P Rosenthal et al. Hepatology 2019; 69: 2326-37. This study examined the efficacy and safety of combined entecavir and Peginterferon for immune-tolerant chronic hepatitis B-infected children (n=60). 48 weeks after completing treatment (week 96), 2 children (3%) achieved the primary outcome of undetectable HBeAg with HBV DNA levels <1000 IU/mL. These two children were also HBsAg negative/anti-HBs positive. In the other children (55 completed study), the ALT and HBV DNA levels were similar to baseline. 37 children experienced adverse events. My take: Entecavir/peginterferon is not very effective in immune-tolerant children infected with chronic HBV.
DL Thomas. NEJM 2019; 380: 2041-50. This article reviews the pathway to the global elimination of chronic hepatitis. Currently, it is estimated that hepatitis C virus (HCV) and hepatitis B virus (HBV) kill more than 1 million persons each year. “In fact, by 2040, deaths from chronic hepatitis are projected to exceed the combined mortality associated with HIV infection, tuberculosis, and malaria.”
JR Dillman et al. J Pediatr 2019; 212: 60-5. This study with 41 patients and 13 patients with biliary atresia prospectively assessed ultrasound shear wave elastography (SWE). The authors found that SWE with a cut-off value of >1.84 m/s had 92% sensitivity and 79% specificity. Also, in their cohort, GGT >320 had a sensitivity of 100% and specificity of 78%.
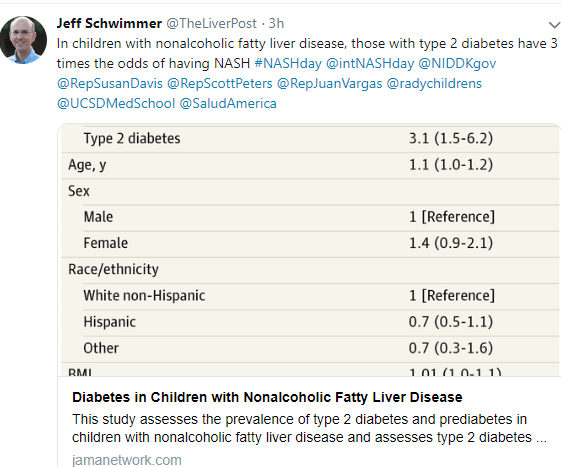
Z Younossi et al. Hepatology 2019; 69: 2672-82. This review provides a global perspective of NAFLD. 25% of the world’s population is currently thought to have NAFLD with highest prevalence in South America at 30.45% and lowest in Africa at 13.5%. This article usggest North America to have 24.1% prevalence rate.