AHY Ho et al. Clin Gastroenterol Hepatol 2026; 24: 763-771. Real-world Experience of Upadacitinib Reinduction and High-dose Maintenance Therapy in Inflammatory Bowel Disease
Methods: This was a prospective cohort study of patients (n=181 — 79 CD, 83 UC, 6 -IBD-U, 13 with IPAA) treated with UPA between April 2022 and November 2023. Included patients responded to UPA induction, had loss of response (LOR) after dose reduction, and subsequently received reinduction therapy with 45 mg QD. They were followed for a median duration 93 weeks.
Key findings:
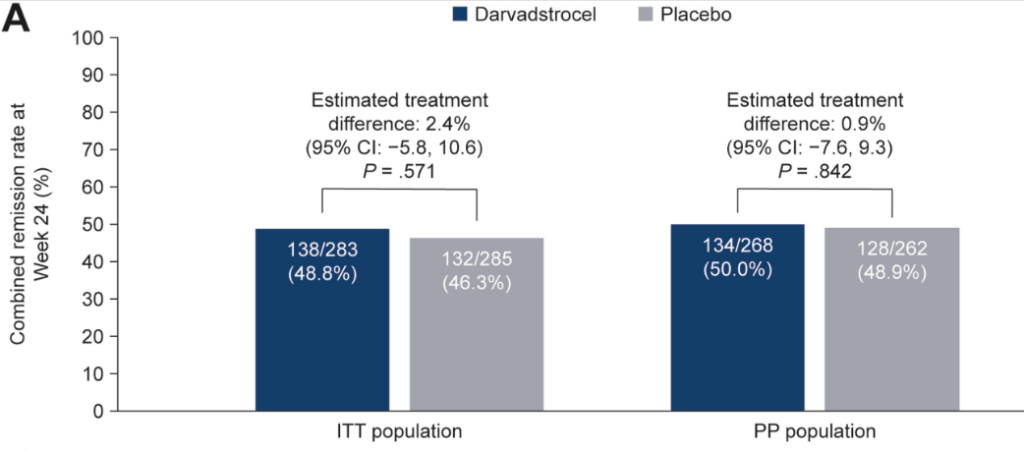
- Dose escalation to 45 mg QD for a median of 13 weeks (IQR, 8–36 weeks) recaptured clinical response in 80.4%
- Among patients who recaptured response, 19 again reduced dose
- 93.8% of patients on 45 mg QD maintained remission vs 21.1% who again dropped to 30 mg QD (P < .001)
- Acne/rosacea was the most common adverse event (39%); there were no serious adverse events
In their discussion, the authors note that dose escalation with another JAK inhibitor, tofacitinib, also has been shown to reverse LOR (in about 50%). In addition, they note that “in our experience, prolonged exposure to 45 mg QDD UPA is safe.” Though, “a longer follow-up period…is required to address long-term safety of UPA in IBD, especially at a higher dose.”
My take: Many patients taking UPA have not responded to multiple other advanced therapy. As such, the potential to recapture response with a higher dose of UPA is an important finding. Dose intensification is an effective strategy for most of the advanced therapies.
Briefly noted: S Honap et al. Clin Gastroenterol Hepatol 2026; 24: 784-793. Open Access! Comparative Effectiveness of Tofacitinib vs Upadacitinib for the Treatment of Acute Severe Ulcerative Colitis In this retrospective study of 111 adults with ASUC, Between days 3 and 7 after treatment initiation, upadacitinib was associated with greater response rates (84% vs 54%), but response/remission was comparable at day 98 (45%/36% vs 55%/48%) and day
182 (29/29% vs 39/34%).
Related blog posts:
- Upadacitinib vs Risankizumab for Crohn’s Disease
- Long-term Efficacy and Safety of Upadacitinib for Ulcerative Colitis
- Upadacitinib’s Effectiveness for Perianal Fistulizing Crohn’s Disease
- IBD Briefs: Upadacitinib in Children, Predicting Crohn’s Disease, and Autoimmune Diseases Associated with IBD
- How Quickly Does Upadacitinib Work for Crohn’s Disease Symptoms?
- Upadacitinib Works Quickly and with High Response