S Buhl et al. NEJM 2022; DOI:https://doi.org/10.1056/EVIDoa2200061. Discontinuation of Infliximab Therapy in Patients with Crohn’s Disease
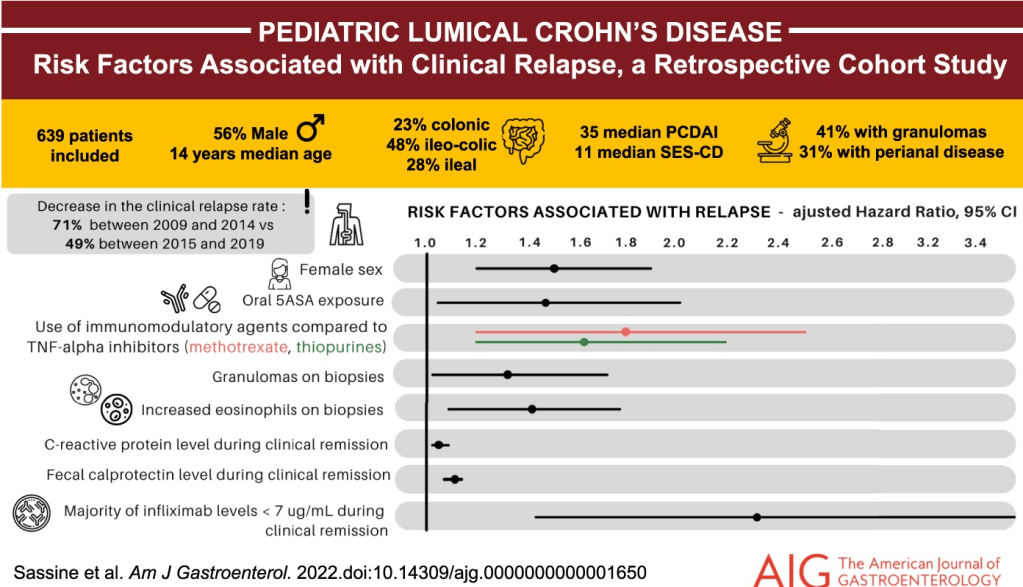
Design: This was a multicenter, randomized, double-blind, placebo-controlled withdrawal study of infliximab in patients (n=115) with Crohn’s disease who were in clinical, biochemical, and endoscopic remission after standard infliximab maintenance therapy for at least 1 year. Patients were randomly assigned 1:1 to continue infliximab therapy or to receive matching placebo for 48 weeks.
Key finding:
- At the end of the trial at week 48, relapse-free survival was 100% in the infliximab-continuation group and 51% in the infliximab-discontinuation group
My take (borrowed from authors): Discontinuation of infliximab for patients with Crohn’s disease receiving long-term infliximab therapy and in clinical, biochemical, and endoscopic remission leads to a considerable risk of relapse
Related blog posts:
- “Do Not Stop Anti-TNF Medications in Children with IBD When They Are Working”
- Biologics in Children with Very Early Onset Inflammatory Bowel Disease
- What happens when anti-TNF therapy is stopped
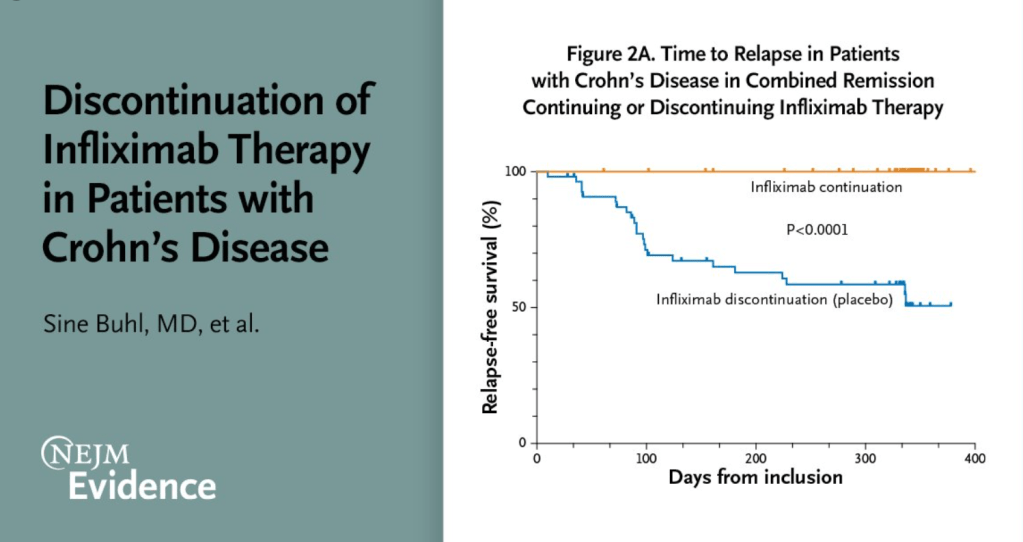
S Sassine et al. AJG 2022; Volume 117 – Issue 4 – p 637-646. doi: 10.14309/ajg.0000000000001650. Risk Factors of Clinical Relapses in Pediatric Luminal Crohn’s Disease: A Retrospective Cohort Study
Key findings–The following variables were associated with clinical relapse:
- female sex (adjusted hazard ratio [aHR] = 1.52, P = 0.0007)
- exposure to oral 5-ASA (aHR = 1.44, P = 0.04),
- use of immunomodulatory agents compared with tumor necrosis factor-alpha inhibitors (methotrexate aHR = 1.73, P = 0.003; thiopurines aHR = 1.63, P = 0.002)
- presence of granulomas (aHR = 1.34, P = 0.02)
- increased eosinophils on intestinal biopsies (aHR = 1.36, P = 0.02)
- high levels of C-reactive protein (aHR = 1.01, P < 0.0001)
- fecal calprotectin (aHR = 1.08, P < 0.0001)
- low serum infliximab levels (<7 mcg/mL) (aHR = 2.32, P = 0.001).