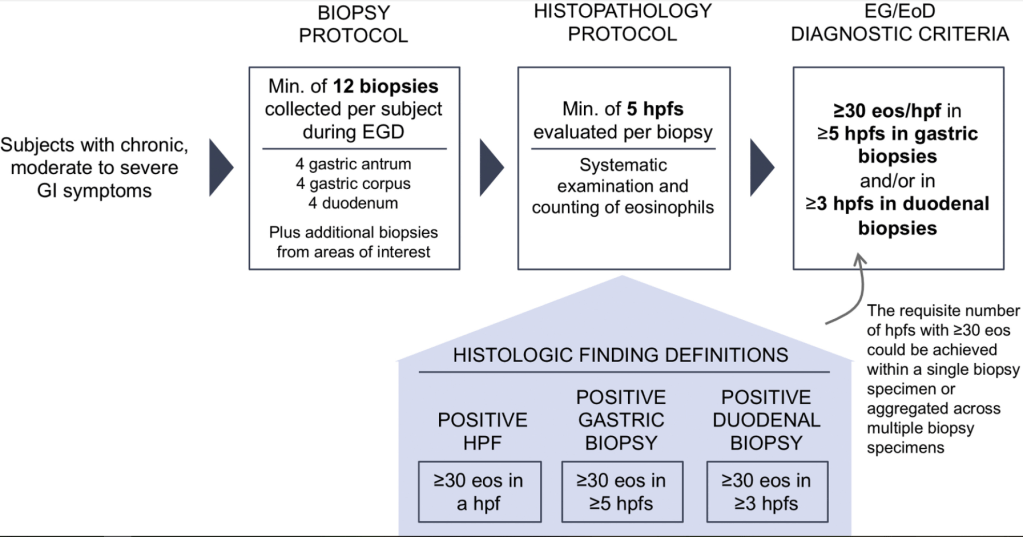
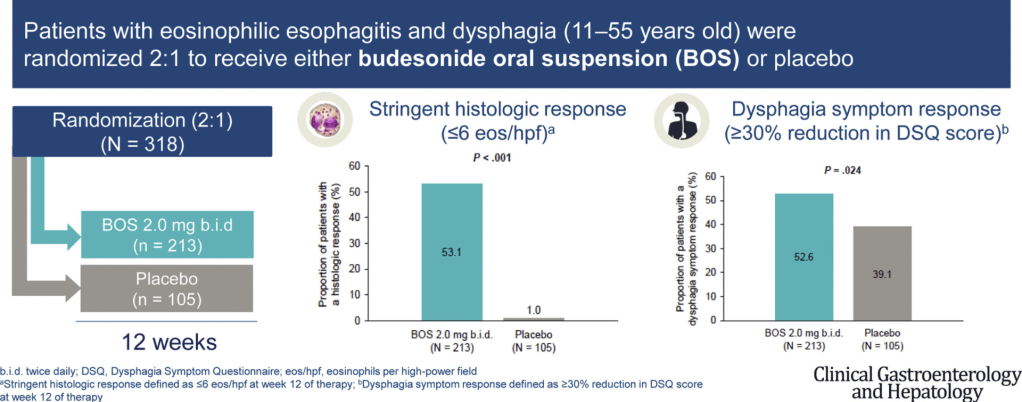
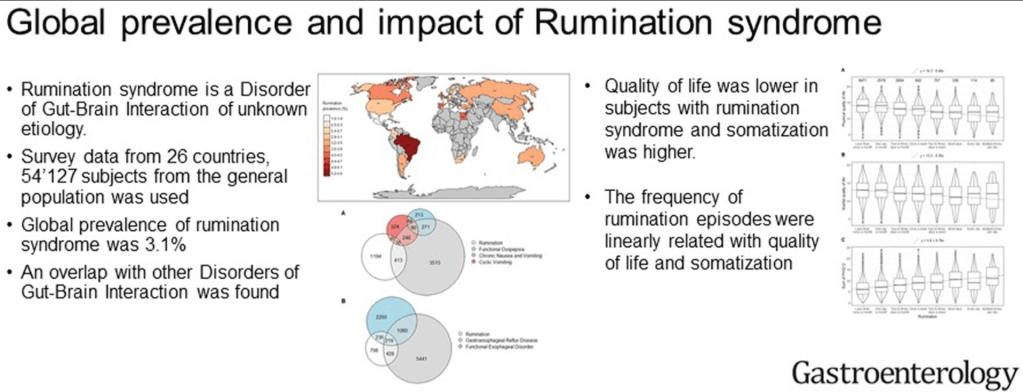
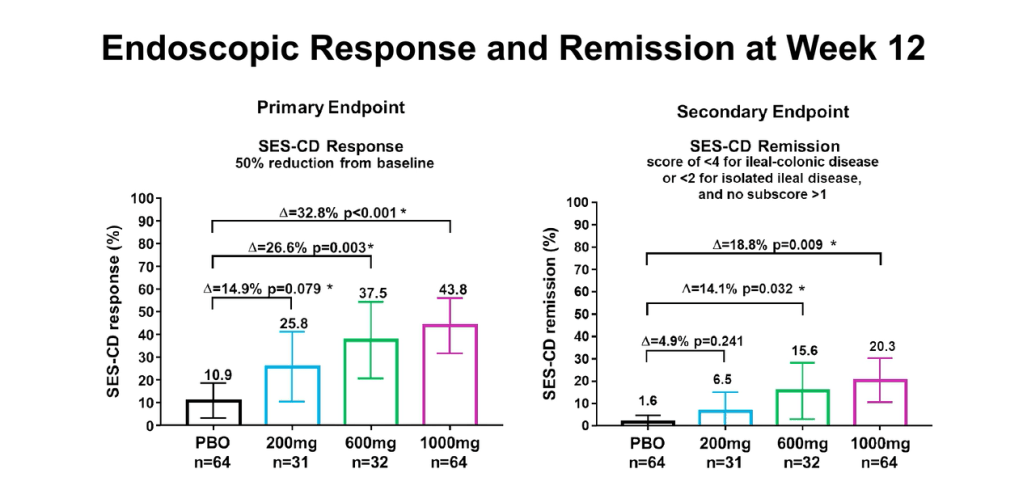
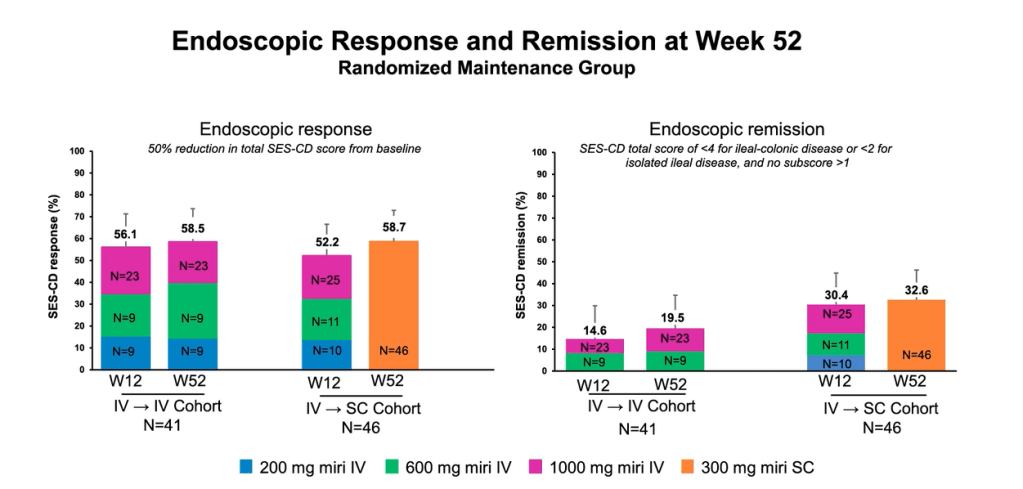
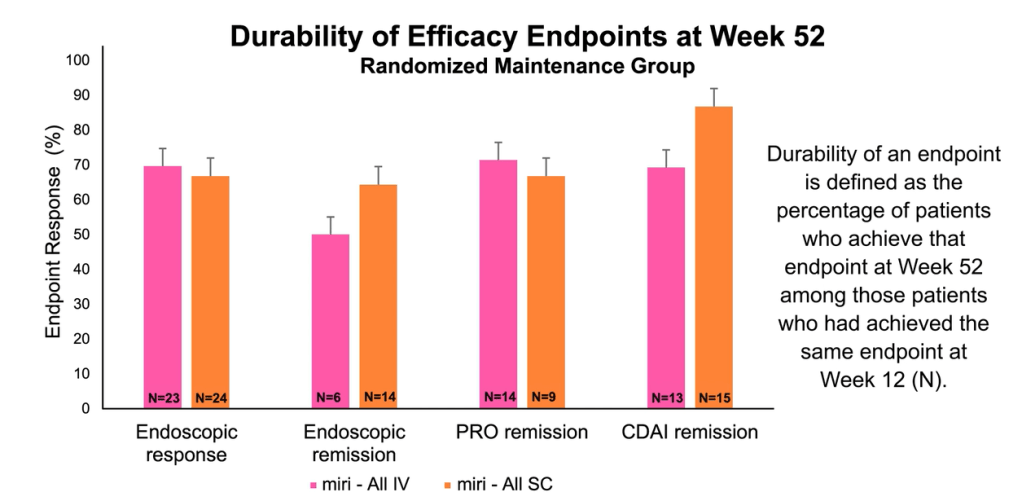
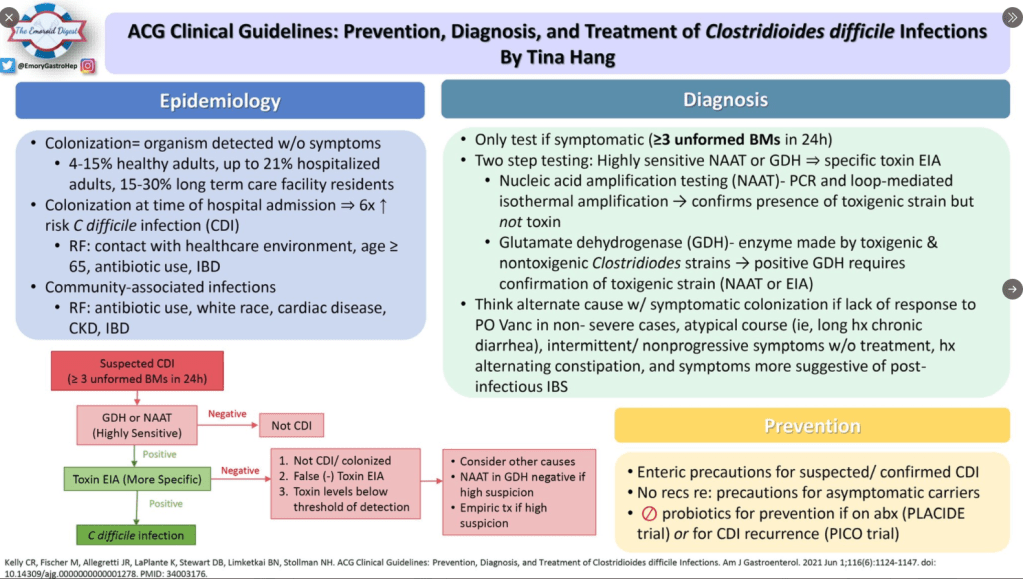
Pharmaceutical Technology (3/17/22): FDA grants approval to AbbVie’s Rinvoq for ulcerative colitis treatment
An excerpt:
The US Food and Drug Administration (FDA) has granted approval to AbbVie’s Rinvoq (upadacitinib) to treat adult patients with moderate-to-severe active ulcerative colitis (UC).
A selective inhibitor of Janus kinase (JAK), Rinvoq is indicated to treat UC patients who had reduced response or are not tolerant to one or more tumour necrosis factor (TNF) blockers.
Per David Rubin: This treatment is a once a day oral pill. In adults, induction consists of 45 mg daily for 8 weeks, then 15 mg or 30 mg for maintenance treatment.
Related blog post: Emerging IBD Treatment: Selective Jak Inhibitor, Upadacitinib

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.