S Ben-Horin et al. Clin Gastroenterol Hepatol 2024; 22: 347-356. Open Access! Curcumin-QingDai Combination for Patients With Active Ulcerative Colitis: A Randomized, Double-Blinded, Placebo-Controlled Trial
This two part study involved a small open-label trial of combination curcumin-QingDai (CurQD) with 10 patients and a placebo-controlled trial with 42 patients with active ulcerative colitis (UC) over 8 weeks.
Background: “Curcumin and QingDai (QD, Indigo) are herbal compounds previously found to be effective in mild–moderate and moderate–severe ulcerative colitis (UC), respectively, but data on their use still are limited.” Curcumin has purported anti-inflammatory and antioxidant properties through downregulation of nuclear factor-kB, regulation the JAK/STAT pathways, and through its effects on the NLRP3 inflammasome. Qing Dai, also known as indigo naturalis, is a traditional Chinese medicine that has demonstrated efficacy in promoting recovery from colitis in animal models and prior human trials, potentially acting through activation of the aryl hydrocarbon receptor.
Methods: CurQD was administered as 3 capsules of 500 mg herbal extract dry powder QD (a total of 1.5 g) and 3 capsules of 500 mg dry powder curcumin (a total of 1.5 g)
Key findings:
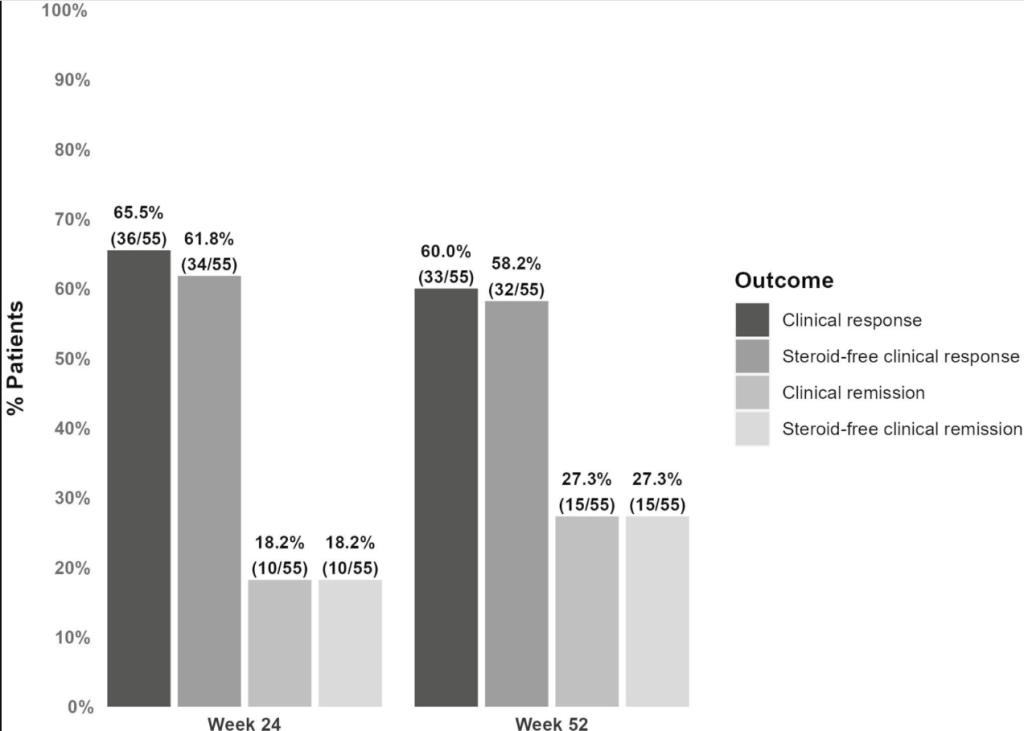
- Clinical response was observed in 85.7% vs 30.7% (P < .001), clinical remission in 14 of 28 (50%) vs 1 of 13 (8%; P = .01), a 50% calprotectin reduction in 46.4% vs 15.4% (P = .08), and endoscopic improvement in 75% vs 20% (P = .036) in the CurQD and placebo groups, respectively.
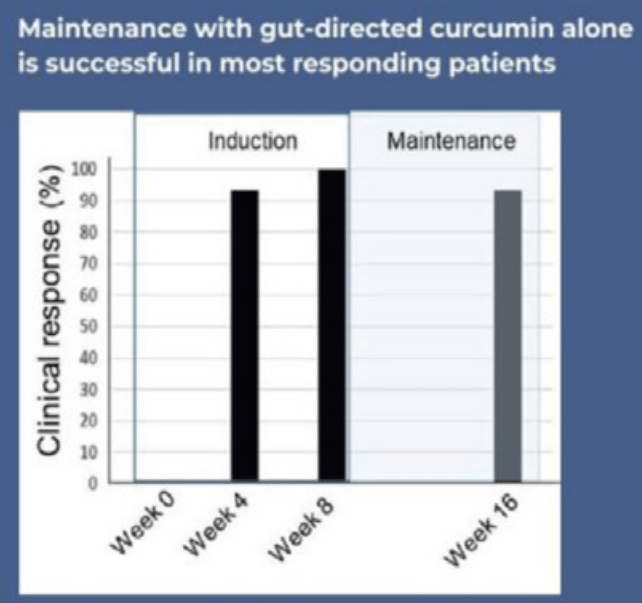
- In the maintenance arm, 11/15 responders in the CurQD arm maintained remission for an additional 8 weeks with just curcumin alone.
The editorial (pg 235 ) notes the following:
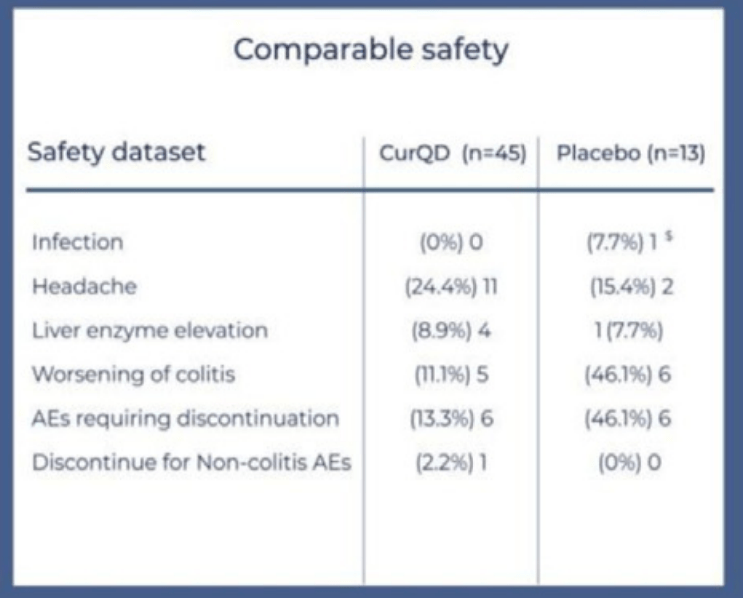
- The number of patients in the study is small and safety and effectiveness of these agents is not certain. Qing Dai has been associated with a rare risk of pulmonary arterial hypertension (especially with long-term use). Thus, further studies are needed.
- “Although it may be preferrable to use these agents in combination with therapies with established efficacy, should the patients’ choice be to use alternative therapy as sole agents for treatment, it is important for us to continue to maintain a trusting physician-patient relationship to ensure that our patients are achieving the treatment targets they need to maximize long-term favorable outcomes, irrespective of the therapeutic agent of choice.”
My take: Curcumin (with combination of Qing Dai for induction) was superior to placebo in achieving meaningful clinical outcomes including clinical response, remission, calprotectin improvement and endoscopic improvement. For future studies, I would favor an active comparator like mesalamine rather than placebo.
Related blog post: Spice It Up? Curcumin for Ulcerative Colitis (2015)