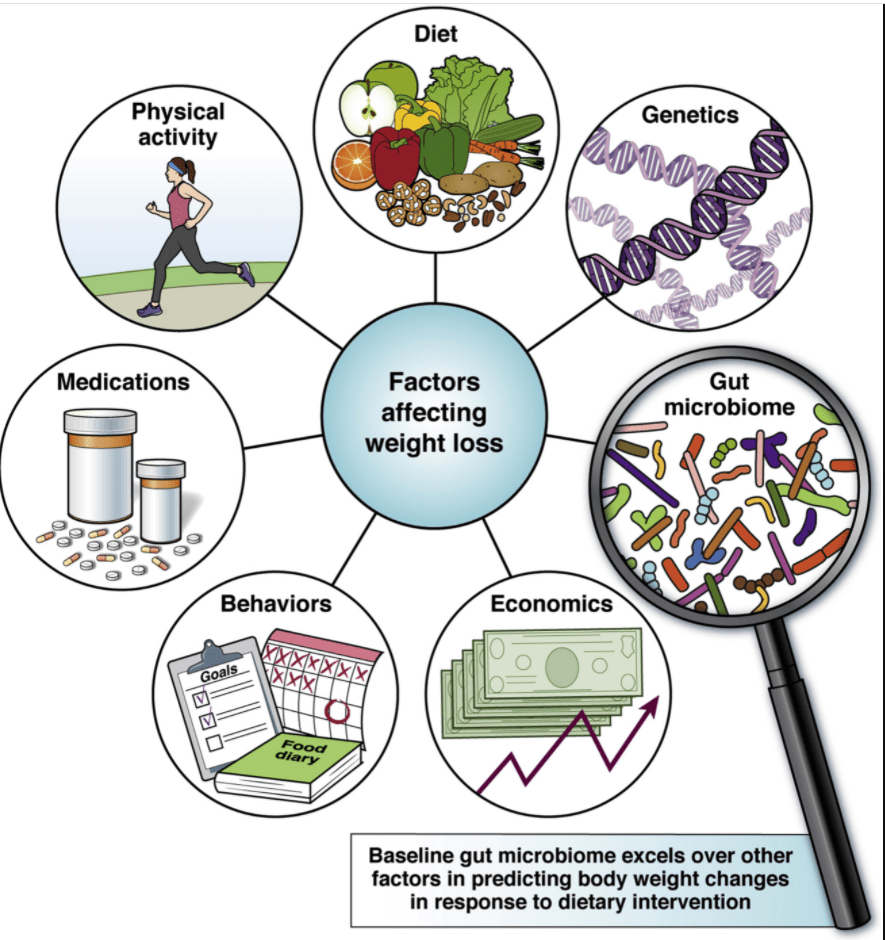
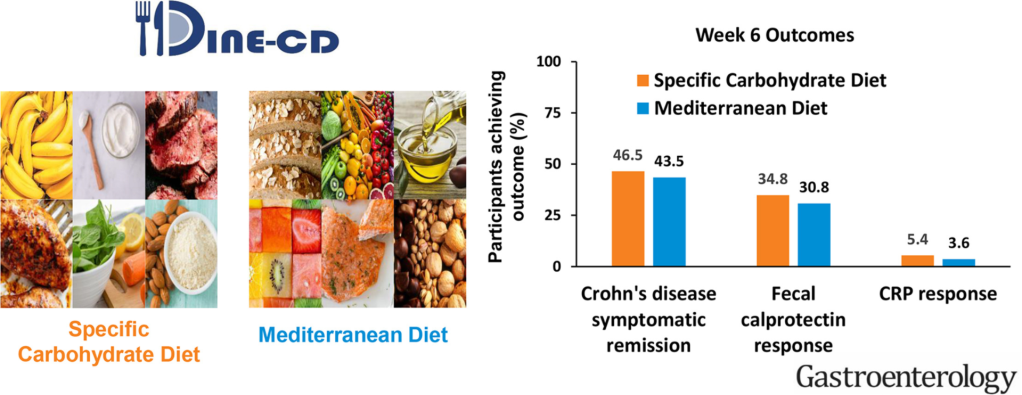
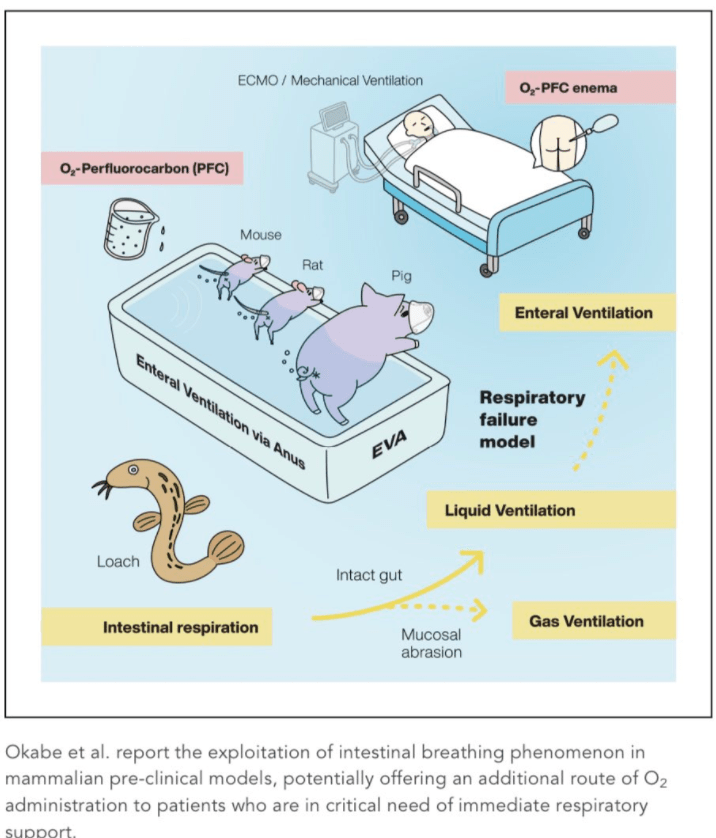
Briefly noted: Z Jie et al. Gastroenterol 2021; 160: 2029-2042. Full text PDF: The Baseline Gut Microbiota Directs Dieting-Induced Weight Loss Trajectories
METHODS: A 6-month weight-reduction program with longitudinal collection of dietary, physical activity, body weight, and fecal microbiome data as well as single-nucleotide polymorphism genotypes in 83 participants was conducted, followed by integration of the high-dimensional data to define the most determining factors for weight loss in a dietician-guided, smartphone-assisted dieting program
Key findings:
- 9 out of 83 subjects achieved long-term weight loss.
- The baseline gut microbiota was found to outperform other factors as a predicting predictor of individual weight loss trajectories
- Blautia wexlerae (MGS0575) and Bacteroides dorei (MGS0187) were the strongest predictors for weight loss when present in high abundance at baseline.
- The microbiome features were more predictive of weight loss than diet, physical activity, and obesity-related host genotype (based on single-nucleotide polymorphism genotypes)
In the associated editorial by RA Reimer et al (pg 1933-1935, full text: Dieting for Success: What Baseline Gut Microbiota Can Tell You About Your Chances of Losing Weight), the authors state that this study supports considering the gut microbiome “as a key component of individual response to dietary interventions.
My take: Much more work is needed in this area to tease out confounding variables (like baseline diet). It is intriguing that our gut microbiome could be instrumental in diet success and perhaps many other characteristics (eg. mental health, longevity, and susceptibility to diseases).
Related blog posts:
- Effects of Fecal Microbiome Transfer in Adolescents with Obesity (lack of effect)
- Not curing obesity with FMT & Remdesivir
- FMT Warning & ‘Get Your Butt in Gear’
- Could Obesity Be Cured/Created at Birth with Manipulation of Microbiome
- NY Times: Frequent Antibiotics May Make Children Fatter | gutsandgrowth
- Antibiotics Given Early in Life Linked to Childhood Obesity…Again | gutsandgrowth
Also NPR article (6/17/21): Bariatric Surgery Works, But Isn’t Offered To Most Teens Who Have Severe Obesity (at website, can also click link for 7 minute audio)