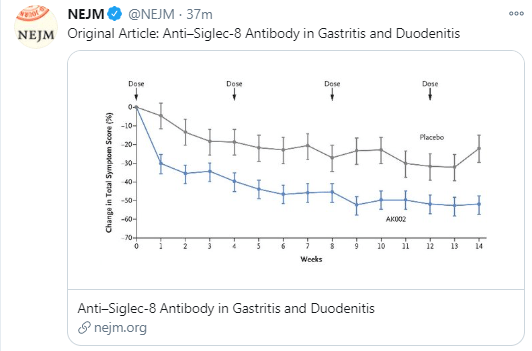
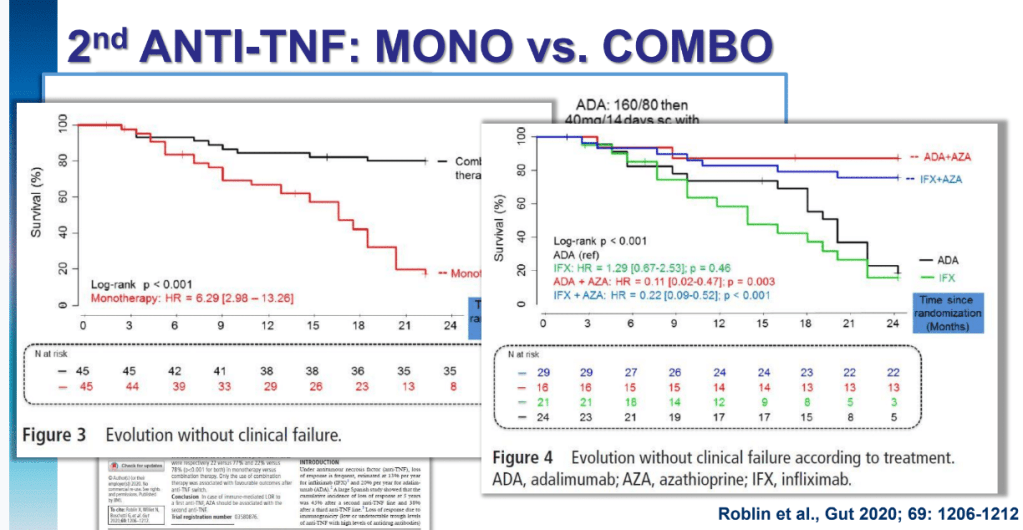
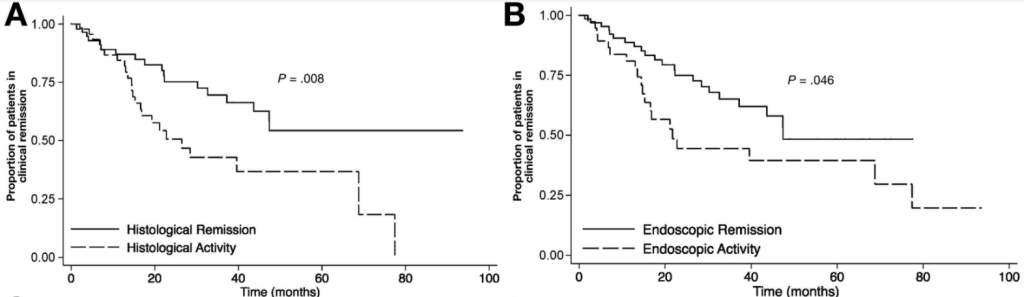
Several recent studies recently evaluated outcomes based on histologic healing compared to endoscopic remission.
RK Pai et al. Clin Gastroenterol Hepatol 2020; 18: 2510-2517. Full text link: Complete Resolution of Mucosal Neutrophils Associates With Improved Long-Term Clinical Outcomes of Patients With Ulcerative Colitis n=281.Key findings:
- “We found histologic evidence of UC activity (Geboes score ≥ 2B.1) in biopsies from 182 patients (65%) and endoscopic evidence of UC activity in 149 patients (53%) (substantial agreement, κ = 0.60).”
- “Histologic features of UC activity were associated with increased rates of systemic corticosteroid use, colectomy, and hospitalization in the entire cohort (P < .05 for all) and associated with increased rates of systemic corticosteroid use in an analysis limited to patients in endoscopic remission (P < .001).”
B Christensen et al. Clin Gastroenterol Hepatol 2020; 18: 2518-2525. Full text link: Histologic Healing Is More Strongly Associated with Clinical Outcomes in Ileal Crohn’s Disease than Endoscopic Healing This was a a retrospective study of 101 patients with CD (52% male) isolated to the terminal ileum. Key findings:
- At ileo-colonoscopy, 63% of patients had endoscopic healing and 55% had histologic evidence of healing. The level of agreement between endoscopic and histologic activity was fair (62%, K = 0.2250, P = .0064)
- On multivariate analysis, only histologic healing was associated with decreased risk of clinical relapse (hazard ratio [HR], 2.05; 95% CI, 1.07–3.94; P = .031), medication escalation (HR, 2.17; 95% CI, 1.2–3.96; P = .011), and corticosteroid use (HR, 2.44; 95% CI, 1.17–5.09; P = .018).
D Kevans et al. Inflamm Bowel Dis 2020; 26: 1722-1729. Histological Markers of Clinical Relapse in Endoscopically Quiescent Ulcerative Colitis Key finding: In endoscopically quiescent UC (n=76), active histological inflammation …[is] adjunctive histological marker associated with increased likelihood of disease relapse. The associated editorial (1730-32 by Asher Kornbluth) quotes Voltaire: “A wise Italian says that the best is the enemy of the good.” He notes that there is “a very real risk of abandoning an effective drug while chasing the goal of some yet to be universally defined histologic remission.” Currently organizational guidelines (ACG, AGA, ECCO, IOIBD) do NOT suggest the use of histologic normalization as an endpoint at this point.
My take: These studies show that histologic healing in ileal Crohn’s disease and in ulcerative colitis are associated with better outcomes that endoscopic appearance. However, there are a lot questions because many patients, possibly a majority, will not achieve histologic healing despite aggressive treatment. Related technical issues include how many biopsies are needed to assess histology and having a validated histologic assessment.
Related blog posts:
- IBD Update Sept 2020 -Deep Remission Article Referenced
- Expert Guidance on IBD (Part 2)
- AGA 2017 Guidelines for Postoperative Crohn’s disease
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.