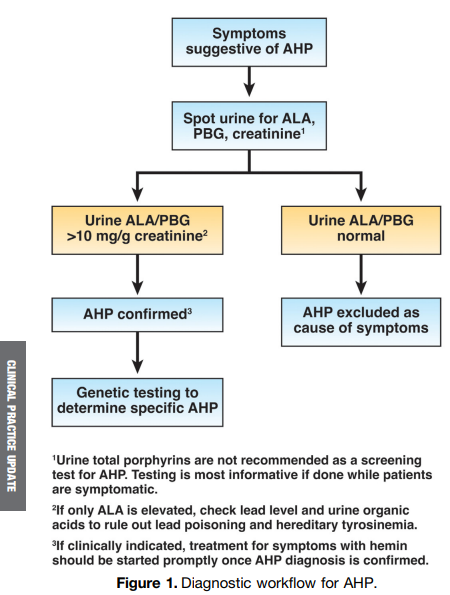
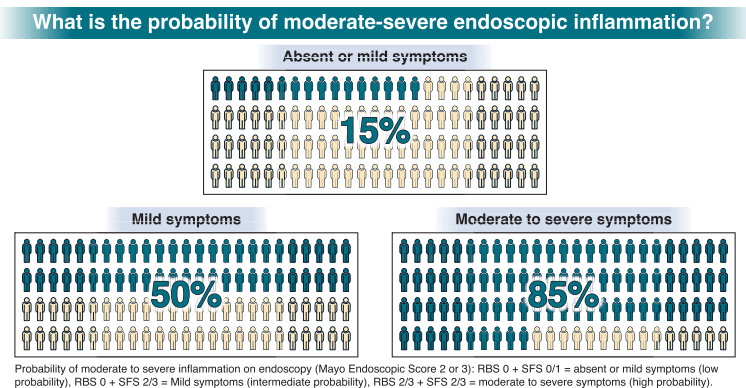
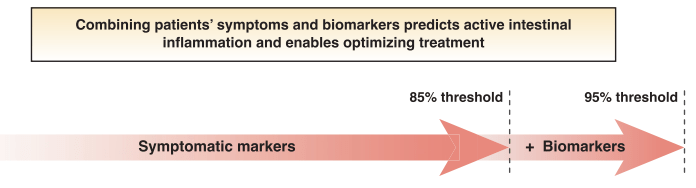
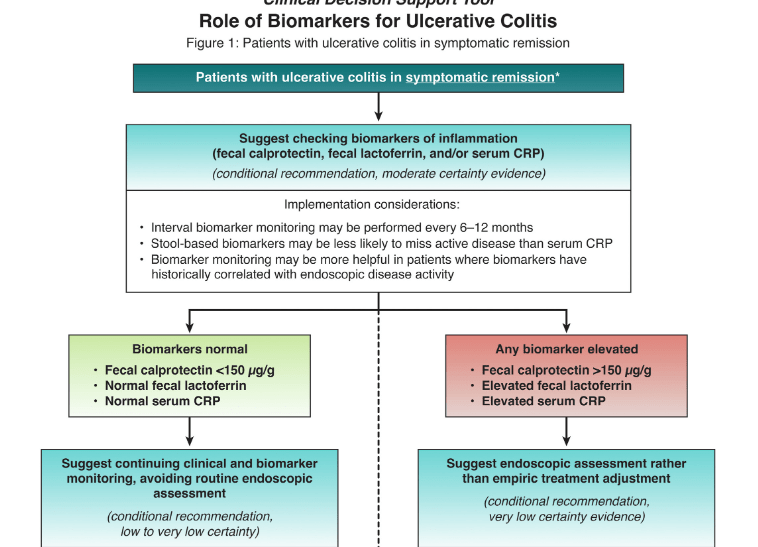
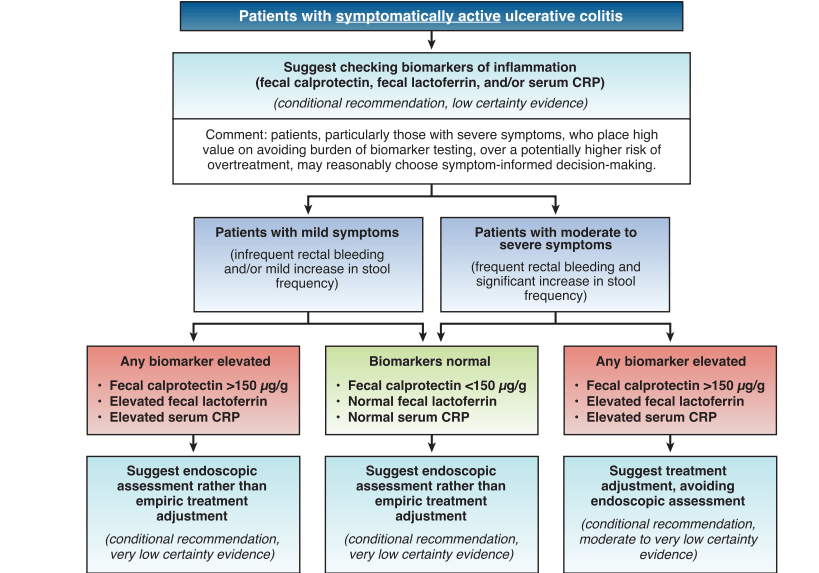
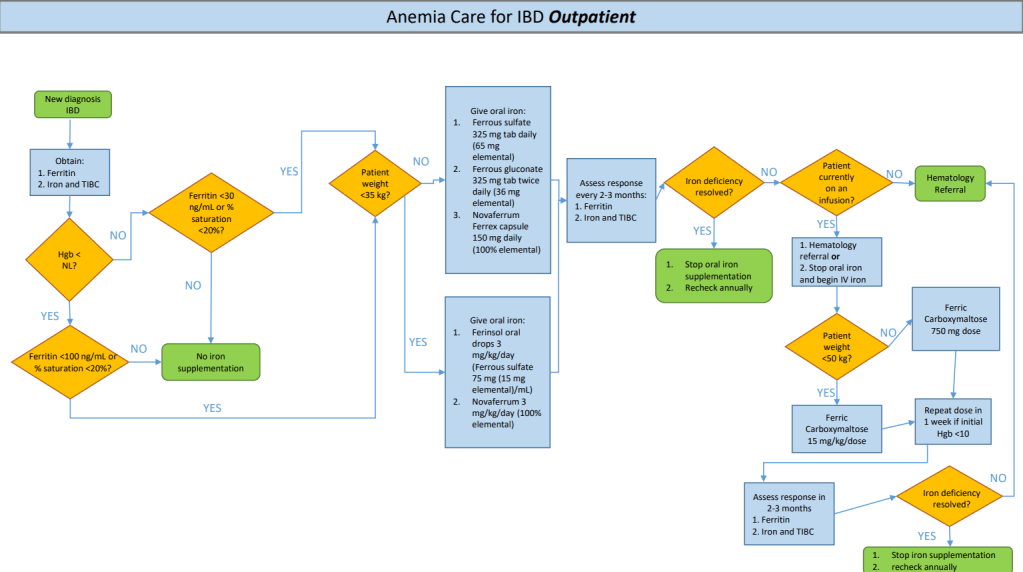
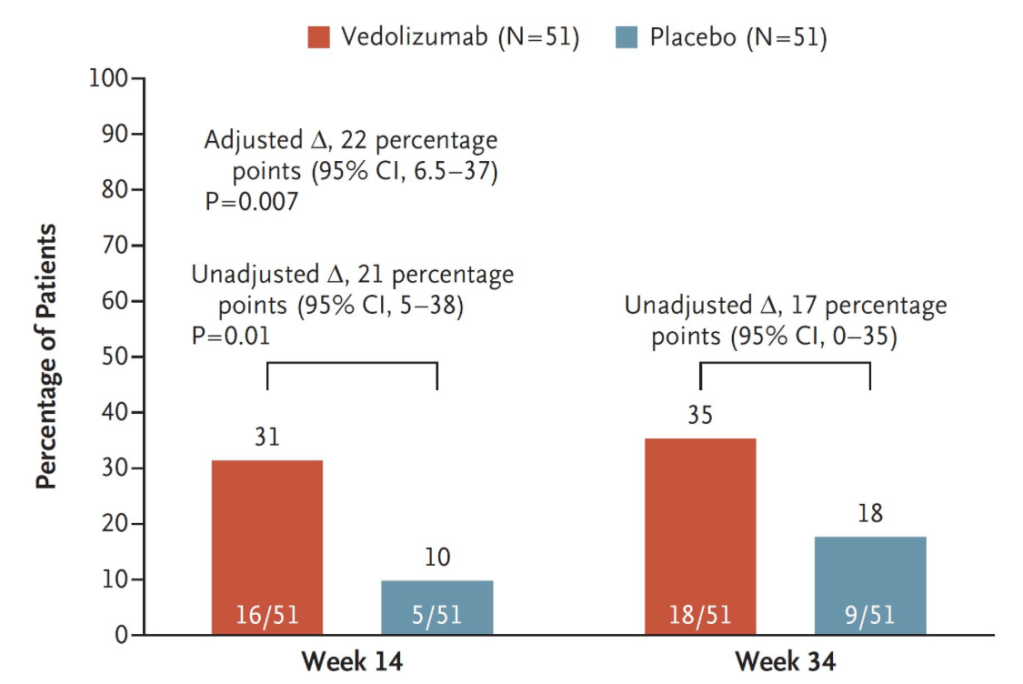
S Travis et al. NEJM 2023; 388: 1191-1200. Vedolizumab for the Treatment of Chronic Pouchitis
Methods: This was a a phase 4, double-blind, randomized trial (n=102 adults,EARNEST trial) to evaluate vedolizumab in adult patients in whom chronic pouchitis had developed after undergoing IPAA for ulcerative colitis.All patients received 4 weeks of ciprofloxacin and the treatment group received standard vedolizumab dosing. The primary end point was modified Pouchitis Disease Activity Index (mPDAI)–defined remission (an mPDAI score of ≤4 and a reduction from baseline of ≥2 points in the mPDAI total score; scores range from 0 to 12, with higher scores indicating more severe pouchitis) at week 14. The mPDAI is based on clinical symptoms and endoscopic findings.
Key findings:
- The incidence of mPDAI-defined remission at week 14 was 31% (16 of 51 patients) with vedolizumab and 10% (5 of 51 patients) with placebo
- Differences in favor of vedolizumab over placebo were also seen with respect to mPDAI-defined remission at week 34 (difference, 17 percentage points, 35% vs 18%)
My take: Vedolizumab is an effective treatment for chronic pouchitis.



Related blog posts: