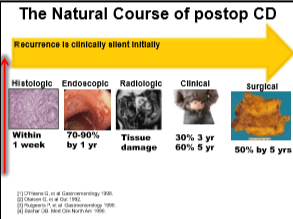
- Rationale for postoperative preventative treatment: high rate of recurrent disease which can be silent for several years despite progressive damage to GI tract
- Large study (PREVENT) to compare infliximab and placebo after surgery. Primary endpoint was clinical recurrence (was endpoint demanded by FDA) even though clinical recurrence can be a late finding. Endoscopic recurrence rate was a secondary endpoint.
Dr. Regueiro’s approach
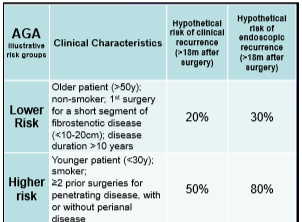
- Low risk patient –repeat scope at 6 months post-op, then every 1-3 yrs if no disease and Rx with anti-TNF or immunomodulator in those with endoscopic recurrence
- Moderate risk patient -possible use of thiopurine or use the ‘low risk’ approach
- High risk patient-combination therapy and if doing well for several years, consider monotherapy
- In pediatrics, the postoperative management is unclear due to difficulty with risk stratification. If postoperative treatment is not given, consider colonoscopy 3-4 months afterwards and treat if recurrence. Then could use calprotectin every 3 months to monitor and when >50, likely will need to be treated
PREVENT Trial Data: