- C Melchior et al. Clin Gastroenterol Hepatol 2022; 20: 1290-1298. Open Access: Food Avoidance and Restriction in Irritable Bowel Syndrome: Relevance for Symptoms, Quality of Life and Nutrient Intake
- Associated editorial: HB Murray, K Staller. Clin Gastroenterol Hepatol 2022; 20: 1223-1225. Open Access: When Food Moves From Friend to Foe: Why Avoidant/Restrictive Food Intake Matters in Irritable Bowel Syndrome
Last week, this blog highlighted a study regarding the prevalence of ARFID in pediatric neurogastroenterology (Prevalence of Avoidant/Restrictive Food Intake Disorders in Pediatric Neurogastroenterology).
Today, this post reviews a study with 955 adult patients from 4 prospective studies who had completed the IBS Quality of Life Instrument (IBS-QOL). The 3 questions constituting the food domain were used to identify patients with reported severe food avoidance and restriction.
Key findings:
- In total, 13.2 % of the patients reported severe food avoidance and restriction, and in these patients all aspects of quality of life were lower (P < .01) and psychological, GI, and somatic symptoms were more severe (P < .05).
The associated editorial provides a lot of information on ARFID in this setting.
Key points:
- “The sine qua non of ARFID is a reduction in food intake, in terms of volume and/or variety, not primarily motivated by body image disturbance”
- “Motivations behind changes in eating in ARFID need to be 1 or more of 3 prototypical presentations: (1) fear of aversive consequences (eg, IBS symptoms), (2) a lack of interest in eating or low appetite, and (3) sensitivity to sensory characteristics of food (eg, taste, texture, smell)”
- “Weight suppression has similar deleterious health effects as is seen in anorexia nervosa, including cardiac abnormalities and bone mineral density loss”
- “Up to 90% of patients in IBS reporting avoidance of specific foods”
- “To identify presence of problematic avoidant/restrictive eating, there are ARFID measures validated with cutoffs (eg, the 9-item ARFID Screen;22,23 the PARDI-ARFID questionnaire).24 Nevertheless, more research is needed on the utility of these screening measures in IBS populations”
My take: Patients with ARFID and IBS need much more careful dietary counseling. So, it is important to consider the possibility of ARFID in this patient population.
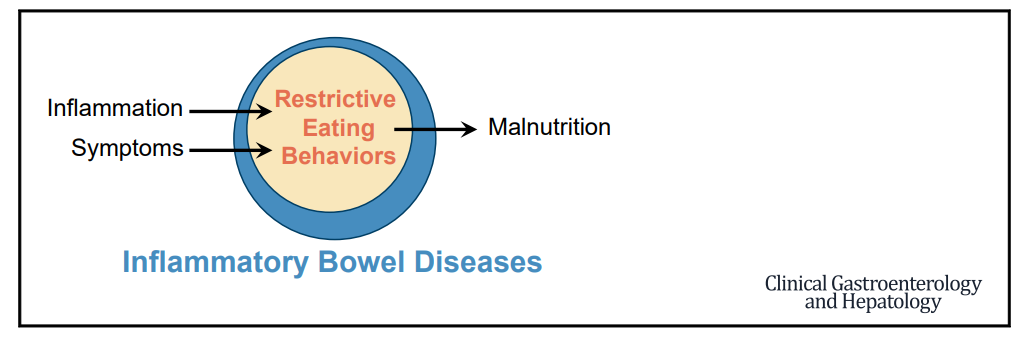
Related article: E Yelencich et al. Clin Gastroenterol Hepatol 2022; 20: 1282-1289. Open Access PDF: Avoidant Restrictive Food Intake Disorder Prevalent Among Patients With Inflammatory Bowel Disease In this cross-sectional study of adults with IBD, 28/161 (17%) had a positive ARFID risk score (>/=24). Most participants (92%) reported avoiding 1 or more foods while having active symptoms, and 74% continued to avoid 1 or more foods even in the absence of symptoms. Patients with a positive ARFID risk screen were significantly more likely to be at risk for malnutrition (60.7% vs 15.8%; P < .01)
Related blog post:
Afraid to Eat -Could be “Avoidant Restrictive Food Intake Disorder”