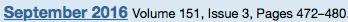Small Pediatric IBD Studies …Briefly Noted
G Wahbeh et al. JPGN 2016; 63: 348-51. This retrospective case series with 4 children (aged 12-17 years) indicated that 2 had a ‘clinical response’ to ustekinumab therapy, though one of these had ongoing elevation of CRP. The dosing may have been too low: 90 mg at week 0 and 4, then every 8 weeks. My take: This study shows that ustekinumab’s use in pediatric IBD seems to be a ‘shot in the dark’ given the lack of coherent data.
Related posts:
- More on Ustekinumab, plus Allopurinol Study | gutsandgrowth
- Using Ustekinumab for Crohn’s Disease | gutsandgrowth
L Zimmerman et al. JPGN 2016; 63: 352-56. Among a cohort of 123 children who had underwent a bowel resection, from 1977-2011, the overall postoperative complication rate was 13%. This included 3 of 24 who had prior infliximab and 9 of 99 who had not received infliximab. It is noteworthy that the infliximab group had more corticosteroid exposure. The authors concluded that preoperative infliximab was not associated with increased complications but noted that their sample size was small. My take: Studies of adults with Crohn’s disease have yielded conflicting results on whether preoperative infliximab increases the risk of complications. This study shows that children likely have a lower rate of postsurgical complications and more pediatric specific data are needed.
Related post:
From KT Park’s Twitter Feed:
Latest on Tofacitinib for Refractory Ulcerative Colitis
From Gastroenterology & Endoscopy News July 2016: Tofacitinib Effective in Refractory and Severe UC
An excerpt:
Tofacitinib (Pfizer), an oral agent already approved for certain patients with rheumatoid arthritis, can induce clinical remission in up to 25% of individuals with moderate to severe, refractory ulcerative colitis (UC) and clinical response in as many as 60% of these patients.
The results, based on two placebo-controlled trials involving more than 1,100 patients, showed the drug also increased the risk for serum lipid elevations but was otherwise safe. Researchers presented the data at the 2016 annual meeting of the European Crohn’s and Colitis Organization (ECCO; oral presentation 019)…
The new data are from the OCTAVE Induction 1 and Induction 2 trials, identically designed, randomized, double-blind and placebo-controlled Phase III studies…In the OCTAVE 1 trial, 476 patients received 10 mg of tofacitinib orally twice daily for eight weeks and 122 received an oral placebo. In OCTAVE 2, 429 and 112 patients were randomized to receive the two regimens, respectively.
Also from Gastroenterology & Endoscopy News August 2016: Update on Diagnosis and Treatment for Ulcerative Colitis This article provides a succinct summary regarding diagnosis and treatments of ulcerative colitis; treatments discussed include emerging therapies like tofacitinib.
Early Preview of Basic Science Review: Biliatresone
Generally, one of the best lectures at our national meeting is the “Basic Science Year in Review.” I would be surprised if a recent study (O Waisbourd-Zinman et al. Hepatology 2016; 64: 880-93) does not get some attention during this review.
Even though biliary atresia (BA) remains the leading cause of pediatric liver transplantation, the exact reasons for its development have not been elucidated. There are data “implicating both immune dysregulation and genetic factors in human BA. Toxin-induced BA is not inconsistent with these findings and may represent a primary injury, with immune dysregulation representing a secondary insult.”
One of the reasons for suspecting a toxin dates back to outbreaks of a BA-like disease in newborn lambs that occurred in New South Wales in 1964 and 1988. During both these periods, severe droughts led to pregnant livestock grazing on atypical flora. Ultimately, a plant toxin termed “biliatresone” was isolated from Australian plants (Dysphania species).
Here’s how this study advanced the science on biliatresone:
- The authors treated mouse cholangiocytes in 3D spheroid culture and neonatal extrahepatic duct exlplants with biliatresone and compounds that regluate glutathione (GSH)
- The authors determined the effects of biliatresone on SOX17 levels and the effects of Sox17 knockdown on cholangiocytes in 3D culture
Key findings:
- “Biliatresoe caused disrupton of cholangiocyte apical polarity and loss of monolayer integrity.”
- “Neonatal bile duct explants treated with the toxin showed lumen obstruction with increased subepithelial staining for α-smooth muscle actin and collagen, consistent with fibrosis”
- “Biliatresone caused a rapid and transient decrease in GSH…and caused a significant decrease in cholangiocyte SOX17, and Sox17 knockdown in cholangiocyte spheroids mimicked the effects of biliatresone.”
- These findings are easiest to appreciate in their figures, particularly Figure 4 and 5.
While pregnant women are not likely exposed to biliatresone, the authors showed that the effects of the toxin on lowering GSH was sufficient for cholangiocyte injury.; in addition, they showed that “SOX17 is required to maintain the epithelial architecture of the gallbladder and the cystic duct.” Thus, there are likely other exposures that could lead to similar outcomes
My take: I will let Dr. Barnard explain the elegant experiments. This study strongly supports maternal dietary factors as a contributing role in the pathophysiology of BA. Now identifying these teratogens is crucial.
Also noted: X Zhao et al. Hepatology 2016; 64: 894-907. This study “strongly support redox stress as a critical contributing factor in biliatresone-induced cholangioctye injury” in Zebrafish. Specifically, the authors identified that gene transcripts involved in redox stress, particularly regarding glutathione were upregulated after exposure to biliatresone.
Related blog posts:
What to Make of A Motility Study of Children with Orthostatic Intolerance
While a recent study (A Darbari et al JPGN 2016; 63: 329-35) provides some interesting data regarding the potential origin of gastrointestinal symptoms in the setting of orthostatic intolerance, I cannot support their conclusion that antroduodenal manometry (ADM) “should” be part of the evaluation of these affected children.
Background:
- Retrospective study which included only subjects with a positive tilt test
What’s interesting:
- Among 35 children with orthostatic intolerance due to either neurally mediated hypotension (NMH) or postural orthostatic tachycardia syndrome (POTS), ADM was abnormal at baseline or during tilt table testing in 26 (75%).
- ADM studies were more often abnormal than gastric emptying studies, which were normal in 12 or 25.
- Specific findings included neurogenic intestinal dysmotility in 15, antral hypomotility in 4, visceral hyperalgesia in 2, and regurgitation in 5.
- GI symptoms of nausea, abdominal pain or vomiting were reproduced during tilt testing in 31 of 35 patients (89%).
Based on the discussion, the authors imply that ADM testing could help determine if the symptoms are due to neurogastrointestinal pathology or if normal, could indicate a central origin for the GI symptoms. Thus, they conclude that motility testing “should” be part of comprehensive” orthostatic intolerance evaluation.
I would argue that this study does not show that ADM testing can reliably distinguish whether symptoms are due to a neurogastroenterological pathology or central pathology. And, in fact, there are better tests to examine for central origin. I wouldn’t be surprised if many of their subjects had brain imaging, though this is not reported.
In addition, the authors acknowledge that ADM testing may not influence therapeutic decisions. “The clinical response to promotility agents in children with POTS is generally low.”
My take: This study provides a useful mechanistic explanation of symptoms associated with orthostatic intolerance. However, “I’m not there yet” on supporting ADM for all children with OI.
Related blog post:
Cutting Edge for Endoscopic Control of Bleeding
A recent review elaborates on the newest methods for endoscopic control of bleeding. Topics included caplock clips, endoscopic suturing, and hemostatic sprays.
Full text: New Endoscopic Technologies and Procedureal Advances for Endoscopic Hemostasis (from Clinical Gastroenterology and Hepatology)
Related blog posts:
- All Bleeding Stops (part 2) | gutsandgrowth
- All bleeding stops | gutsandgrowth
- Watch the Bleeding Ulcer Stop! | gutsandgrowth
Latest Vaccine Recommendations
As a pediatric specialist, I do not administer vaccines in our office; at times I wonder how the recommendations for immunizations may have changed. Relatively new vaccines include HPV and MenB. For those who want an up-to-date guidance for 2016 from CDC/ACIP:
- For adults:http://www.cdc.gov/vaccines/schedules/downloads/adult/adult-schedule-easy-read.pdf Latest guideline changes discussed: Ann Intern Med. 2016;164(3):184-194. doi:10.7326/M15-3005 & specific conditions vis-a-vis vaccines reviewed in Table.
- For teens:http://www.cdc.gov/vaccines/who/teens/downloads/parent-version-schedule-7-18yrs.pdf
- For infants/children: http://www.cdc.gov/vaccines/parents/downloads/parent-ver-sch-0-6yrs.pdf
HBV Vaccination Prevents Cancer
In a Gastroenterology study from Taiwan, HBV vaccination has been shown to reduce the risk of Hepatocellular Carcinoma.
Results
Of the 1509 patients with HCC, 1343 were born before, and 166 were born after, the HBV vaccination program began. HCC incidence per 105 person-years was 0.92 in the unvaccinated cohort and 0.23 in the vaccinated birth cohorts. The RRs for HCC in patients 6–9 years old, 10–14 years old, 15–19 years old, and 20–26 years old who were vaccinated vs unvaccinated were 0.26 (95% confidence interval [CI], 0.17–0.40), 0.34 (95% CI, 0.25–0.48), 0.37 (95% CI, 0.25–0.51), and 0.42 (95% CI, 0.32–0.56), respectively. The RR for HCC in 6- to 26-year-olds was lower in the later vs the earlier cohorts (born in 1992–2005 vs 1986–1992; P < .001 and 1986–1992 vs 1984–1986; P < .002). Transmission of HBV from highly infectious mothers and incomplete immunization were associated with development of HCC.
Celiac Hepatopathies 2016
A recent study (GJ Lee et al. JPGN 2016; 63: 340-3) adds a little bit more information regarding hypertransaminasemia in newly diagnosed celiac disease. Some previous information was summarized in a previous blog: Celiac Hepatopathies (2013)
In this retrospective, single center study, 185 children had transaminases obtained at the time of celiac diagnosis (185/388 = 47.7%).
Key findings:
- Among this group, 28 (15.1%) had elevated transaminases, with an average of ALT 2.52 x ULN and AST 1.87 x ULN.
- Patients with elevated liver transaminases tended to be younger (mean 6.3 yrs compared with 11.0 without elevation). Among those who had followup blood testing available, 15/21 (71.4%) normalized their values over an average of 210 days.
- For the 6 who had persistent elevation of transaminases, 3 were suspected to have poor adherence, 1 was thought to have a fatty liver, 1 had only mild elevation, and 1 remained unexplained.
My take: This study indicates that elevated transaminases are common in children with celiac, particularly younger children. As with other studies, the majority resolve on a gluten-free diet. As there is a recognized association with autoimmune hepatitis, in those with elevated ALT, followup after institution of a gluten free diet seems prudent.
Hepatitis E Update
Briefly noted:
B Fischler et al. JPGN 2016; 63: 288-94. This position paper by ESPGHAN suggests testing children with increased transaminases for hepatitis E virus (HEV) and notes that immunocompromised children with HEV may require treatment (with ribavirin or lowering immunosuppression). Best tests: serology for IgM and IgG and HEV RNA PCR.
F Huang et al Hepatology 2016; 64: 350-59. This study from China showed frequent Hepatitis E excreted in milk from infected cows and that this milk is capable of transmit infection when given to rhesus macaques, even if pasteurized. Also, the article notes that seroprevalence of HEV in recent years has been higher than prior estimates (21% from 1988-94 and 6% from 2009-2010).