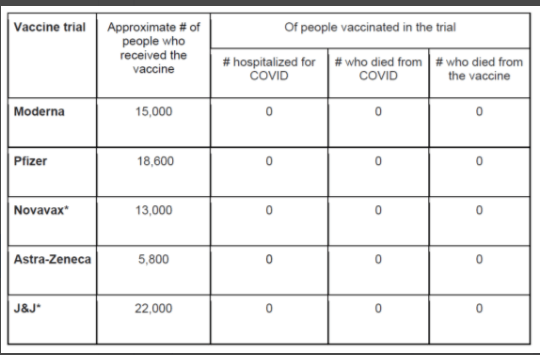
A recent study (GL Fell et al. J Pediatr 2020; 227: 69-76. Optimizing Duration of Empiric Management of Suspected Central Line-Associated Bloodstream Infections in Pediatric Patients with Intestinal Failure) showed that 98% of blood cultures returned positive within 24 hrs.
This prospective single-institution cohort study with 73 patients had 128 Central Line-Associated Bloodstream Infections (CLBSI) in 35 patients during the study period (2015-2018).
Key findings:
- The probability of a blood culture becoming positive after 24 hours was 2.3%; only 1 blood culture became positive after 30 hours (none beyond 48 hrs).
- The median time from blood sampling to positive culture was 11.1 hours.
- Elevated C-reactive protein and neutrophil predominance in white blood cell count were associated with positive blood cultures
My take: 98% is not good enough. For now, 48-hours is the safest policy.
Related blog posts:
- Antibiotic Selection for Suspected Central Line Infections
- #NASPGHAN19 Intestinal Failure Session Part 1

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.