SS C Rao et al. Gastroenterol 2023; 164: 1202-1210. Randomized Placebo-Controlled Phase 3 Trial of Vibrating Capsule for Chronic Constipation
Methods: This was a a phase 3, double-blind, placebo-controlled trial of patients with chronic constipation, who were randomized to receive either a vibrating or placebo capsule, once daily, 5 days a week for 8 weeks. The primary efficacy end points were an increase of 1 or more complete spontaneous bowel movements per week (CSBM1 responder) or 2 or more CSBMs per week (CSBM2) from baseline during at least 6 of the 8 weeks
Key findings:
- A greater percentage of patients receiving the vibrating capsule achieved both primary efficacy end points compared with placebo (39.3% vs 22.1%, P = .001 for CSBM1; 22.7% vs 11.4% P = .008 for CSBM2).
- Spontaneous bowel movements per week, adjusted mean change: 1.40 vs 1.24 per placebo (0.16 per week difference)
The capsule used is 24 mm x 11 mm and includes a motor for vibrations, a battery, a computer chip and a latex-free plastic shell. The control group had received a dissolvable sham capsule.
I looked up cost of this new treatment and it is approximately $89/month which equates to $139 price per poop (PPP). To my knowledge, the PPP is a new metric –I have not seen it previously. For the vibrating capsule, I derived this figure by dividing the monthly cost into a weekly cost and dividing it by 0.16 (mean difference in weekly stooling with vibrating capsule). The PPP may be competitive with some of the constipation medications which cost in the range of ~$500 per month on GoodRx (like prucalopride and linaclotide) but is much more costly than senna products which can be purchased for ~$5/month.
My take: The vibrating capsule is an expensive way to help with constipation

Related article: L Chang, WD Chey, AJ Lembo et al. Gastroenterol 2023; 164: 1086-1106. Open Access! American Gastroenterological Association-American College of Gastroenterology Clinical Practice Guideline: Pharmacological Management of Chronic Idiopathic Constipation
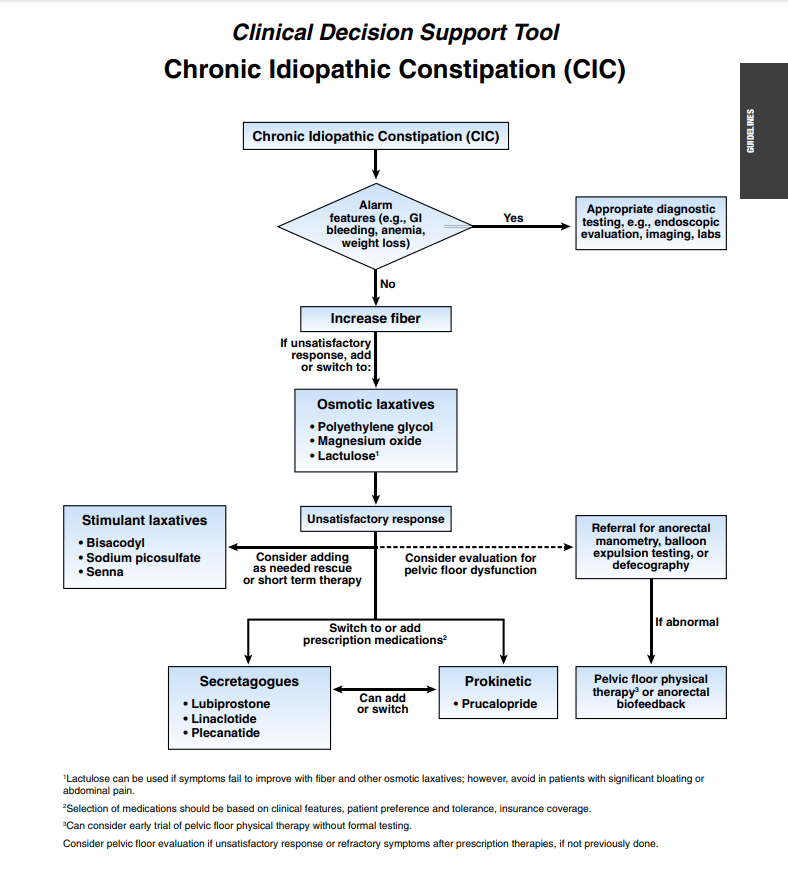
Gastroenterol 2023; 164: 1107. Open Access! Clinical Decision Support Tool
These guidelines for adults with constipation are similar to guidelines published in 2020 with the addition of prucalopride.