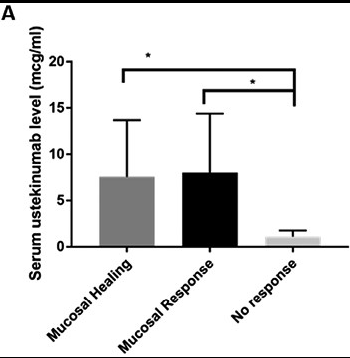
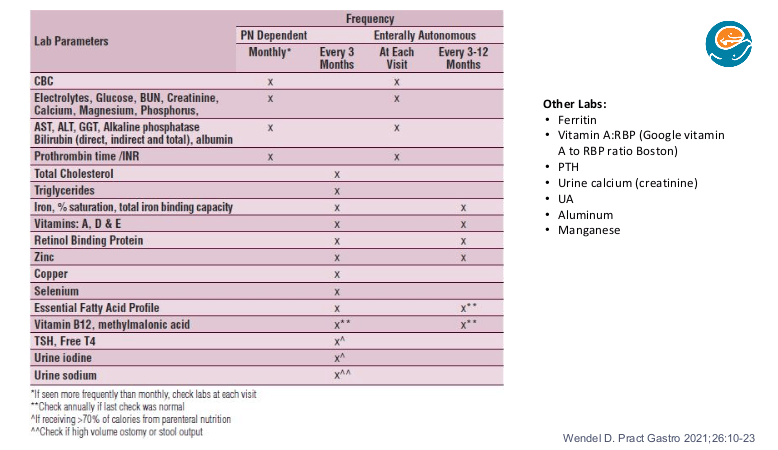
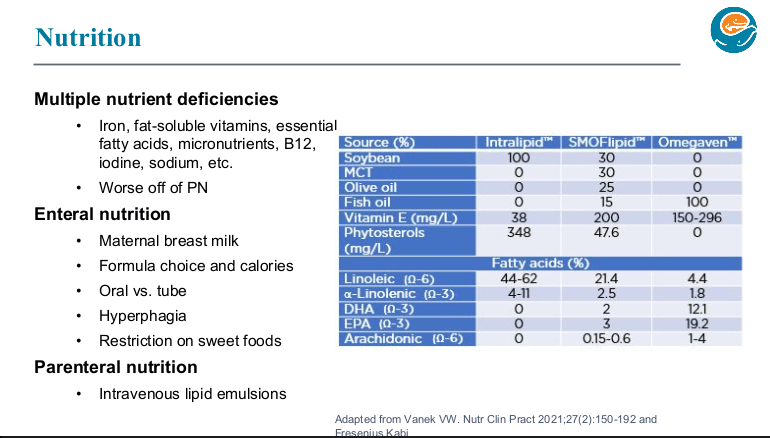
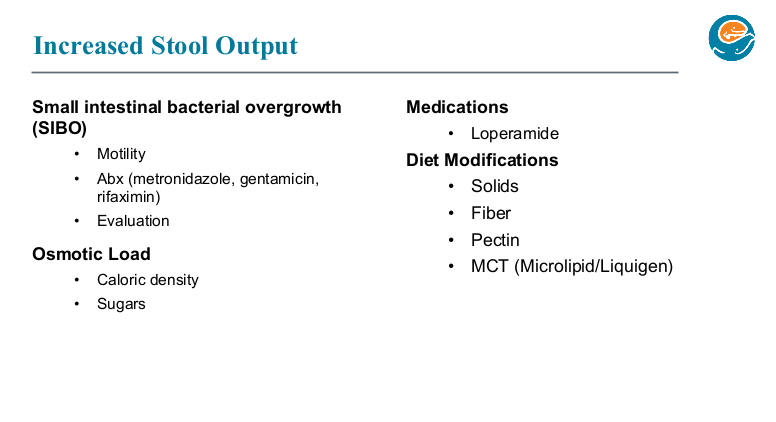
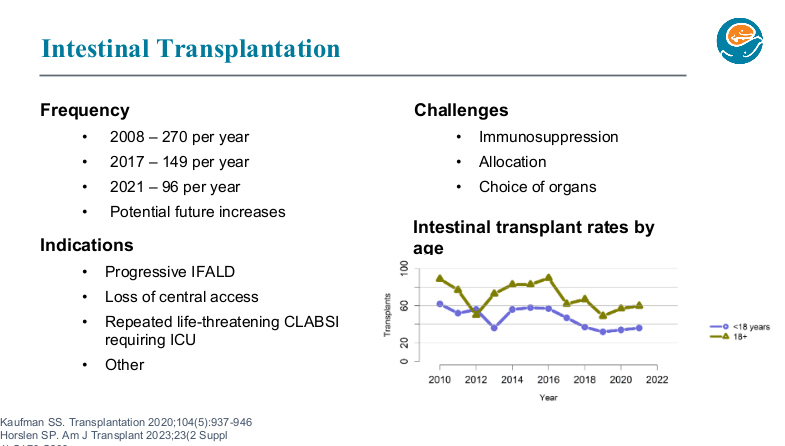
K Gkikas et al. Inflamm Bowel Dis 2024; Volume 30, March 2024, 347–356. Open Access! YouTube as a Source of Information for Food, Diet-Related Items, and Advisory Comments for the Management of Inflammatory Bowel Disease
The authors assessed videos discussing dietary aspects (food, diet-related items, and advisory comments [FODRIACs]) on YouTube. Of 1800 videos screened, a total of 160 were included in the final analysis.
Background: “Currently, the only well-established dietary treatment in IBD is exclusive enteral nutrition (EEN), which is used for induction of remission mostly in pediatric CD.5,6 Over the past 2 decades, several food-based exclusion diets have been suggested as potential treatments for CD and UC, some of which demonstrated promising efficacy signals such as the CD-TREAT (Crohn’s disease treatment with eating) diet and the CDED (Crohn’s disease exclusion diet).7-9 Nonetheless, current societal guidelines do not recommend the use of any solid food–based exclusion diet as a treatment option for the induction or maintenance of clinical remission in IBD.6“
Key findings:
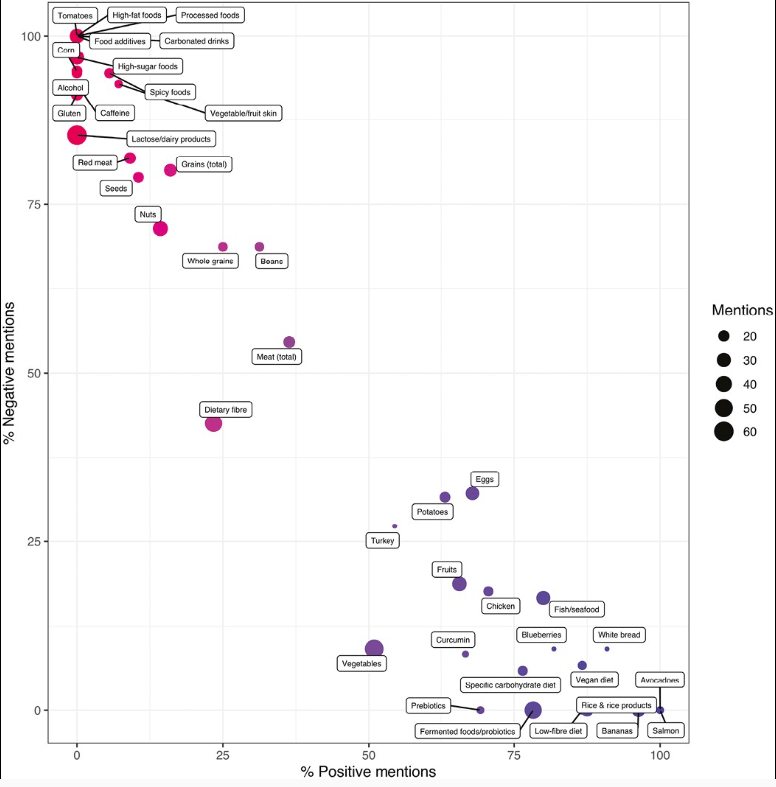
- Foods pertinent to a prudent dietary pattern (ie, fish, chicken, avocado, blueberries), foods high in pre- and probiotics, and certain food exclusion diets (eg, SCD) were primarily portrayed as beneficial.
- Foods often associated with a Western dietary pattern, including processed foods, high-sugar foods and high-fat foods, red meat, and alcohol, were considered detrimental for disease outcomes in patients with IBD.
- Neutral opinions were expressed about fiber and vegetables.
- There was a higher video interaction rate and number of likes in patient-generated videos compared with videos from healthcare professionals.
- Only 3% of all patient videos and 35% of videos from healthcare professionals cited any form of scientific evidence.
- Problems with diet advice is that extensive dietary restrictions and adherence to extreme diets may lead to the development of disordered eating as well as nutrient deficiencies (nutritionist involvement is important to avoid this). In addition, reliance on unproven diets may lead some patients to forgo proven therapies.
My take: Though there are some overlapping advice in these videos, much of the dietary advice on YouTube (and elsewhere) is conflicting. In part, this reflects the lack of evidence-based dietary guidelines for IBD. Physicians should review information on dietary therapies at diagnosis and request that families contact them (or well-qualified nutritionists) when considering dietary modifications.
Related blog posts:
- IBD Updates: Preventing Inflammatory Bowel Disease with a Healthy Diet and Medication Safety Pyramid
- EEN: It Only Works If You Do It
- Dietary Therapy for Inflammatory Bowel Disease This is good lecture review on dietary therapy
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- Trial by Diet for Pediatric Crohn’s Disease
- Can IBD Be Treated with Diet Alone?
- Good Food and Bad Food for Crohn’s Disease -No Agreement | gutsandgrowth
- Pushing the Boundaries on Dietary Therapy for Crohn’s Disease: CD-TREAT
- Position Paper: Nutrition in Pediatric IBD
- CDED + PEN: An Alternative Diet to Exclusive Enteral Nutrition?