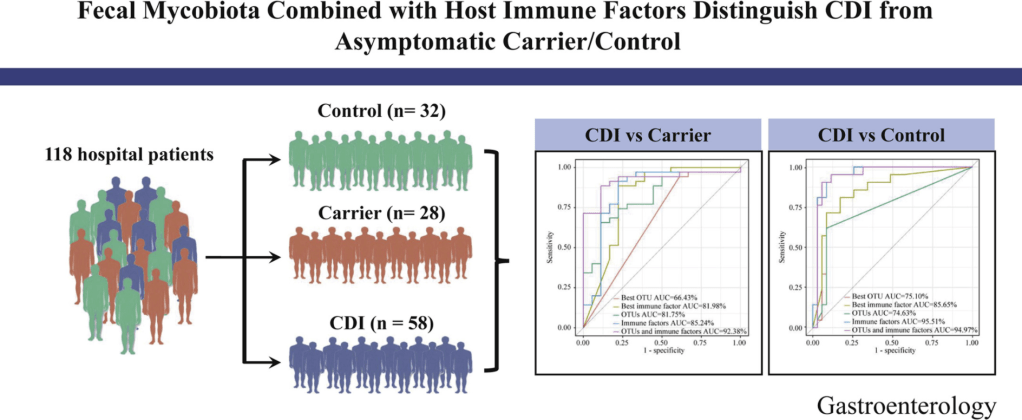
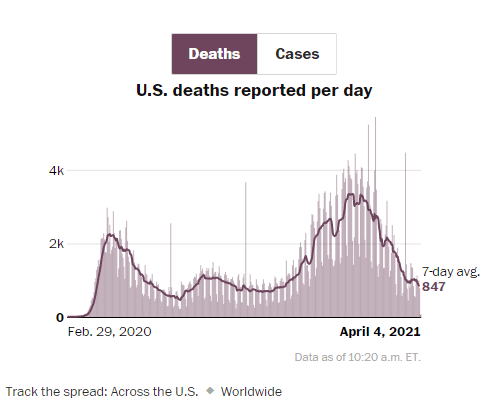
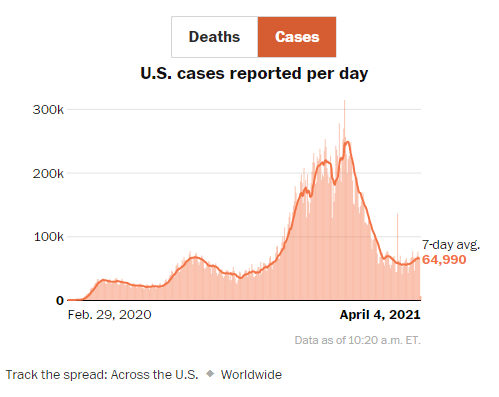
Y Cao et al. Gastroenterol 2021; 160: 2328-2339. Fecal Mycobiota Combined With Host Immune Factors Distinguish Clostridioides difficile Infection From Asymptomatic Carriage
Key findings:
- The ratio of Ascomycota to Basidiomycota was dramatically higher in patients with CDI than in Carrier and Control (P < .05).
- Using 4 fungal operational taxonomic units combined with 6 host immune markers in the random forest classifier can achieve very high performance (area under the curve ∼92.38%) in distinguishing patients with CDI from Carrier.
My take: It is interesting that fecal fungal diversity (mycobiome), in addition to bacterial diversity, is reduced in those with Clostridium difficile infection (CDI) compared to both control groups and those with Clostridium difficile asymptomatic carriage.
Related blog posts:
- Why It Is Still A Bad Idea To Test Asymptomatic Patients For Clostridium Difficile
- ACG Clostridium difficile Guidelines (2021)
- Overdiagnosis of CDI with PCR Assays
- Clostridium difficile: Colonization vs. Symptomatic Infection …
- Clostridium difficile Epidemiology | gutsandgrowth
- Precise Identification of C difficile Transmission …
- Long-term Safety of FMT
- Real-world Efficacy for FMT
- Large Study Show FMT Efficacy/Safety in Children (n=335)
- Clostridium difficile/Fecal Microbiota Transplantation Video