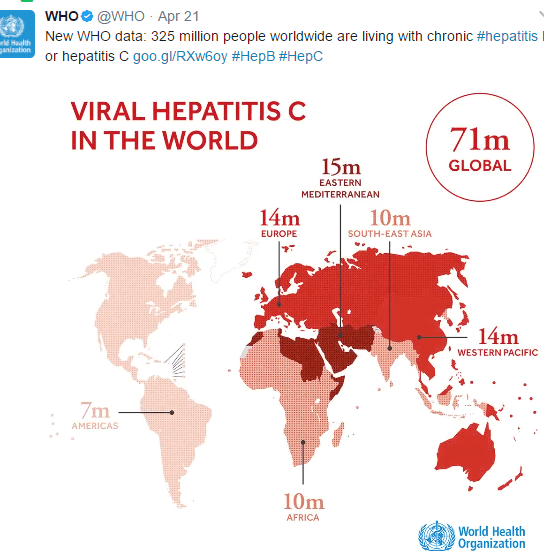
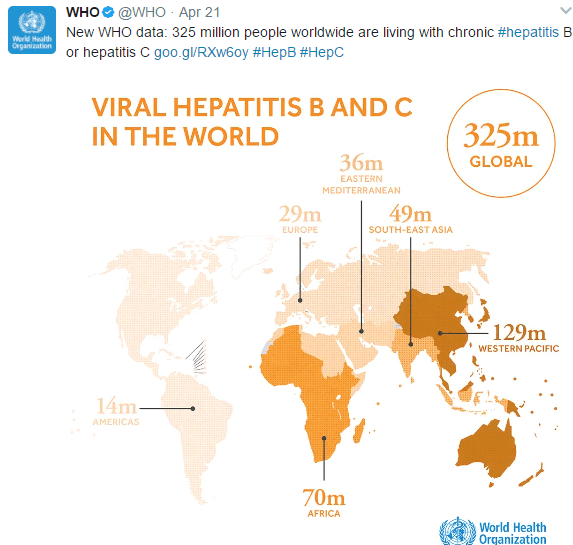
Related blog posts:
Category Archives: Pediatric Gastroenterology Intestinal Disorder
Pancreatitis Update (Part 2)
Our group received a very helpful update on pancreatitis from Maisam Abu-El-Haija (GI) and Jaime Nathan (surgery). These notes focus on the surgical perspective. My notes may include some errors in transcription and errors of omission. Some pictures of the slides are included below as well.
Key points:
- Several surgical procedures can be considered in chronic pancreatitis. Prior surgical procedures, though, could reduce islet cells if TPIAT needed later.
- TPIAT –Cincinnati experience: 17 cases in last 2 years. Highly selected group. Operation time takes about 10 hours (or more). GJ tube placed due to anticipated poor gastric emptying for 4-6 weeks. Fevers expected during 1st post-operative week due to systemic inflammatory response. ~15% of children remain on opioids after TPIAT, likely due to long-standing problems prior to TPIAT.
- Post-TPIAT care: PERT, vitamins, insulin (may wean off). During 1st year, anticoagulation, hydroxyurea/aspirin (if high platelets), and penicillin prophylaxis. Prior to TPIAT, patients receive vaccines (due to anticipated splenectomy).
Related blog post:
Pancreatitis Update (part 1)
Our group received a very helpful update on pancreatitis from Maisam Abu-El-Haija (GI) and Jaime Nathan (surgery). My notes may include some errors in transcription and errors of omission. Some pictures of the slides are included below as well.
Key points:
- About 30% of acute pancreatitis patients have a 2nd bout of pancreatitis. Obesity is a risk factor for recurrence.
- There has been a recent increase in incidence of acute pancreatitis.
- Cincinnati has a gene panel to examine the four most common mutations which cause hereditary pancreatitis (PRSS1, SPINK1, CFTR, and CRTC) along with 6 other relevant genes. (28 day turnaround) In addition, there is a pancreatitis insufficiency panel.
- Discussed screening for pancreatic insufficiency. Directly measuring pancreatic enzymes are more sensitive for early insufficiency, but may be unnecessary if good growth and normal stool elastase.
- There are NO proven medical/dietary therapies to prevent recurrent or chronic pancreatitis and eliminate pain symptoms.
Related blog posts:
- Consensus Pancreatitis Recommendations
- Risk of pancreatitis with celiac disease | gutsandgrowth
- INSPPIRE for pancreatitis | gutsandgrowth
- For the pediatric pancreatologists | gutsandgrowth
- Chronic Pancreatitis in Pediatrics -Descriptive Study | gutsandgrowth
- RECURRENT PANCREATITIS AND GENETIC UNDERPINNINGS | GUTSANDGROWTH
- DOES PANCREAS DIVISUM CAUSE PANCREATITIS? | GUTSANDGROWTH
- HOW HELPFUL ARE ANTIOXIDANTS FOR CHRONIC PANCREATITIS PAIN …
- Why an ERCP Study Matters to Pediatric Care
FPIES Guidelines
Recently, international consensus guidelines (A Nowak-Wegrzyn et al. J Allergy Clin Immunol 2017; 139: 1111-26) for the diagnosis and management of food protein-induced enterocolitis (FPIES) have been published.
The report starts with a review of epidemiology and diagnosis. Table 1 outlines features:
- early vs. late: <9 months or >9 months
- severity: mild-to-moderate =repetitive emesis with or without diarrhea, mild lethargy, severe =repetitive projectile emesis, pallor, lethargy, dehydration, hypotension
- timing: acute vs chronic. Acute occurs with intermittent exposures with emesis 1-4 h following exposure. Chronic occurs with repetitive food exposures (eg. formula in young infants)
- IgE positivity: classical FPIES is IgE negative. Atypical FPIES is IgE positive
Some recommendations:
- #4. “Consider specific IgE testing of children with FPIES to their trigger food because comorbid IgE-mediated sensitization to triggers, such as CM [cow’s milk], can infer a greater chance of persistent disease.”
- #8. Conduct food challenges “in patients with suspected FPIES in medically supervised settings in which access to rapid fluid resuscitation is available and prolonged observation can be provided, if necessary.”
- #14. Do not routinely obtain endoscopic evaluation as part of the evaluation of FPIES.
- #17. Acute FPIES should be considered a medical emergency. “Approximately 15% of patients can have hypovolemic shock.”
- #19. Consider ondansetron treatment as an adjunct (if >6 months of age)
- #21. Do not recommend routine maternal dietary elimination of offending triggers while breast-feeding if the infant is asymptomatic.
- #23. FPIES can occur to multiple foods. “The majority of children (65% to 80%) have FPIES to a single food, most commonly CM.” In one study, 5% to 10% of children reacted to more than 3 foods.
- #26. Use hypoallergenic formula in infants who can no longer breast-feed and are given a diagnosis of FPIES caused by CM. Most will tolerate extensively hydrolyzed formulas; some may require an amino acid based formula
- #29. Reviews natural history. “The age of CM tolerance appears to be around 3 years” but there has been variability in reports. For FPIES due to grains, average age of tolerance is 35 months and other solid foods is 42 months. The average age for soy is 12 months (later in some studies), for rice 4.7 years and 4.0 years for oats. For CM-FPIES with positive SPT response, a much protracted course has been reported, with older age of tolerance (~13.8 years)
Table III lists a differential diagnosis for FPIES and distinguishing features. This list includes gastroenteritis, necrotizing enterocolitis, anaphylaxis, food aversions, inborn errors of metabolism, cyclic vomiting/neurologic disorders, gastroesophageal reflux, Hirschsprung’s enterocolitis, eosinophilic gastroenteritis, celiac disease, immune enteropathies/IBD, intestinal obstruction, and primary immune deficiencies. Not listed on this table, but worth a mention, would be medical child abuse (aka Munchausen syndrome by proxy).
With regard to inborn errors of metabolism, these include urea cycle defects, hereditary fructose intolerance, hyperammonemic syndromes, Beta-oxidation defects, proprionic/methylmalonic academia, mitochondrial defects and others. Typically, features could include developmental delay, neurologic manifestations, organomegaly, and in some reaction to fruits.
Table IV specifies diagnostic criteria with the major criteria for acute FPIES: vomiting 1- to 4-h period after ingestion of the suspect food and absence of classic IgE-mediated allergic skin or respiratory symptoms. Minor criteria include extreme lethargy, pallor, need for emergency room evaluation/IV fluids, and diarrhea in 24 h (usually 5-10 h).
Table VI details management of FPIES. With moderate bouts, IV fluids with 20 mL/kg normal saline is recommended. For severe episodes, “consider administering intravenous methylprednisolone, 1 mg/kg; maximum 60-80 mg/dose” in addition to fluid resuscitation.
Table IX provides empiric guidelines for selecting weaning foods in infants with FPIES. The recommendations need to be considered based on whether the infant has shown tolerance for a number of foods, which can indicate the acceptability of a more liberal approach. Age-specific guidance:
4-6 months:
- Begin with smooth, thin purees and progress to thicker purees
- Lower-risk foods: vegetables, broccoli, cauliflower, parsnip, turnip, pumpkin
- Moderate-risk: squash, carrot, white potato, green bean
- Higher-risk: sweet potato, green peas
6-8 months:
- Continue to expand vegetables and fruits; in breast-fed, high-iron foods and/or supplemental iron are needed (1 mg/kg/day)
- Lower-risk: fruits, blueberries, strawberries, plum, watermelon, peach, avocado
- Moderate-risk: apple, pear, orange
- Higher-risk: banana
8-12 months:
- Offer soft-cooked and bite-and-dissolve textures
- Lower-risk: high iron foods, lamb, fortified quinoa, millet
- Moderate-risk: beef, fortified grits, corn cereal, wheat, barley
- Higher-risk: fortified infant rice and oat cereals
12 months:
- Offer tolerated table foods: chopped meats, soft vegetables, grains
- Lower-risk: tree nuts
- Moderate-risk: peanut, other legumes (besides green pea)
- Higher-risk: milk, soy, poultry, egg, fish
Overall, with regard to food introduction: While children with FPIES have increased reactions to other foods, “current early feeding guidelines do not recommend delay in introducing complementary foods past 6 months of life because of FPIES. A practical ordering for introducing solids at about 6 months of age at home could start with fruits and vegetables.” For infants with history of severe reactions, “supervised (eg. in-office) introduction can be considered…and prevent unnecessary avoidance.” As with new foods in the home setting, starting with small amounts is recommended and then gradual build up in serving size.
Related blog post: SEED Journal Club: FPIES
Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
More IBD Cases Than Ever in Young Canadian Children
Summary of recent article (Link to full study: Benchimol EI, et al. Am J Gastroenterol. 2017;doi:10.1038/ajg.2017.97) by Healio Gastroenterology: IBD incidence rapidly increasing in young Canadian children
An excerpt:
To evaluate the recent incidence, prevalence and trends in childhood-onset IBD in Canada, Benchimol and colleagues used health administrative data from five provinces to identify children aged younger than 16 years who were diagnosed with IBD between 1999 and 2010. During this period, 3,462 children were diagnosed with Crohn’s disease, 1,382 with ulcerative colitis and 279 with unclassifiable IBD, for an overall IBD incidence of 9.68 (95% CI, 9.11-10.25) per 100,000 children.
Throughout the study period, the annual percentage change in overall IBD incidence remained statistically stable, increasing by just 2.06% per year, but the incidence increased significantly among children aged younger than 5 years, rising by 7.19% per year.
Further, the annual percentage change in the prevalence of IBD increased significantly throughout the study period (4.56%), and at the end of the study period IBD prevalence was 38.25 (95% CI, 35.78-40.73) per 100,000 children.
The investigators noted their findings confirmed the predominant form of pediatric-onset IBD was Crohn’s disease, and that more boys were affected than girls.
My take: While Canada has high prevalence of IBD, I expect that there will be similar trends in epidemiology in multiple regions in young children. When one looks at the increases in IBD prevalence over the last 100 years (see previous post) and the emergence of IBD in non-Western countries, it is quite alarming.
Also, last week a blog post discussed hepatic problems associated with IBD (Liver problems with IBD): here is full article text link: Hepatic Issues and Complications Associated with IBD
Related blog posts:
Liver Problems with Inflammatory Bowel Disease
A recent review (Full text: LJ Saubermann et al. JPGN 2017; 64: 639-52) discusses the hepatic issues and complications associated with inflammatory bowel disease.
Key topics:
- Primary Sclerosing Cholangitis (PSC)
- Autoimmune Hepatitis (AIH)
- Autoimmune Sclerosing Cholangitis (ASC)
- Portal Venous Thrombosis/hypercoagulability
- Cholelithiasis (more common in Crohn’s disease if diseased terminal ileum)
- Viral hepatitis
- Drug-Induced Liver Disease
- Fatty Liver disease
Many of these topics have been discussed previously on this blog. A couple of pointers in this review:
PSC:
- Greater risk of colorectal carcinoma
- IBD-PSC patients are at higher risk for pouchitis
- GGT of >252 U/L “was highly sensitive (99%) and had good specificity (71%) for PSC” [or ASC]
- The authors recommend “screening all newly diagnosed patients with IBD with ALT and GGT
- Immunosuppressive therapy is NOT effective
- Vancomycin therapy is currently being tested (clinical trials: NCT02137668 & NCT01802073)
AIH:
- Less frequent in IBD patients than PSC
- Most common treatment is prednisone/azathioprine
- 40-80% of children have cirrhosis at AIH diagnosis, but “progression to end-stage liver disease is rare and …with appropriate treatment, 80% of patients achieve remission.”
ASC:
- ASC is an overlap syndrome between AIH and PSC
- “It is important that children with IBD and apparent AIH are routinely investigated for evidence of biliary disease with MRCP”
- “ASC responds to the same immunosuppressive combination therapy used for AIH”
HAV/HBV Immunization:
- HAV vaccination is effective in patients with IBD…although the rate [seroconversion] was significantly lower” in patients receiving anti-TNF therapy (92.4% vs 99.1% in one study).
- In those needing HBV immunization: “One strategy evaluated to improve HBV immunity in adults with IBD is an accelerated course with double vaccine doses at 0, 1, and 2 months.”
Methotrexate (MTX):
- “The extent of histological features of hepatotoxicity secondary to long-term MTX use in IBD has been infrequently described; however, the inicdence of significant abnormal histological findings appears to be rather low.”
My take: This article is a good starting point for liver-related issues in IBD. For concerns regarding medications, the NIH livertox website is more useful and much more comprehensive.
Related blog entries:
DILI:
PSC:
- Population-Based Outcomes for Primary Sclerosing Cholangitis
- Should we care about subclinical PSC?
- Screening for subclinical PSC in IBD? | gutsandgrowth
- Vedolizumab for Primary Sclerosing Cholangitis (with … – gutsandgrowth
- Population-based outcomes for PSC gutsandgrowth
AIH:
- Withdrawing Immunosuppression with Autoimmune Hepatitis
- Diagnosing Autoimmune Hepatitis
- Adult versus Pediatric Data for Autoimmune Hepatitis
Safety of Long-term Adalimumab in Pediatrics; Weighted PCDAI
A recent study (W Faubion et al. Inflamm Bowel Dis 2017; 23: 453-60) reports on the long-term safety/effectiveness of Adalimumab in pediatric patients entering the IMAgINE 2 trial (& who completed the 52 week IMAgINE 1 trial).
Patients with a PCDAI <10 were considered to be in remission and those who had a drop in PCDAI of 15 or more were considered to have had a treatment response.
Key findings:
- Of the 100 patients enrolled in IMAgINE 2, 41% achieved remission and 48% had a treatment response at week 240.
- >80% of patients were “able to discontinue use of corticosteroids.”
- Adalimumab treatment was associated with growth normalization.
- No new safety signals were identified.
While this study provides some reassurance regarding long-term adalimumab use, it should be noted that the instruments used to assess efficacy in this trial (& many others) are suboptimal.
A recent study (D Turner et al. JPGN 2017; 64: 254-60) showed that PCDAI (and several similar versions) had “poor correlation with calprotectin” and none of the PCDAI versions “can give a valid assessment of mucosal healing.” This study had used prospectively collected data from the ImageKids study of 100 children with Crohn’s disease. For the weighted PCDAI, the “best cut-off to identify endoscopic mucosal healing was <12.5 points” with a sensitivity of 58% and specificity of 84%.\
wPCDAI:
History: (recall 1 week):
- Abdominal Pain 0=None, 10=Mild (does not interfere with activities, brief), 20=Moderate/Severe
- Patient functioning 0=No limitations, 10=Occn difficulty with activities (below par), 20=frequent limitations
- Stools per day 0=0-1 liquid stools, no blood, 7.5=up to 2 semiformed stools with blood or 2-5 liquid nonblood, 15=Gross bleeding or ≥6 liquid stools or nocturnal diarrhea
Laboratory
- ESR 0 points if <20, 7.5 points if 20-50, 15 points if >50
- Albumin 0 points if ≥3.5 g/dL, 10 points if 3.1-3.4 g/dL, and 20 points if ≤3.0 g/dL
Examination
- Weight 0= Weight gain or stable or voluntary weight loss, 5=involuntary weight loss 1-9% or involuntary weight stable, 10= weight loss ≥10%
- Perirectal Disease 0=None or asymptomatic tags, 7.5= 1-2 indolent fistula, scant drainage, no tenderness, 15=active fistula, drainage, tenderness or abscess
Extraintestinal Manifestatons: Fever for 3 days (≥38.5), definite arthritis, uveitis, erythema nodosum, or pyoderma gangrenosum
- Points: 0=None, 10 ≥1
Total Score 0-125: ______________________
As compared with PCDAI, the weighted PCDAI drops height velocity, abdominal examination, and hematocrit. Turner et al note “their exclusion does not mean that they have no role in reflecting disease activity, but that the other included items, as a whole, are inclusive of the contribution of the 3 items.” Also, the weighted PCDAI simplifies the “extraintestinal manifestation” into a simple choice; overall, this affects few scores due to the low frequency of these manifestations.
Related blog posts:
How Many Times Have You Done This?
Two recent studies illustrate the need for better endoscopic training for fellows:
- AM Banc-Husu et al. JPGN 2017; 64: e88-e91.
- EA Mezoff et al. JPGN 2017; 64: e96-e99.
In the first study from CHOP, the authors performed a retrospective review of their endosocpic database from 2009-2014. Out of 12,737 upper endoscopies, 15 patients underwent 17 upper endoscopies which required a therapeutic intervention to control nonvariceal bleeding (1:750 procedures). therefore, among their 24 fellows, this resulted in less than 1 therapeutic endoscopy per fellow.
In the second study, “a recent study suggests that fellows are largely unable to achieve the prescribed case volume recommended to achieve competence.” The authors found that control of nonvariceal bleeding [and other advanced endoscopy cases] “were performed exclusively but relatively infrequently by members of this advanced endoscopy service. Fellows…participated in relatively few.”
My take: Fortunately, life-threatening nonvariceal bleeding cases are infrequent. The downside of the rarity of these cases is the lack of subspecialty expertise, particularly in recently trained physicians. My recommendations:
- Work with experience physicians (adult and pediatric) until sufficient expertise is developed.
- Even experienced physicians should collaborate on these difficult cases
- Efforts to improve simulation would be welcome –similar to aviation pilots.
Related blog posts:
- Training Not Meeting Procedure Thresholds for Fellows
- All bleeding stops | gutsandgrowth
- All Bleeding Stops (part 2) | gutsandgrowth
- Cutting Edge for Endoscopic Control of Bleeding | gutsandgrowth
- NASPGHAN Postgraduate Course 2014 -Endoscopy… | gutsandgrowth
- Watch the Bleeding Ulcer Stop! | gutsandgrowth
- Transfusion strategy in acute GI bleeding | gutsandgrowth
- Is a Continuous IV Proton Pump Inhibitor Really Necessary for GI Bleeders?
A Better Budesonide for Eosinophilic Esophagitis (Part 2)
A recent study (ES Dellon et al Gastroenterol 2017; 152: 776-86) provides more data indicating that a premixed solution of budesonide improves eosinophilic esophagitis (EoE). This study complements a recent report highlighted in a blog post earlier this year:
A Better Budesonide for Eosinophilic Esophagitis
In the present study by Dellon et al, the authors performed a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial of 93 EoE patients (ages 11-40). All patients had dysphagia and active EoE. The active treatment group received 2 mg twice daily.
Key findings:
- Dysphagia symptom questionnnaire (DSQ) scores improved more in the active treatment group compared to placebo. At baseline, the DSQ scores were 29.3 and 29.0 respectively. After 12 weeks, the the scores were 15.0 and 21.5 respectively.
- Similarly, the active treatment group peak eosinophil counts improved more. At baseline, the treatment group had a count (per hpf) of 156 and this dropped to 39; in contrast, the placebo group started at 130 and dropped to 113.
- The overall histologic response (≤6 eos/hpf) was 39% for the treatment group and 3% for the placebo group.
- No significant adverse effects could be attributed to budesonide. There was 1 case of esohageal candidiasis. “There were no notable differences between the groups in cortisol levels.”
My take: Budesonide suspension is useful for EoE but not effective in all patients. A reliable composition from a manufacturer, if not too expensive, would be a big improvement for many kids with EoE. Higher doses of budesonide may be warranted in some cases of EoE.
Related blog posts:
- Adrenal Insufficiency due to Fluticasone in … – gutsandgrowth
- Higher Doses of Topical Steroids for Eosinophilic … – gutsandgrowth
- Do We Still Need PPI-REE? | gutsandgrowth
- Eosinophilic Esophagitis Slide Set | gutsandgrowth
- Eosinophilic Esophagitis Review -NEJM | gutsandgrowth
- How Common is Eosinophilic Esophagitis? | gutsandgrowth
- Nexium versus Fluticasone for EoE | gutsandgrowth
- Predicting Response to Topical Steroids in Eosinophilic Esophagitis …
- Putting in Place a Big Piece of the Eosinophilic Esophagitis Puzzle …
- Sweet Alternative to Splenda for Budesonide | gutsandgrowth
- Putting in Place a Big Piece of the Eosinophilic Esophagitis Puzzle …
Better to Do a Coin Toss than an ENT Examination to Determine Reflux
A recent study (R Rosen et al. J Pediatr 2017; 183: 127-31) adds additional data to the literature which has shown that ENT doctors are NOT able to tell if there is reflux by examining the airway.
Prior post on this topic: Accuracy of ENT diagnosis of Reflux Changes
This prospective, cross-sectional cohort study of 77 children correlated ENT examinations with “reflux finding score” (RFS) by three blinded otolaryngologists with objective measures of reflux: pH-metry and impedance. All children had chronic cough and underwent bronchoscopy and esophagogastroduodenoscopy.
Key findings:
- “There was no correlation between pH-MII variables and mean RFS”
- The concordance correlation for RFS between ENT doctors was low (intraclass correlation coefficient =0.32)
- Using pH-metry as a gold standard, the positive predictive value for the RFS was 29% whereas with MII as the gold standard, the positive predictive value for the RFS was 40%.
My take: ENT doctors are unable to tell if a patient has reflux. The finding of a red or swollen airway has poor predictive value in determining the presence of reflux –a coin toss is more reliable. Based on this study and others, starting a PPI because of an abnormal airway exam does not make sense.
Related blog posts:
- PPI Webinar NAPSPGHAN
- Treating reflux does not help asthma | gutsandgrowth
- Salivary Pepsin Doesn’t Pass Muster for Reflux
- Does Reflux Lead to Increased Aspiration Pneumonia? | gutsandgrowth
- How Likely is Reflux in Infants with “Reflux-like … – gutsandgrowth
- No Effect of Proton Pump Inhibitors and Irritability on … – gutsandgrowth
- pH Testing; Rumors of My Death are Premature
- The prosecution rests…PPIs on trial gutsandgrowth
- PPI Side Effects: “Dissecting the Evidence” | gutsandgrowth
- Do medicines work for GERD infants? | gutsandgrowth
- Failure of PPI test | gutsandgrowth
- One for the PPI team | gutsandgrowth