Thanks to Seth for sharing this twitter link: Holy Cow on Twitter (7 secs). The comments are amusing too.


Thanks to Seth for sharing this twitter link: Holy Cow on Twitter (7 secs). The comments are amusing too.

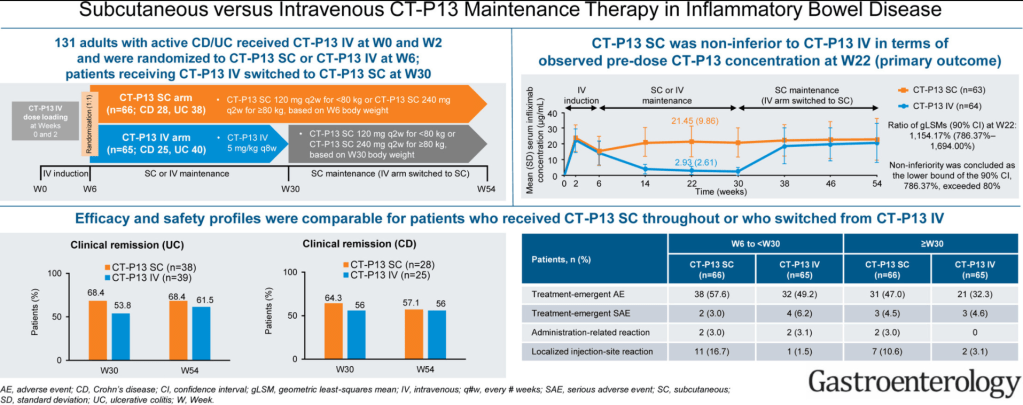
S Schreiber, S Ben-Horin et al. Gastroenterol 2021; 160 2340-2353. Full text: Randomized Controlled Trial: Subcutaneous vs Intravenous Infliximab CT-P13 Maintenance in Inflammatory Bowel Disease
Methods: Overall in this phase 1 randomized, open-label study in patients with either ulcerative colitis or Crohn’s disease, 66 and 65 patients were randomized to CT-P13 SC (every 2 weeks) and CT-P13 IV, respectively
Key findings: The pharmacokinetic noninferiority of CT-P13 SC to CT-P13 IV, and the comparable efficacy, safety, and immunogenicity profiles
These findings are in agreement with similar studies performed in patients with Rheumatoid Arthritis.
My take: If confirmed with additional studies, it is likely that SC infliximab treatment will be a useful alternative to intravenous infliximab. This is similar to data presented with vedolizumab which is currently administered intravenously.
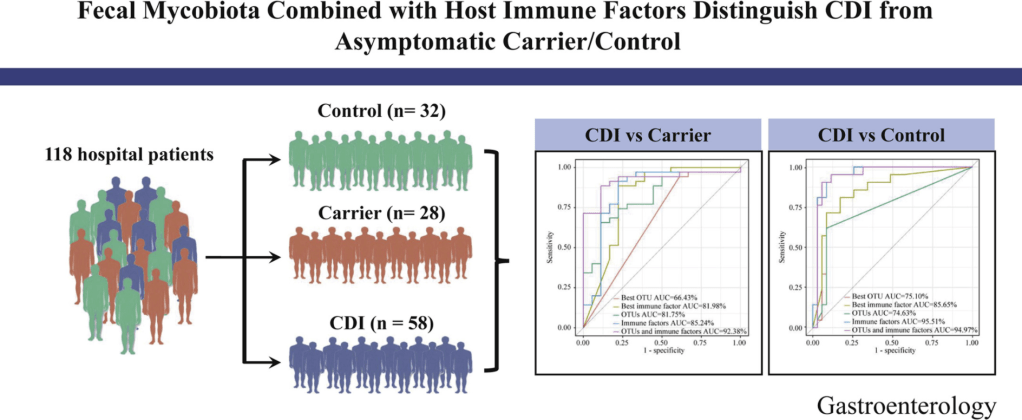
Y Cao et al. Gastroenterol 2021; 160: 2328-2339. Fecal Mycobiota Combined With Host Immune Factors Distinguish Clostridioides difficile Infection From Asymptomatic Carriage
Key findings:
My take: It is interesting that fecal fungal diversity (mycobiome), in addition to bacterial diversity, is reduced in those with Clostridium difficile infection (CDI) compared to both control groups and those with Clostridium difficile asymptomatic carriage.
Related blog posts:

A recent study (P Mansueto et al. Am J Gastroenterol 2021; 116: 1015-1023. Autoimmunity Features in Patients With Non-Celiac Wheat Sensitivity). Thanks to Ben Gold for this reference.
The authors prospectively and consecutively studied 91 patients with Non-Celiac Wheat Sensitivity (NCWS) (F?M ratio 7:1); 76 healthy blood donors (HBD) and 55 patients with a diagnosis of irritable bowel syndrome (IBS) unrelated to NCWS served as controls.
NCWS was diagnosed based on absence of celiac serology, absence of villous atrophy (while receiving a gluten-containing diet), negative IgE-testing for wheat allergy (either serum or skin prick tests) along with resolution of symptoms off wheat and symptom reappearance wiht a DBPC wheat challenge.
Key findings:
The associated editorial by Galipeau et al notes that only 16% of those who self-report as “gluten-sensitive” will actually fulfill the current consensus criteria for a diagnosis of NCWS. The diagnosis is problematic because of the absence of a validated biomarker. While the current study shows association with autoimmune markers, other studies have shown some have markers of immune activation and others with non-IgE-mediated food sensitivities.
My take: These type of studies help us understand NCWS. Yet, without a more definitive biomarker, many people will be on a gluten-free diet needlessly.
Related blog posts:

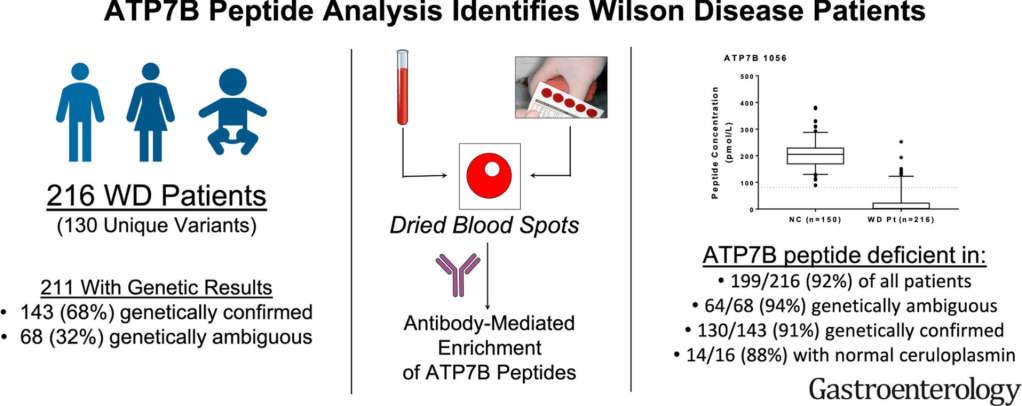
CJ Collins, F Yi et al. Gastroenterol 2021; 160: 2367-2382. Full text: Direct Measurement of ATP7B Peptides Is Highly Effective in the Diagnosis of Wilson Disease
There continues to be challenges in the diagnosis of Wilson’s disease (WD). Genetic testing, per the authors and Vasrome (varsome.com), have found more than 649 pathogenic mutations and another 692 mutations that are VUS. Definitive diagnosis with genetic testing requires 2 known pathogenic variants. Other features, including Kayser-Fleischer rings and ceruloplasmin, have limited sensitivity and/or specificity.
Methods: Two hundred and sixty-four samples from biorepositories at 3 international and 2 domestic academic centers and 150 normal controls were used.
Key findings:
Discussion: As with other tests, ATPB7 peptide testing has limitations. Most patients with WD have pathogenic mutations that often result in protein misfolding, absence of decay of messenger RNA and enhanced degradation; hence, low ATPB7 levels; however, disease-causing mutations that affect protein activity but not protein concentration will generate false-negative results.
My take: “ATP7B peptide analysis identified WD patients in a large majority of cases and reduced ambiguities resulting from genetic analysis and Cp (ceruloplasmin) levels. This noninvasive assay can serve as an adjunctive test for the diagnosis of WD and is expected to fundamentally advance the use of proteomic technology for a rapid screening tool.
Related blog posts:
NY Times: Covid Is a Greater Risk to Young People Than the Vaccines (July 4, 2021)
This article elaborates on the risks of vaccination, especially due to myocarditis, compared to the risks posed by COVID-19 infection. Even using very cautious estimates, the authors find that the risks of hospitalizations, cardiac morbidity, and deaths are likely to be much lower in those who receive the vaccine.
Key points:
My take: 12-17 year olds are at less risk from COVID-19 infection than other age groups, however, this risk is still greater risk than the risk of vaccination. Protecting them with immunizations also protects other vulnerable populations and may decrease the risk of vaccine-resistant variants.
Related article: Eric Topol NY Times: It’s Time for the F.D.A. to Fully Approve the mRNA Vaccines An excerpt: “Now more than 180 million doses of the Pfizer vaccine and 133 million of Moderna’s have been administered in the United States, with millions more doses distributed worldwide. In the history of medicine, few if any biologics (vaccines, antibodies, molecules) have had their safety and efficacy scrutinized to this degree…it’s frankly unfathomable that mRNA vaccines have been proved safe and effective in hundreds of millions of people and yet still have a scarlet “E”.”

A recent study (F Fernandez-Banares et al. Am J Gastroenterol 2021; 116: 1036-1043. Persistent Villous Atrophy in De Novo Adult Patients With Celiac Disease and Strict Control of Gluten-Free Diet Adherence: A Multicenter Prospective Study (CADER Study) shows that there is a high likelihood of persistent villous atrophy among adults with celiac disease (CD) despite adherence with a gluten-free diet (GFD). Thanks to Ben Gold for showing me this paper.
Key findings:
Discussion:
Editorial:
My take: This study shows that there is ongoing gluten exposure in the majority of patients even in those with excellent or good adherence to a GFD; in addition, it shows that clinical/serological markers are NOT effective in predicting mucosal healing in adults. Nevertheless, it is not clear that followup endoscopy is beneficial.
Related blog posts:

Forbes (7/1/21): 99.5% Of People Killed By Covid In Last 6 Months Were Unvaccinated, Data Suggests

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition
A recent retrospective single-center in Turkey study (2013-2018) reports 5 cases of colorectal cancer (CRC).
E Polat et al. JPGN Reports; 2021 – Volume 2 – Issue 1 – p e039 Full text: Colorectal Carcinoma in Childhood
Key points:

K Vaidy et al. JPGN Reports 2020; Full text: Treatment of Pyoderma Gangrenosum in Pediatric Inflammatory Bowel Disease
This in-depth report reviews pyoderma gangrenosum including the differential diagnosis, the pathophysiology/genetics, presentation/diagnosis and treatment approaches. Anti-TNF therapy: “Currently available published data support using an anti-TNF-α biologic agent as first-line therapy for severe PG therapy in pediatric IBD, as well as for those cases that have not responded to local therapies.”

Related blog posts -PG:
Most often a letter to the editor would not grab my attention. A recent letter did: Full Text: Tofacitinib Is Safe and Effective When Used in Combination With Infliximab for the Management of Refractory Ulcerative Colitis (R Gilmore et al. Clin Gastroenterol Hepatol 2021; 1302-1303; reply 1303-1304 by JA Berinstein et al.)
This reported case series with 5 patients with severe ulcerative colitis (UC) who received a combination of tofacitinib and infliximab for at least 90 days were retrospectively reviewed. Tofacitinib dosing was de-escalated to 5 mg twice daily after 8 weeks. Thiopurine therapy was stopped with tofacitinib initiation.
Key findings:
The authors letter title regarding tofacitinib being “safe and effective” is clearly overstated. The reply notes that in limited experience the group from the University of Michigan had a 50-year-old man develop severe pulmonary and CNS disease due to acquisition of legionnaires disease while on combination tofacitinib and infliximab.
My take: (borrowed from reply) “Efficacy and safety data obtained through rigorous randomized trials are needed…it is possible that long-term use of combination tofacitinib and infliximab will lead to an unacceptable risk of infection.”
Another study of tofacitinib: GR Lichtenstein et al. Inflamm Bowel Dis 2021; 27: 816-825. Tofacitinib, an Oral Janus Kinase Inhibitor: Analysis of Malignancy (Excluding Nonmelanoma Skin Cancer) Events Across the Ulcerative Colitis Clinical Program Key finding: With an exposure of 2576.4 patient years & 124 overall cohort tofacitinib-treated patients, 20 developed a malignancy
Related blog post:

