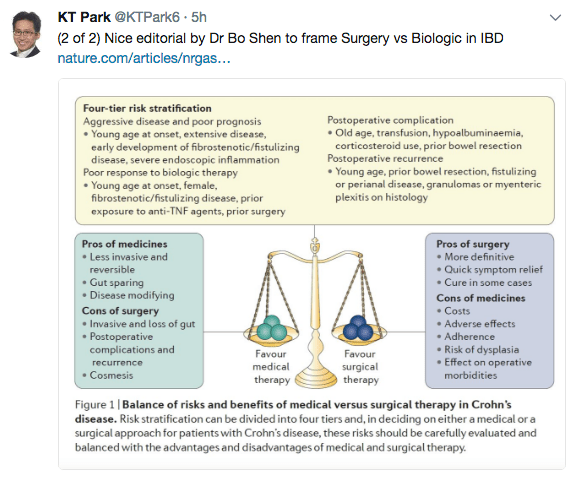
Continued from yesterday…
The third study (HM Kansen et al. JPGN 2017; 65: 425-29) retrospectively (2009-2014) examined 162 children (with available data) with Crohn’s disease (CD) for the development of anti-infliximab antibody (ATI) while receiving either monotherapy or concomitant therapy. This was a collaborative study from the Kids with Crohn’s Colitis (KiCC) working group (Netherlands). In the majority of their patients (222 of 229), IFX was initiated as step-up therapy. Key findings:
- 15% developed ATIs
- 6 of 62 (10%) developed ATIs while receiving ongoing concomitant immunosuppression
- 11 of 81 (14%) developed ATIs after receiving early concomitant immunosuppression (median of 6.2 months of concomitant immunosuppression followed by IFX monotherapy) 10 of 11 who developed ATIs within the first 12 months, compared to 1 of 26 (4%) after 12 months.
- 8 of 19 (42%) developed ATIs on IFX monotherapy
In their discussion, the authors note concerns regarding the safety of thiopurines. However, they point out that “the benefit of combination therapy (reduction of ATI formation) relative to IFX monotherapy should outweigh the risk of serious infections and malignancies to achieve an optimal treatment strategy for paediatric CD.” The authors: “combination therapy for approximately 12 months from initiation of IFX, followed by IFX monotherapy, may be equally effective alternative to continuous combination therapy.”
Overall, the totality of the evidence favors combination therapy for most patients with CD. Yet, there is wide variation in clinical practice. As I was thinking about this, I came across a recent commentary: “The Power of Regret” (J Groopman, P Hartzband. NEJM 2017; 377: 1507-9). The authors note that “disappointment is an unavoidable aspect of making difficult choices…but disappointment is not associated with self-recrimination and thus differs notably from regret.” They indicate that “process regret” occurs when patients do not consider information about all available choices before making a decision. I wonder if many patients/families fear using concomitant therapy because they worry they will regret the decision if a complication occurs. Perhaps, working with all available information, some reluctant patients/families will feel better about their decision if the process for their decision was thorough, considering the risks/benefits of the treatment but also the risks/benefits of not choosing the treatment. .
My take: Overall, for most pediatric patients with CD, to date, concomitant therapy has been the most effective treatment. More prospective studies are needed to determine more conclusively the benefit and optimal duration/timing of combined therapy, particularly with the more frequent use of therapeutic drug monitoring. Also, as will be noted in future posts from annual meeting, thiopurine use is declining.
Related blog posts:
- Combination therapy with adalimumab –is it helpful?
- ‘Don’t Believe Our Study’
- Should All Pediatric Patients with Crohn’s Disease Continue …
- Don’t be Fooled About Withdrawing Immunomodulator …
- Digging into the COMMIT Study | gutsandgrowth
- Support for Step-Up Therapy and Thiopurines | gutsandgrowth
- More NASPGHAN Meeting Notes –Hot Topics