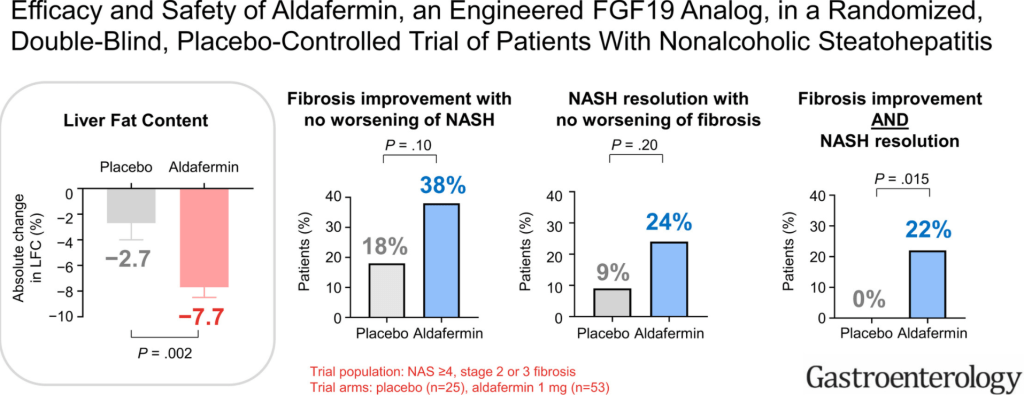
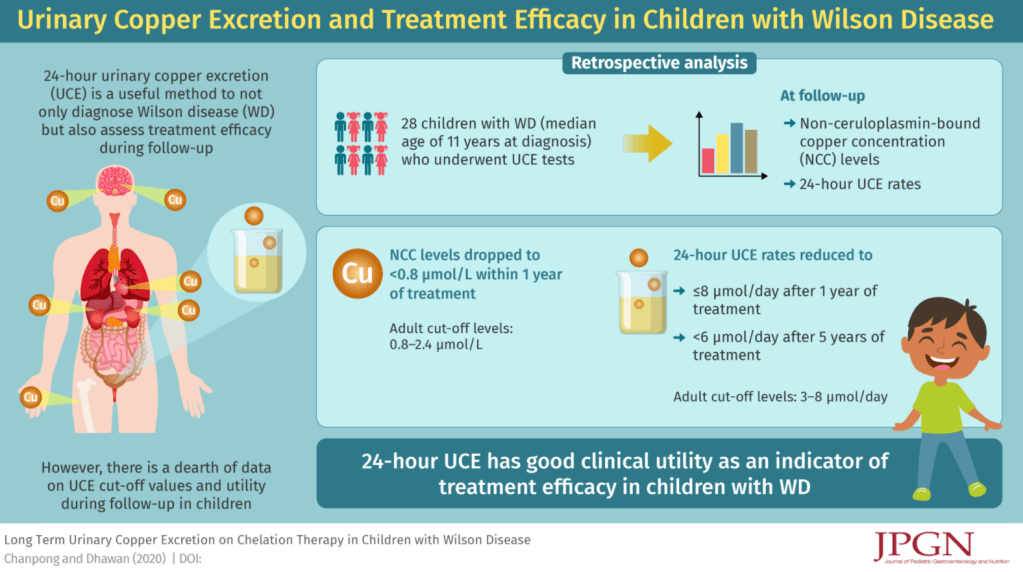
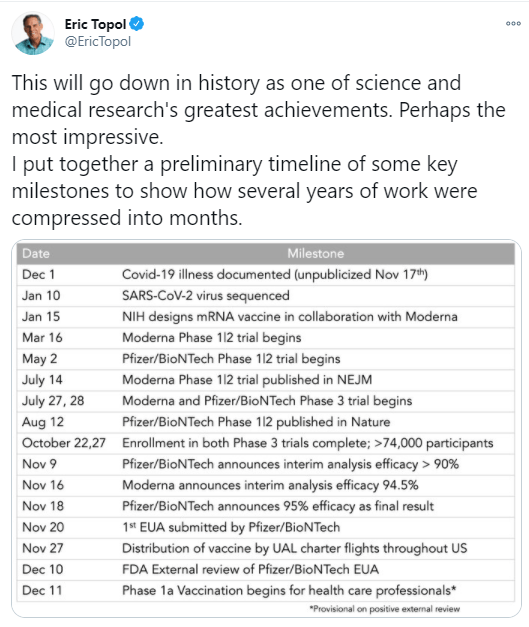
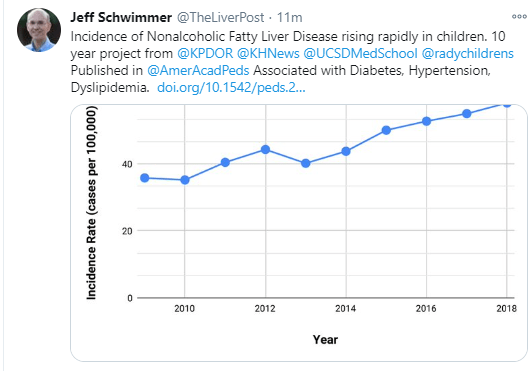
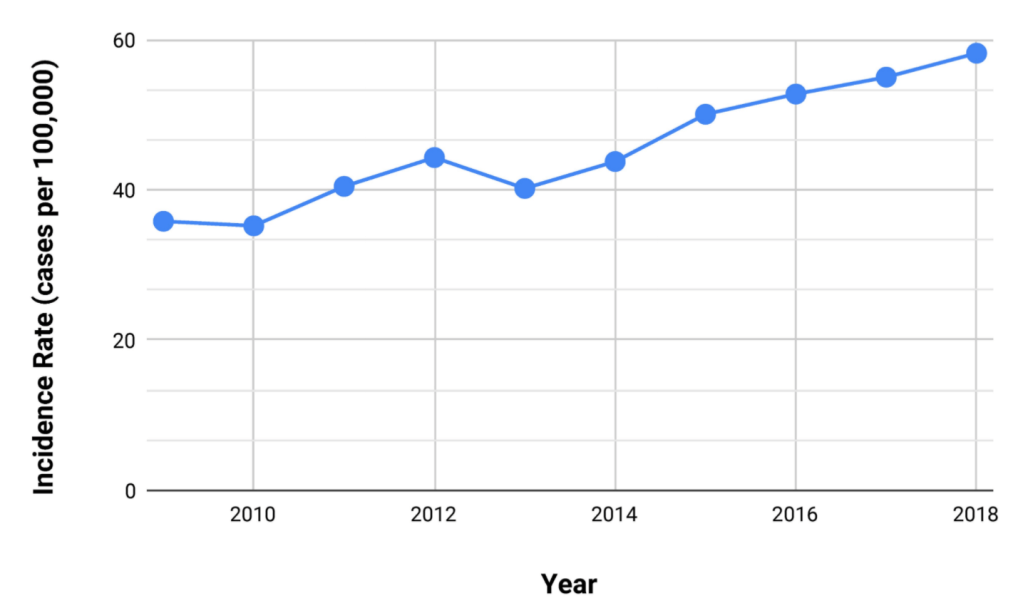
M Biewenga et al. Liver Transplantation 2020; 26: 1573-1581. Full text: Early Predictors of Short-Term Prognosis in Acute and Acute Severe Autoimmune Hepatitis
Key points:
- After the start of immunosuppressive therapy, bilirubin, albumin, and INR normalized in 70%, 77%, and 69%, respectively, in a median of 2.6 months, 3 months, and 4 weeks, respectively, in patients with A-AIH and AS-AIH
- Deterioration of liver function (bilirubin, INR) after 2 weeks of treatment should lead to rapid evaluation for LT and consideration of second-line medication.
I Ziogas et al. J Pediatr 2021; 228: 177-182. Mortality Determinants in Children with Biliary Atresia Awaiting Liver Transplantation
Key points:
- The cumulative incidence of waitlist mortality was 5.2%. Median waitlist time was 83 days.
- In multivariable analysis (n = 2253), increasing bilirubin level ( P < .001), portal vein thrombosis ( P = .03), and ventilator dependence ( P < .001) at listing were associated with a higher risk, whereas weight ≥10 kg at listing ( P = .009) was associated with a lower risk of waitlist mortality.
References Only:
HM DuBrokc, MJ Krowka. Hepatology 2020; 1455-1460. The Myths and Realities of Portopulmonary Hypertension
Related blog posts:
- Portopulmonary Hypertension -A Little More Data | gutsandgrowth
- Pulmonary Complications Associated with Chronic Liver Disease …
- Expert advice on portal hypertension | gutsandgrowth
- Portopulmonary Hypertension | gutsandgrowth
H Oh et al. Clin Gastroenterol Hepatol 2020; 18: 2793-2802. Full text: No Difference in Incidence of Hepatocellular Carcinoma in Patients With Chronic Hepatitis B Virus Infection Treated With Entecavir vs Tenofovir Related blog post: Is Tenofovir the Best Medication for HBV?