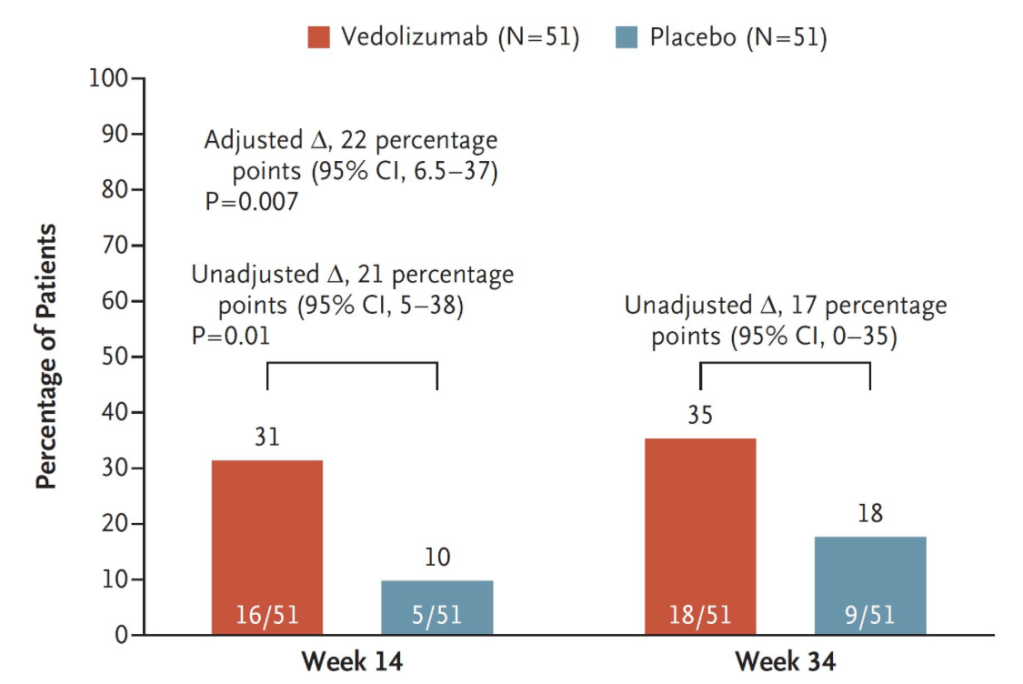
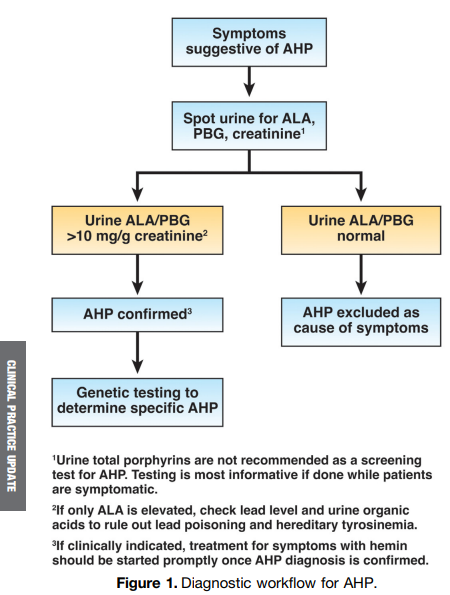
Y Usui et al. NEJM 2023; 388: 1181-1190. Helicobacter pylori, Homologous-Recombination Genes, and Gastric Cancer
Background: Gastric cancer is the fifth most common neoplasm and the fourth leading cause of death from cancer worldwide.1 Helicobacter pylori has been classified as a group I carcinogen and is an environmental risk factor for gastric cancer.2 Although H. pylori infection affects more than half the world population
Methods: This study evaluated the association between germline pathogenic variants in 27 cancer-predisposing genes and the risk of gastric cancer in a sample of 10,426 patients with gastric cancer and 38,153 controls from BioBank Japan. This retrospective study also assessed the combined effect of pathogenic variants and H. pylori infection status on the risk of gastric cancer and calculated the cumulative risk in 1433 patients with gastric cancer and 5997 controls from the Hospital-based Epidemiologic Research Program at Aichi Cancer Center (HERPACC).
Key findings:
- Germline pathogenic variants in nine genes: (APC, ATM, BRCA1, BRCA2, CDH1, MLH1, MSH2, MSH6, and PALB2) were associated with the risk of gastric cancer.
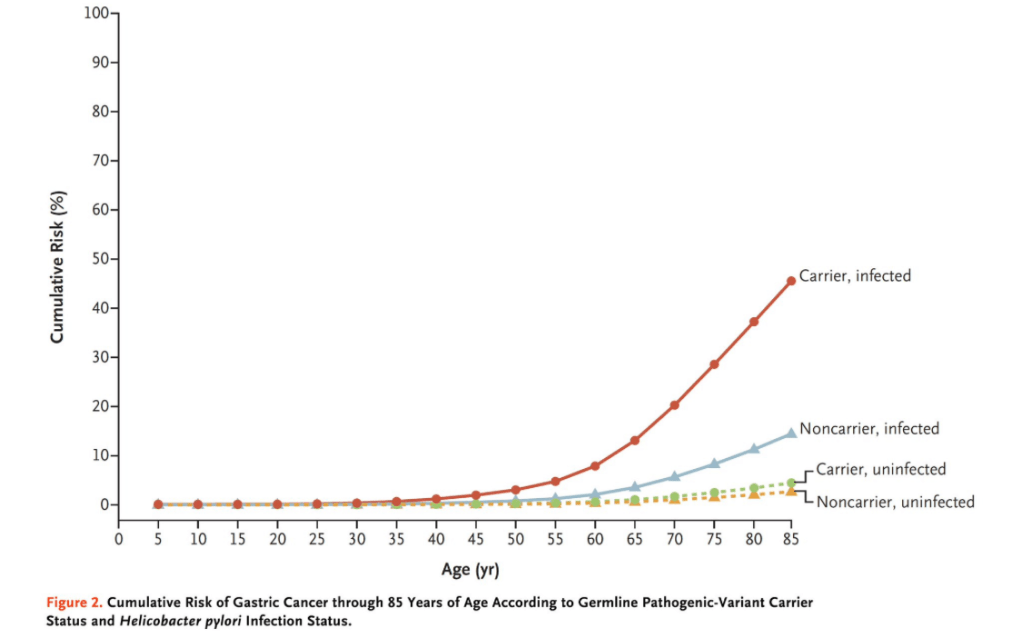
- At 85 years of age, persons with H. pylori infection and a pathogenic variant had a higher cumulative risk of gastric cancer than noncarriers infected with H. pylori (45.5% vs. 14.4%).
- Limitation: The study population was from East Asia and thus, the findings may be different in other populations.
My take: H. pylori infection has a synergistic effect in increasing the risk of gastric cancer in individuals with germline pathogenic variants in homologous-recombination genes. To minimize the risk of gastric cancer, H pylori eradication is important; however, it is especially in those with cancer-predisposing variants.
Related blog posts:
- Treating Helicobacter Pylori Lowers The Risk of Gastric Cancer
- Understanding FDA Approval of Vonoprazan-Based Therapies for Helicobacter Pylori
- Helicobacter Pylori: Relationship to Cancer and Dubious Beneficial Claims
- Helicobacter Pylori 2019 Review
- It is Getting Harder to Treat H pylori -Here’s Why