R Lujan, R Buchuk et al. Gastroenterol 2024; 166: 815-825.Early Initiation of Biologics and Disease Outcomes in Adults and Children With Inflammatory Bowel Diseases: Results From the Epidemiology Group of the Nationwide Israeli Inflammatory Bowel Disease Research Nucleus Cohort
Methods: All patients diagnosed with CD or UC in Israel (2005–2020) were included in the Epidemiology Group of the Israeli Inflammatory Bowel Disease Research Nucleus cohort. The authors compared disease duration at biologics initiation (ie, 0–3 months, >3–12 months, >1–2 years, and >2–3 years) using the cloning, censoring, and weighting by inverse probabilities method to emulate a target trial, adjusting for time-varying confounders and selection bias.
Of the 34,375 included patients (of whom 5240 [15%] were children), 7452 of 19,264 (39%) with CD and 2235 of 15,111 (15%) with UC received biologics. To attempt to adjust for patient characteristics, “essentially, each patient was cloned 4 times and 1 clone was assigend to each treatment strategy at baseline…[they were removed] subsequently if they did not receive the biologic within the acceptable time window.” Key findings:
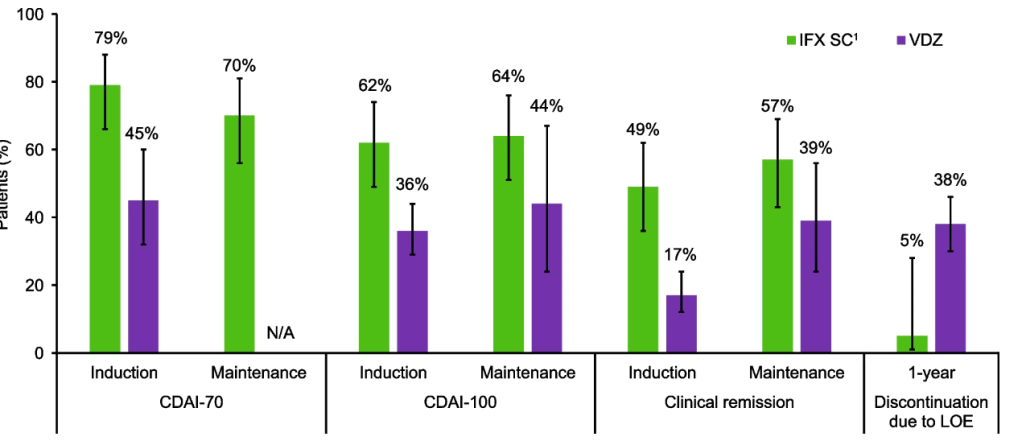
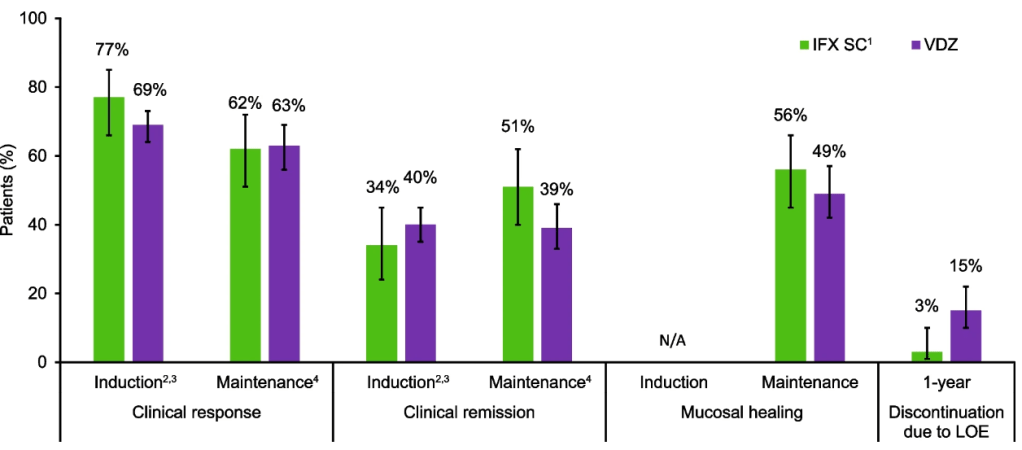
- In CD, by 10 years postdiagnosis, the probability of CD-related surgery decreased gradually but modestly with earlier initiation of biologics; a significant difference was noted between >2–3 years (31%) and 0–3 months (18%; P = .02; number needed to treat, 7.7)
- In CD, the 10-year probability of steroid dependency for the 0–3-month period (19%) differed both from the >2–3-year (31%; P < .001) and 1–2-year periods (37%; P < .001).
- In UC, no significant differences in colectomy or steroid dependency rates were observed between the treatment initiation periods.
- Similar trends were noted in the pediatric population.
Thus, overall, the study found that early initiation of biologics was associated only with a modest reduced risk of surgery and steroid dependency for Crohn’s disease. It was not found to reduce risk of colectomy or steroid dependency in UC.
My take: In my view, this study probably underestimates the benefits of early biologic therapy. Even though, lead time bias can skew interpretation, inadequately-treated disease likely leads to long-term damage. The associated editorial (pg 728) by Murphy notes that despite the sophisticated statistical methodology, all observational studies cannot fully control for unmeasured confounders.
Related blog posts:
- Is It RISKy Not To Use Anti-TNF Therapy for Pediatric Crohn’s Disease?
- Early Treatment Can Prevent Fistulas in Pediatric Crohn’s Disease
- What is Mild Crohn’s Disease and How to Treat It
- Paris Classification Quiz and Explanation
- CCFA: Updates in Inflammatory Bowel Disease 2017 (part 3)
- Paris Classification of Pediatric Crohn’s Disease
- Validated SEMA-CD Score For Crohn’s Disease

This past weekend there were two fun events in Atlanta -Porchfest (Virginia Highlands) and Midtown Garden Stroll. Porchfest featured more than 50 local bands.
Here’s a couple photos from the Midtown Garden Stroll: