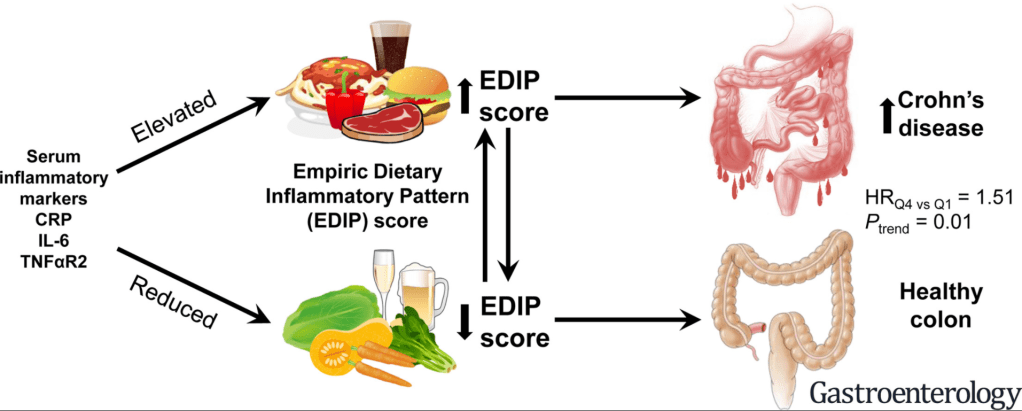
A recent study (AG Feldman et al. J Pediatr 2020; 225: 252-258. Subacute Liver Failure Following Gene Replacement Therapy for Spinal Muscular Atrophy Type 1) describes two children who developed subacute liver failure after treatment with onasemnogene (AVXS-101). This gene therapy was approved by the FDA in 2019 and more than 335 children have been treated. Both children presented about 3-8 weeks after their AVXS-101 infusion (despite steroid therapy), at 6 months of age and 20 months of age respectively, with ALT values above 1600 and INR of at least 1.5 (despite Vitamin K). Both had liver biospies and then were treated with methylprednisolone, starting at 20 mg/kg/day.
Key points:
- The authors speculate that subacute liver failure was due to a systemic hyperinflammatory reaction
- The authors recommend screening prior to AVXS-101 therapy with LFTs, GGT, and INR; if baseline labs are elevated, further workup is recommended (eg. A1AT, HBV, HCV, ANA, anti-SMA, anti-LKM, and ultraound)
- While this reaction has been with AVXS-101, there are other gene therapies with adenovirus-vector which could trigger similar reactions
- The authors note that the “package insert for onasemnogene recommends prednisolone (1 mg/kg/day) should be given in the 24 hours before infusion and should be continued for 30 days after infusion.”
- After infusion, it is “necessary to monitor liver tests frequently in the first 2 months”
My take: This new therapy’s risks are substantial; however, the benefits from treatment can be life-altering as well.
Related blog post: Understanding the New Therapies for Spinal Muscular Atrophy