Briefly noted: J Remmler et al. Clin Gastroenterol Hepatol 2018; 16: 730-7. This retrospective study with 474 patients showed that blood levels of interleukin 6 were associated with mortality. In this cohort, those with levels in the lowest quartile (< 5.3 pg/mL) had zero fatalities within 1 year. In those with the highest quartile (37 pg/mL or more), had a 67.7% mortality rate within 1 year. The associated editorial (pg 630-32) notes that IL6 functions include liver regeneration, infection defense, and metabolic homeostasis. “IL6 is synthesized during inflammatory conditions…persistent activation of the IL6 pathway may have detrimental effects in the livers and in other tissues.”
Author Archives: gutsandgrowth
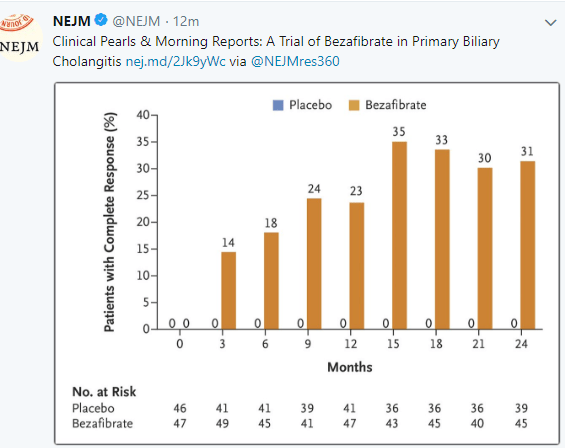
New Treatment for Primary Biliary Cholangitis
Another treatment is emerging for biliary cholangitis (PBC): C Corpechot et al (NEJM 2018; 378: 2171-81) shows that an inexpensive medication, bezafibrate, is effective in patients with PBC who have not responded adequately to ursodeoxycholic acid. An associated editorial (2234-35 by Elizabeth Carey) notes that this medication is not available in the U.S., though a similar medicine, fenofibrate “has shown similar efficacy” in PBC (off-label).
IBD Shorts June 2018
AL Granstrom et al. JPGN 2018; 66: 398-401. Using a nationwide Swedish registry, the authors determined that patients with a Hirschsprung disease had an increased risk of receiving a diagnosis of IBD (OR 4.99). In total 20 of 739 HD patients, developed IBD.
T Card et al. Inflamm Bowel Dis 2018; 24: 953-9. This article questions the ‘what is the risk of progressive multifocal leukoencephalopathy ..with vedolizumab? The authors are not certain. But they state that after reviewing 54,619 patient-years “there have been no cases of PML reported in association with vedolizumab use.”
LCT Buer et al. Inflamm Bowel Dis 2018; 24: 997-1004. This case report of 10 patients describes combination therapy with anti-TNF therapy with vedolizumab. “At the end of follow-up, all patients were in clinical remission, and 8 patients could discontinue anti-TNF treatment.”
OJ Adedokun et al. Gastroenterol 2018; 154: 1660-71. This study examined pharmocokinetics and response of ustekinumab in patients with Crohn’s disease from 701 patients in phase 3 studies.. “Trough concentrations was approximately threefold higher in patients given ustekinumab at 8-week intervals compared with 12-week intervals…Trough concentrations of 0.8 (or even up to 1.4 mcg/mL) or greater were associated with maintenance of clinical remission.” Also, “concentrations of ustekinumab did not seem to be affected by cotreatment with immunomodulators.”
Predicting the Need for Gastrojejunostomy Tube Placement Instead of Gastrostomy Tube Placement
A recent study (ME McSweeney et al. JPGN 2018; 66: 887-92) determined that preoperative characteristics were unable to determine which patients who had gastrostomy tube (GT) placement would ultimately need conversion to gastrojejunostomy (GJ) placement.
This retrospective study matched 79 GJ patients with 79 GT patients.
Key points:
- These patients had similar rates of successful preoperative nasogastric feeding trials (GT 84.5% vs GJ 83.1%), and similar rates of abnormal swallow studies (53.8% and 62.2% respectively).
- In the entire cohort, 11 patients had fundoplication (all GJ patients)
- GT patients were more likely to have tube permanently removed: 20.5% vs 2.5% for GJ patients. Many (45.6%) of the GJ converted patients went back to GT feeds
- Overall, from an initial cohort of 902 patients, 8.8% “required conversion” to GJ feeds
- GJ-converted patients had a trend towards fewer hospitalizations.
While not a result in the study, the issue of GJ compared with fundoplication is briefly discussed. The authors in their discussion of preoperative workup state that
“the complications of fundoplication are more significant and the risks are higher than GJ placement”
In my view, this is one of the most consequential parts of their discussion. While the authors have extensive experience, I think the issue regarding GJ tube placement and fundoplication is more murky. GJ tubes can be difficult to maintain and I have not seen long-term well-controlled studies comparing outcomes between GJ placement and fundoplication.
Other pointers in the discussion:
- Fundoplication has “minimal impact/no impact” to reduce respiratory-related admissions, mainly because the main mechanism is aspiration rather than reflux
- For isolated oropharyngeal dysphagia, one could argue that “an enteral tube is not indicated anymore” based on published data
My take: This is an important retrospective study that illustrates how difficult it is to know preoperatively which patients need GJ placement (or fundoplication) compared to GT placement alone. In our institution, we are reluctant to place GT placement if a patient has not demonstrated tolerance of nasogastric feeds.
Related blog posts:
How Gastrostomy Tube Placement Influences Gastroesophageal Reflux
A recent prospective observational study (M Aumar et al. J Pediatr 2018; 197: 116-20) examined the effect of percutaneous gastrostomy (PEG) tube placement on gastroesophageal reflux disease (GERD) over a 13 year period. This study included 326 patients, 56% who had neurologic impairment and had a median follow-up of 3.5 years (and in some cases follow-up to 15 years). GERD was defined as gastroesophageal reflux causing troublesome symptoms and/or complications. Routine pH studies or impedance were not performed.
Key findings:
- GERD was present in 242 of 326 patients at baseline (74%). GERD appeared in 11% of patients after PEG and was aggravated in 25% with preexisting GERD.
- Factors associated with worsening GERD were neurologic impairment and preexisting GERD.
- 53 patients (16%) required anti-reflux surgery with 22 (6%) in the year following PEG. The only factor identified with the need for surgery was neurologic impairment.
- At last followup, PEG remained in place in 133 children (41%), and had been removed in 99 (30%). 94 children (29%) were deceased, including 2 from an early procedure-related complication. In those who were deceased, the vast majority occurred related to evolution or complication of their underlying disease.
The authors note that studies have shown that PEG increases GERD, but “the majority of these studies were of low methodologic quality.”
My take: Routine antireflux surgery at the time of PEG placement is NOT needed in the majority of patients, even in those with baseline GERD. Less than 20% of patients with GERD required antireflux surgery.
Related blog posts:
- 2018 Pediatric Gastroesophageal Reflux Clinical Guidelines
- Fundoplication in children with neurologic impairment
- How Many Kids with Reflux have Reflux?
- Esophageal Diseases Special
- pH Probe Testing: Rumors of My Death are Premature
- Better to do a coin toss than an ENT exam to determine reflux
- PPI Webinar NAPSPGHAN
- Treating reflux does not help asthma | gutsandgrowth
- Salivary Pepsin Doesn’t Pass Muster for Reflux
Managing Life with IBD
From Nationwide Children’s and ImproveCareNow: Managing Life with IBD This 2 minute video, geared to patients and kids, discusses ways of managing stress, 504 plans, and potential help from therapists.
Another two videos:
How Often Should an Endoscopy Be Normal?
An interesting retrospective study (S Wang et al. JPGN 2018; 66: 876-81) looks at a total of 218 endoscopies in 164 children and examines findings in relation to gastrointestinal symptoms. The results focus mainly on upper endoscopy as there were only 20 who had isolated colonoscopy.
Key findings.
- 164 of 612 (26.8%) of all new patients had an endoscopy
- Among upper endoscopy, 72% were histologically normal, and 56% were macroscopically normal. The authors suspect those with abnormal macroscopic appearance and normal histology were mainly errors in interpretation.
- In those with isolated colonoscopy, 25% were histologically and macroscopically abnormal; thus, complete concordance among the 20 cases.
- In those with combined procedures, the likelihood of abnormalities was higher at 53% for both histology and macroscopically being abnormal. This higher rate was driven mainly by the increased suspicion and diagnosis of inflammatory bowel disease in this subset of 74 patients.
- Overall, 49% of all first diagnostic endoscopies were completely normal, though 65% were histologically normal.
- For upper endoscopy, in those who had performance for isolated abdominal pain, the histologic yield was 11%. It was also 11% for reflux. The symptom with highest yield was vomiting, yet even for this, the findings were normal in about 80%.
- For colonoscopy, rectal bleeding had the highest yield, but 72% were normal in this small cohort, indicating that a trial of conservative management may be appropriate.
An important point in the discussion. The authors take an exception to the idea that normal findings are reassuring. “When procedures are performed despite the expectation of normality this has not been shown to lead to better clinical outcomes of patients…abdominal pain was unaffected by whether or not the patient had undergone endoscopy>”
My take: I agree with the authors that the histologic findings are more likely significant than macroscopic findings in the majority of patients. However, it is worth noting that mild histologic findings are of dubious importance in many cases. Ultimately, identifying strategies to maximize diagnostic yield is needed to provide more cost-effective care and minimize the risks of unnecessary procedures.
Taking Young Children from Their Parents -How Can That Happen Here?
The Impact of Maternal Antibiotics on Necrotizing Enterocolitis and Death in Neonates
A recent study (BD Reed et al. J Pediatr 2018; 197: 97-103) showed that prenatal antibiotic exposure was associated with lower rates of necrotizing enterocolitis (NEC) or death.
In this secondary analysis of a progressive study with 580 infants (<32 weeks) that were cared for in Level III neonatal intensive care units, the authors examined the outcomes of the neonates in relation to whether their mothers had prenatal antibiotics within 72 hours of delivery.
Key findings:
- Two-thirds of mothers received antibiotics w/in 72 hrs of birth, mainly ampicillin (37%) and azithormycin (26.4%). Most (~60%) of the mothers who received antibiotics received more than 1 antibiotic.
- In this cohort, NEC occurred in 7.5%, late onset sepsis (LOS) in 11.1%, and death in 9.6%. The combined outcome of any of these events occurred in 21.3% of study infants.
- Prenatal antibiotics were associated with a reduced risk of NEC (OR 0.28, CI 0.14-0.56), reduced risk of death (OR 0.29, CI 0.14-0.60) but not LOS (OR 1.59, CI 0.84-2.99). Thus, the first two outcomes were within the confidence limits but not LOS.
The authors indicate that their initial hypothesis was that maternal antimicrobials (w/in 72 hours of birth) would increase the risk of NEC but in fact provided a 3-fold protection and reduced the risk of death. The effects presumably would be mediated by changes in the infant microbiome related to infant’s exposure to microbial environment at birth and/or transplacental passage of antibiotics. The authors note that their study did not consider mothers to be in the exposure to antibiotics group if their only exposure was antibiotics at the time of cesarean section due to limited time to effect neonate. They did review this group and noted that if they were included in the exposure group that it would not have significantly changed the findings.
My take: This intriguing finding that NEC and death occurred less often in infant’s whose mothers received antibiotics prior to delivery needs further study as does the long-term effect.
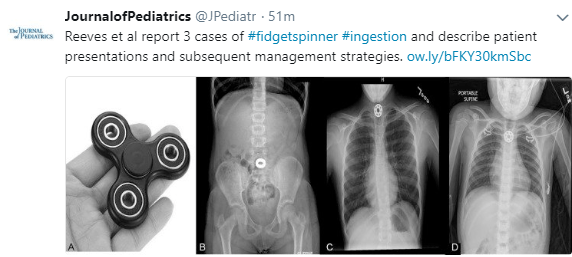
Fidget Spinner Ingestions
A recent study (PT Reeves et al. J Pediatr 2018; 197: 275-9) reviews 3 new cases of fidget spinner ingestion; there had already been 13 cases reported to Consumer Product Safety Risk Management System since 2016.
Key points:
- “If the object is located within the esophagus, urgent endoscopic retrieval is indicated in some cases, specifically if the object is sharp; has electromagnetic capabilities…)eg. batteries, magnets, light-emitting diodes); or fails to pass into the stomach after 24 hours; or the patient cannot tolerate oral secretions.”
- The article recommends using the NASPGHAN foreign bodies guidelines (link below)
Related blog posts: