CJ Lee, DS Dellon. Clin Gastroenterol Hepatol 2024; 22: 252-258. Open Access! Real-World Efficacy of Dupilumab in Severe, Treatment-Refractory, and Fibrostenotic Patients With Eosinophilic Esophagitis
Rationale for the retrospective study: ” Although it is the first Food and Drug Administration–approved treatment for EoE, eligibility criteria for the clinical trial program excluded several characteristics of the most severe EoE patients seen in clinical practice…Therefore, the purpose of this study was to determine the real-world efficacy of dupilumab in patients with severe, treatment-refractory, and fibrostenotic EoE.”
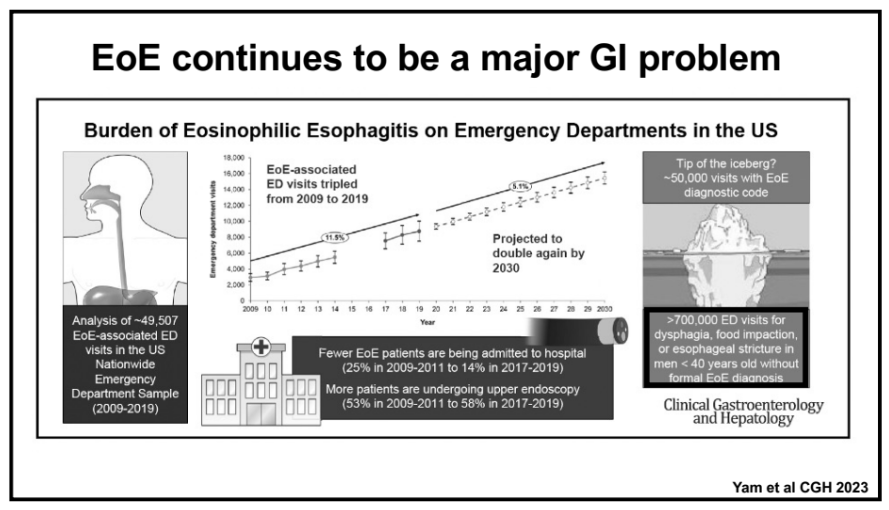
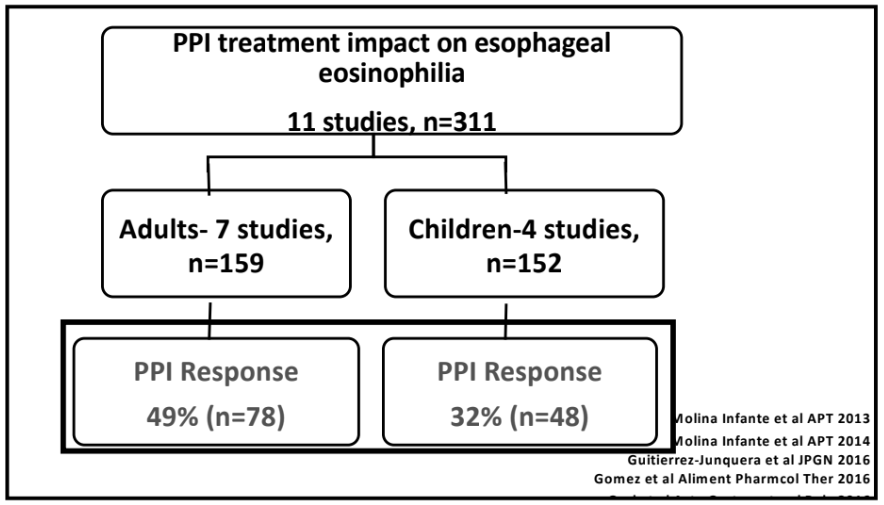
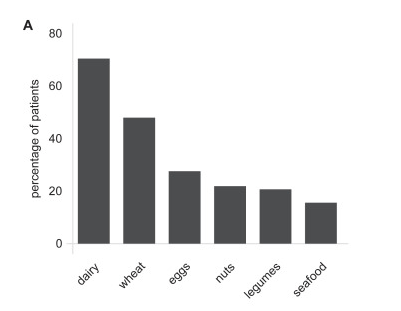
This cohort of 46 patients with severe disease including 39 (85%) who had prior esophageal dilatation (mean of 9). Patients had a mean age of 39 and had had symptoms for a mean of 13 years. Patients were considered treatment-refractory as all had received PPIs and topical steroids; in addition, most (87%) had tried elimination diets.
Key findings:
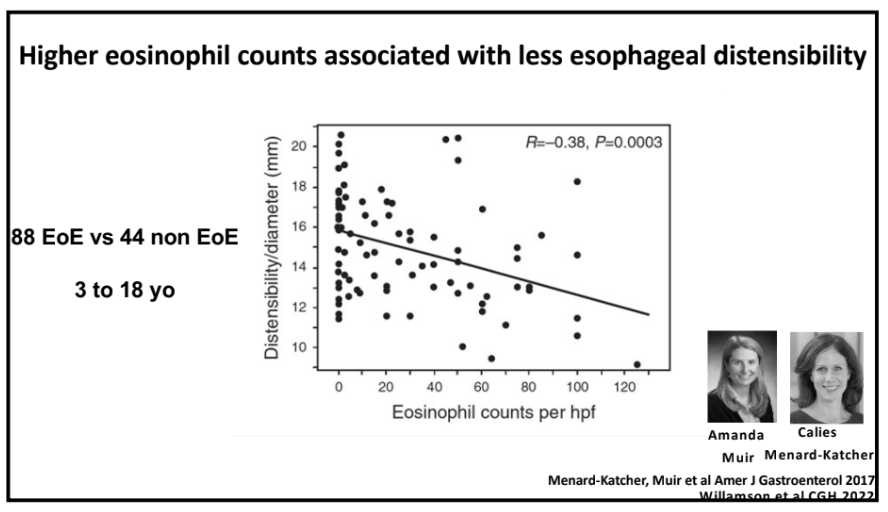
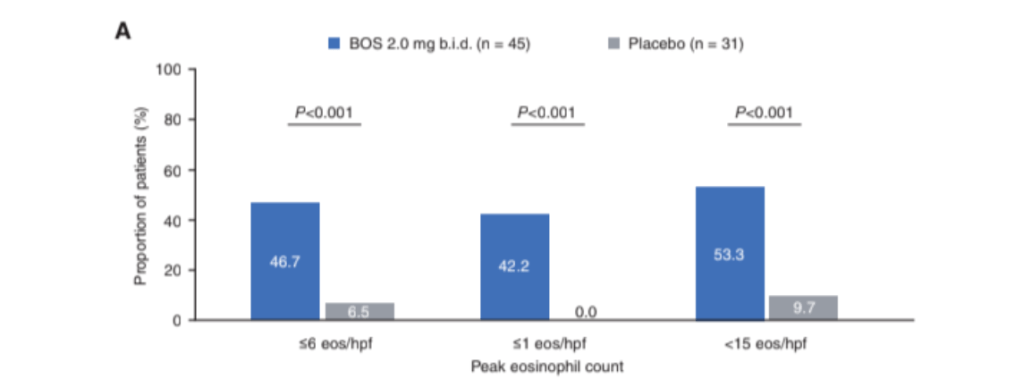
- The peak eosinophil counts decreased markedly, and postdupilumab histologic response rates were 80% and 57% for fewer than 15 eosinophils per high-power field and 6 or fewer eosinophils per high-power field, respectively. Mean eosinophil count dropped from 70 to 9 following dupilumab treatment.
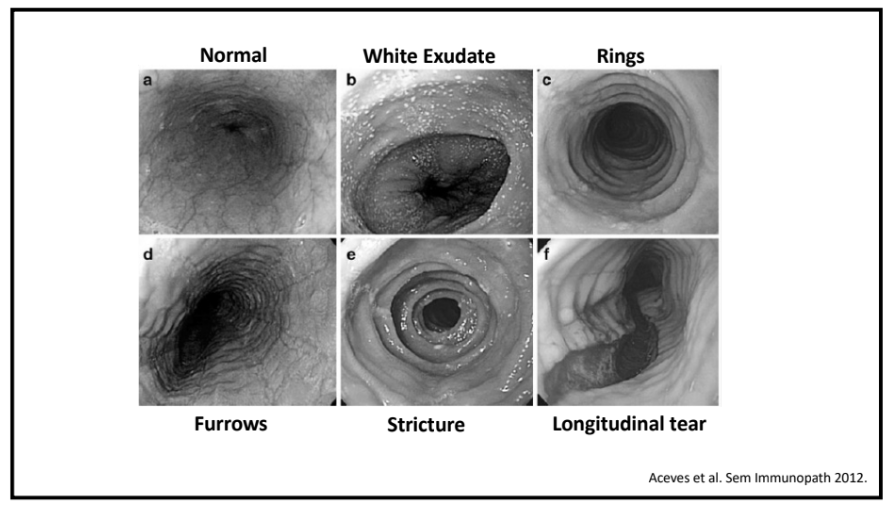
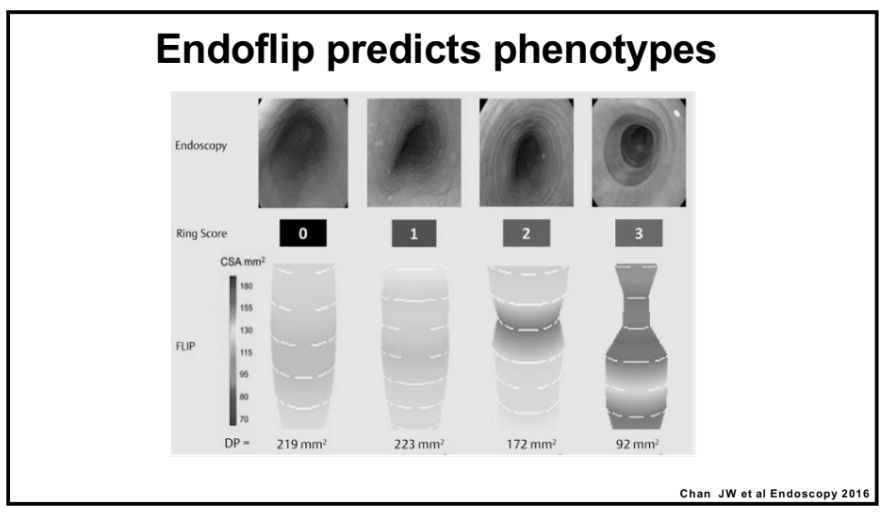
- The Endoscopic Reference Score (EREFS) decreased from 4.62 to 1.89 with improvement in all categories: exudates, rings, edema, furrows and strictures.
- Global symptom improvement was reported in 91% (P < .001).
My take: Many clinical studies are not representative of typical patients with various ailments, often excluding those with the most severe manifestations. This study indicates that dupilumab is an effective agent for patients with severe fibrostenotic eosinophilic esophagitis.
Related blog posts:
- Dupixent Approved in Younger Children (15 kg+)
- Practical Tips for Eosinophilic Esophagitis
- Dupilumab: FDA Approval for Eosinophilic Esophagitis
- “Tug” Sign For Eosinophilic Esophagitis and EoE Bowel Sounds Tips
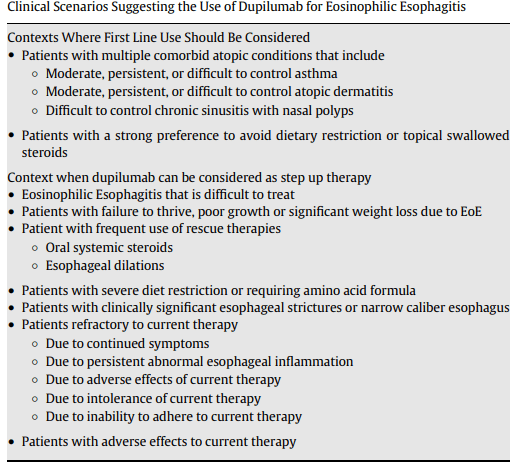
- When to Use Dupilumab for Eosinophilic Esophagitis: Multispecialty Guidelines
- According to the study which you would never qualify for… (2023)
- Landmark Dupilumab Study for Eosinophilic Esophagitis