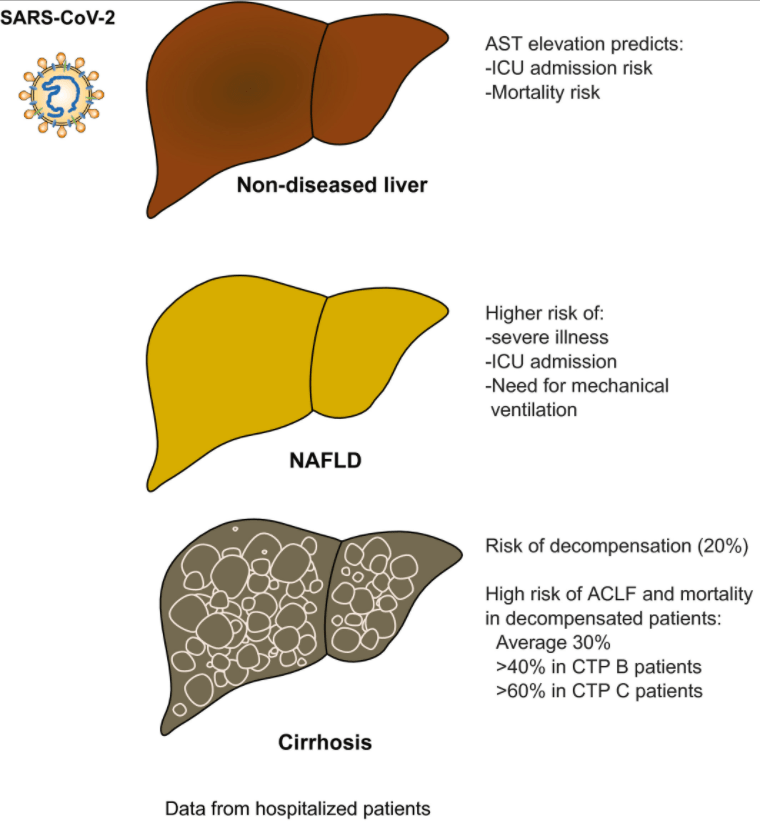
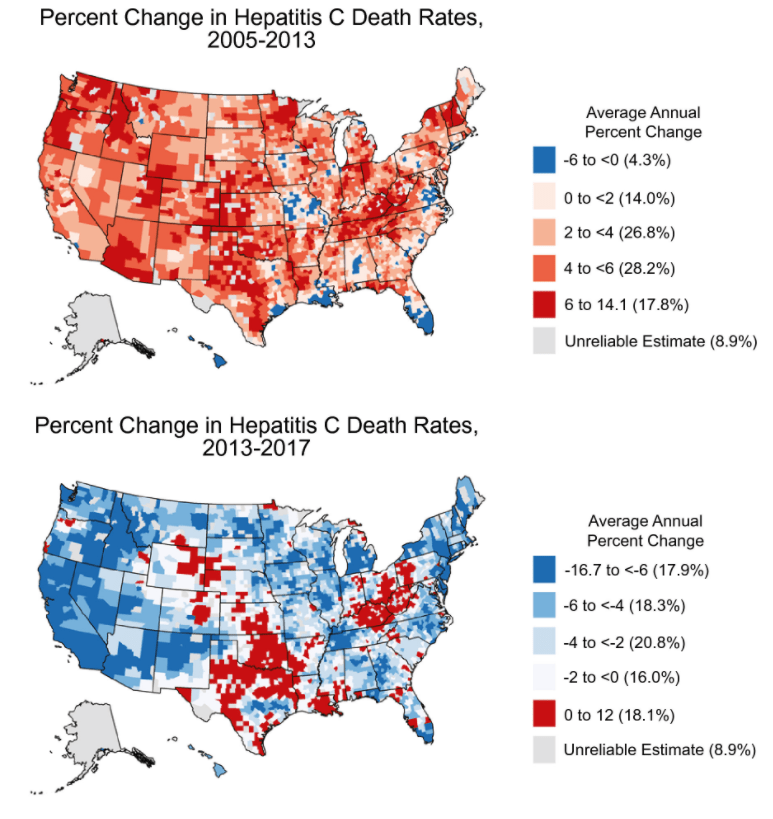
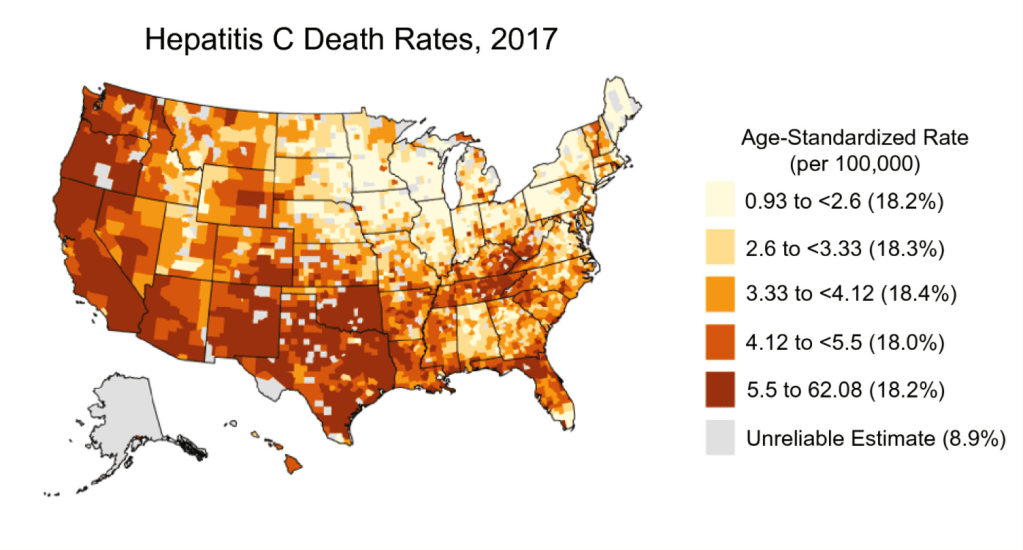
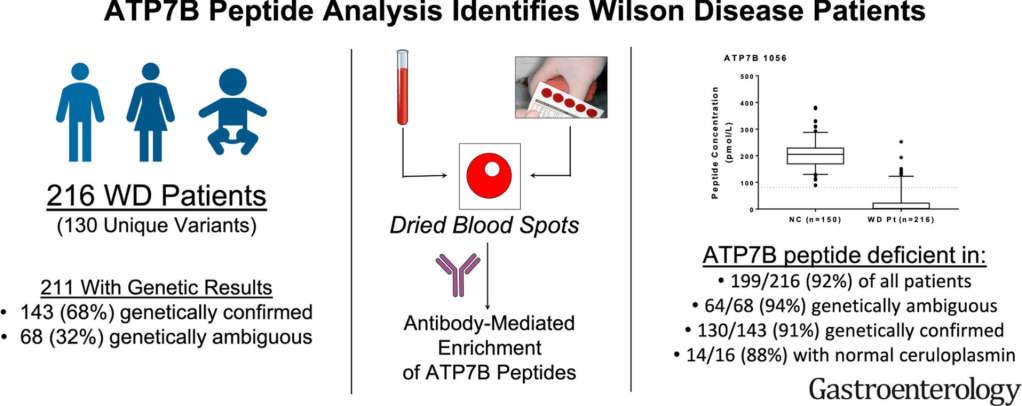
2021 Halloween Pics:

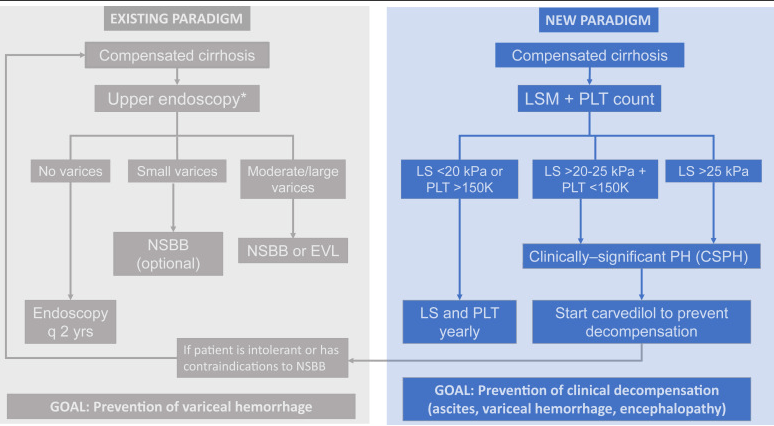
G Garcia-Tsao, JG Abraldes. Gastroenterol 2021; 161: 770-773. Open Access: Nonselective Beta-Blockers in Compensated Cirrhosis: Preventing Variceal Hemorrhage or Preventing Decompensation?
Key points:
- Carvedilol (not approved in children) is a NSBB that has additional α1 adrenergic blocking activity that enhances the portal pressure–reducing effect, compared to other NSBBs (eg. nadolol, propranolol).
- New paradigm focuses on “the most relevant end point in compensated cirrhosis…development of decompensation” (ascites, variceal hemorrhage, and/or hepatic encephalopathy).
- In the PREDESCI study with 201 patients with compensated cirrhosis and CSPH, with no or small varices, to NSBBs or placebo…”clinical decompensation, was significantly lower in the NSBB arm than in the placebo arm (from 27% to 17% over a median follow-up of 37 months: HR 0.51, 95% CI 0.26–0.97)”
- This model favors carvedilol over endoscopic variceal ligation; the “only RCT of carvedilol vs EVL in the prevention of first variceal hemorrhage showed a survival benefit of carvedilol over EVL.” Carvediol has been associated with improved survival in patients with cirrhosis (McDowell H.R. et al. Carvedilol is associated with improved survival in patients with cirrhosis: a long-term follow-up study. Aliment Pharmacol Ther. 2021; 5: 531-539)
- This model “consists of identifying those with CSPH (by means of noninvasive methods) and treating them with carvedilol, with the goal of preventing any decompensating event (not only variceal hemorrhage).”
- Screening endoscopy would still be recommended in newly-diagnosed decompensated cirrhosis and those intolerant to NSBBs.
My take: This new paradigm is one approach for portal hypertension in adults. More studies are needed in the pediatric age group to help determine whether medical therapy can obviate the need for EVL in most children with cirrhosis.
Related blog posts:
- #NASPGHAN19 Liver Symposium (excellent presentation by Dr. Romero on varices in children and portal hypertension)
- Transient Elastography in Pediatric Liver Disease
- Elastography-Accuracy in Children (great picture of Lake Moraine)
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.