B Constant et al. AJG 2024; DOI: 10.14309/ajg.0000000000002851. Insurer-Mandated Medication Utilization Barriers are Associated With Decreased Insurance Satisfaction and Adverse Clinical Outcomes: An Inflammatory Bowel Disease Partners Survey
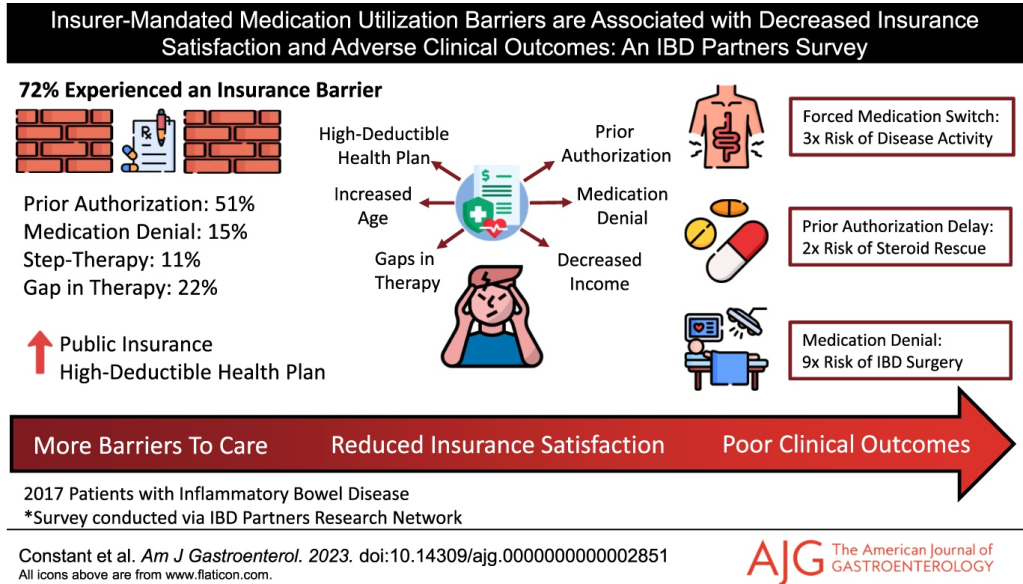
Key findings: In this longitudinal survey with 2017 patients, 72% experienced an insurer-mandated barrier, most commonly prior authorizations (51%). Fifteen percent were denied an IBD medication by their insurer, 22% experienced an insurance-related gap in therapy, and 8% were forced by their insurer to switch from an effective medication. Several insurance barriers were linked to negative downstream clinical outcomes, including prior authorizations associated with corticosteroid rescue (odds ratio [OR] 2.24]), forced medication switches associated with continued disease activity (OR 3.28), and medication denials associated with IBD-related surgery (OR 8.92).
Related blog posts:
- “Denials, Dilly-dallying and Despair”
- NY Times: ‘What’s My Life Worth?’ The Big Business of Denying Medical Care
- What’s Changing in IBD Care: Hospitalization Rates and Authorizations
- The Consequences of Prior Authorizations
- FDA IBD Workshop -Take-Home Points Off-label does not equate to experimental FDA Statement: The FD&C Act does not, however, limit the manner in which a physician may use an approved drug. Once a product has been approved for marketing, a physician may prescribe it for uses or in treatment regimens or patient populations that are not included in approved labeling. Such “unapproved” or, more precisely, “unlabeled” uses may be appropriate and rational in certain circumstances, and may, in fact, reflect approaches to drug therapy that have been extensively reported in medical literature
- “We Need More Information to Process This Claim” | gutsandgrowth
S Danese et al. Lancet Gastroenterol Hepatol 2024; 9: 133-146. Efficacy and safety of 48 weeks of guselkumab for patients with Crohn’s disease: maintenance results from the phase 2, randomised, double-blind GALAXI-1 trial
In this phase 2 randomised, multicentre, double-blind trial with 309 adults, the authors report on the safety and efficacy of subcutaneous guselkumab maintenance regimens to week 48 in the GALAXI-1 study. Key findings:
- “At week 48, the numbers of patients with CDAI clinical remission were 39 (64%) in the guselkumab 200→100 mg group, 46 (73%) in the guselkumab 600→200 mg group, 35 (57%) in the guselkumab 1200→200 mg group, and 37 (59%) in the ustekinumab group.”
- “Eendoscopic remission was seen in 11 (18%), 11 (17%), 20 (33%), and four (6%) patients, respectively.”
Related blog posts:
- Guselkumab: Expanding the GALAXI of Treatments for Crohn’s Disease
- New IBD Medication: Guselkumab for UC (QUASAR study)
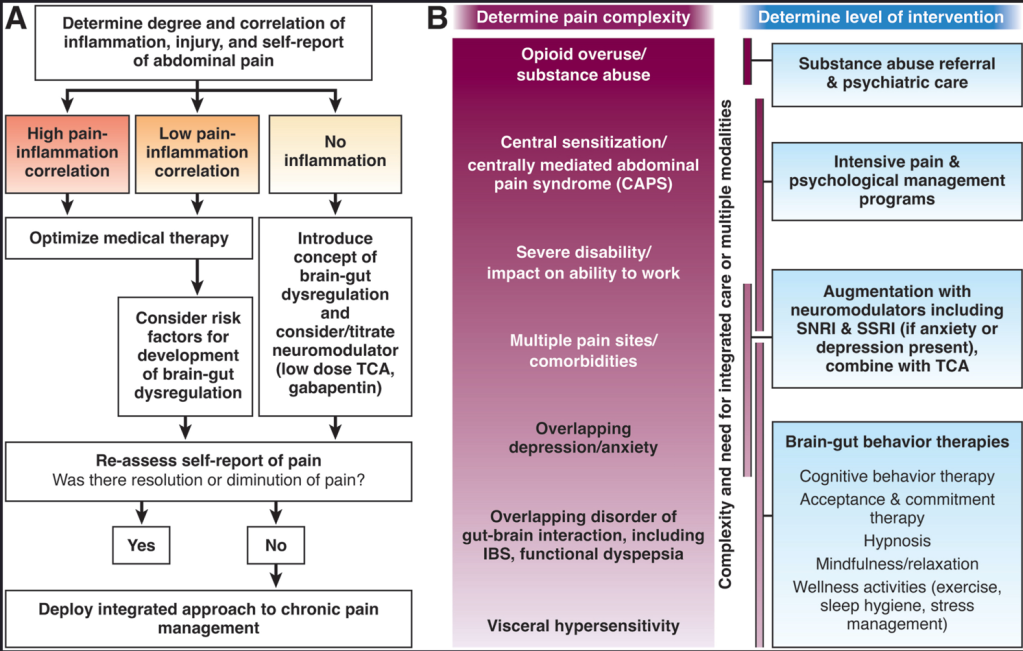
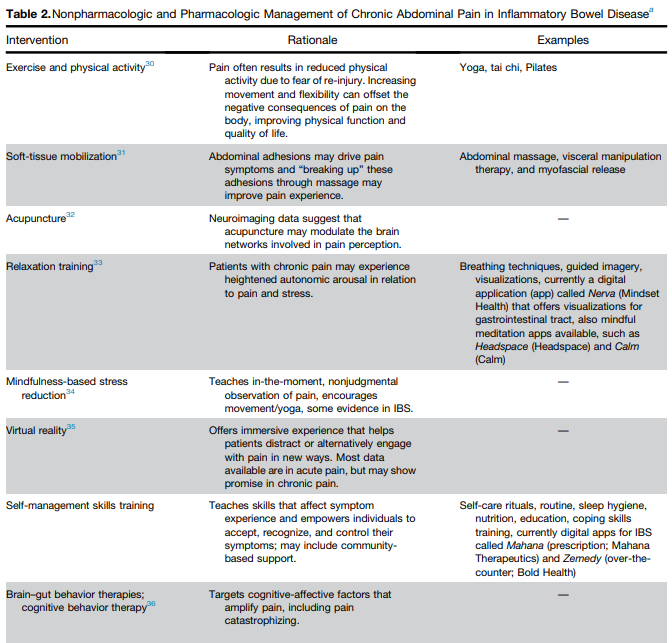
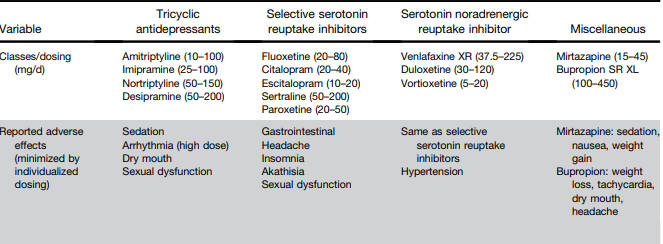
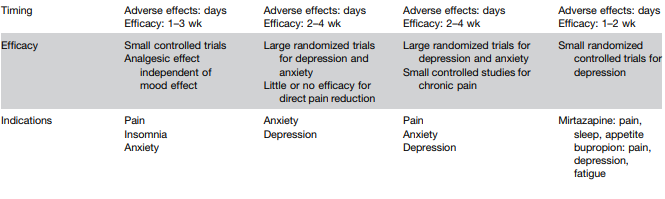
L Keefer et al. Gastroenterology 2024; 166: 1182-1189. AGA Clinical Practice Update on Pain Management in Inflammatory Bowel Disease: Commentary
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.