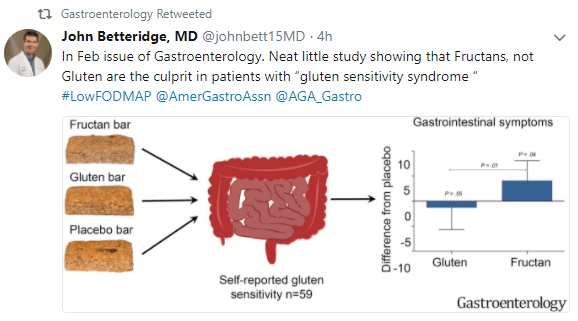
A recent study (R Francavilla et al. Am J Gastroenterol advance online publication, 30 January 2018; doi: 10.1038/ajg.2017.483 ) examined “non-celiac gluten sensitivity” (NCGS) in a multicenter prospective trial from Italy (2013-16).
This study included 1,114 children with chronic gastrointestinal symptoms and negative for both celiac disease and wheat allergy. To determine if these children, had a positive correlation between symptoms and gluten ingestion they were evaluated consecutively through the following phases: run-in, open gluten-free diet (GFD) and DBPC crossover gluten challenge.
Design: If there was a correlation between symptoms and gluten ingestion, then patients were randomized to gluten (10 g/daily) and placebo (rice starch) for 2 weeks each, separated by a washout week. The gluten challenge was considered positive in the presence of a minimum 30% decrease of global visual analogue scale between gluten and placebo.
- Out of 1,114 children, 96.7% did not exhibit any correlation with gluten ingestion.
- Among the 36 patients who seemed to show a correlation between gluten ingestion and symptoms, 28 patients entered the DBPC gluten challenge. Of these 28 children, eleven children (39%) tested positive.
- “No predictive laboratory tests can help in identifying NGCS”
Also, it is worthwhile to quote the authors from their last paragraph: “philosopher Immanuel Kant [said], ‘all our knowledge begins with the senses, proceeds then to understanding, and ends with reason’. NCGS begins in the gut feeling of patients, and we are still in the process of understanding it, hoping that reason is not too far behind.'”
Related blog posts:
- What Happens When Patients with Nonceliac gluten sensitivity are Challenged with Gluten?
- Is a Gluten-Free Diet a Healthy Diet for those without Celiac Disease?
- An Unexpected Twist for “Gluten Sensitivity” | gutsandgrowth
- The Science Behind IBS Dietary Interventions | gutsandgrowth
- Gluten sensitivity without celiac disease | gutsandgrowth
- Food choices, FODMAPs, and gluten haters | gutsandgrowth