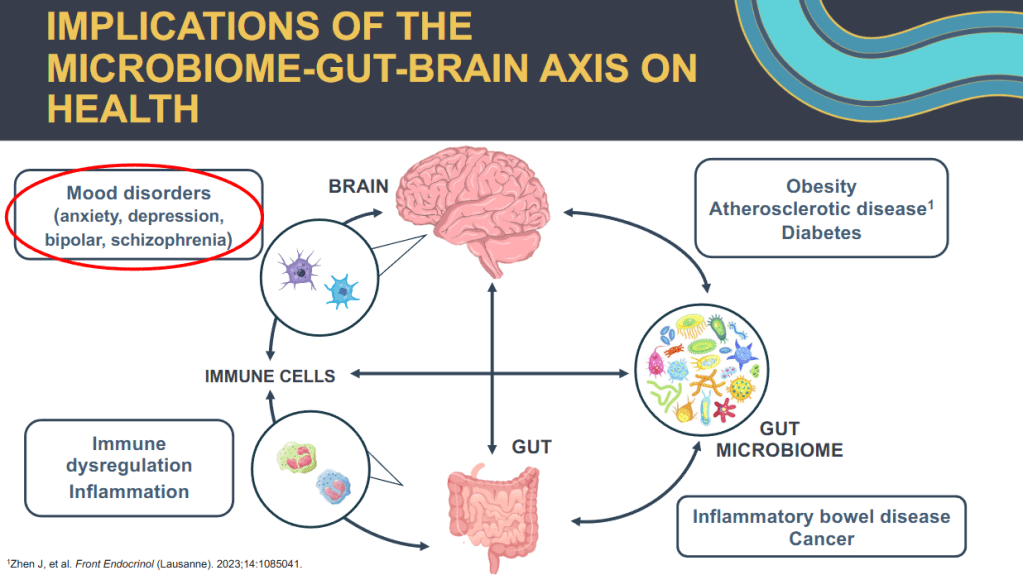
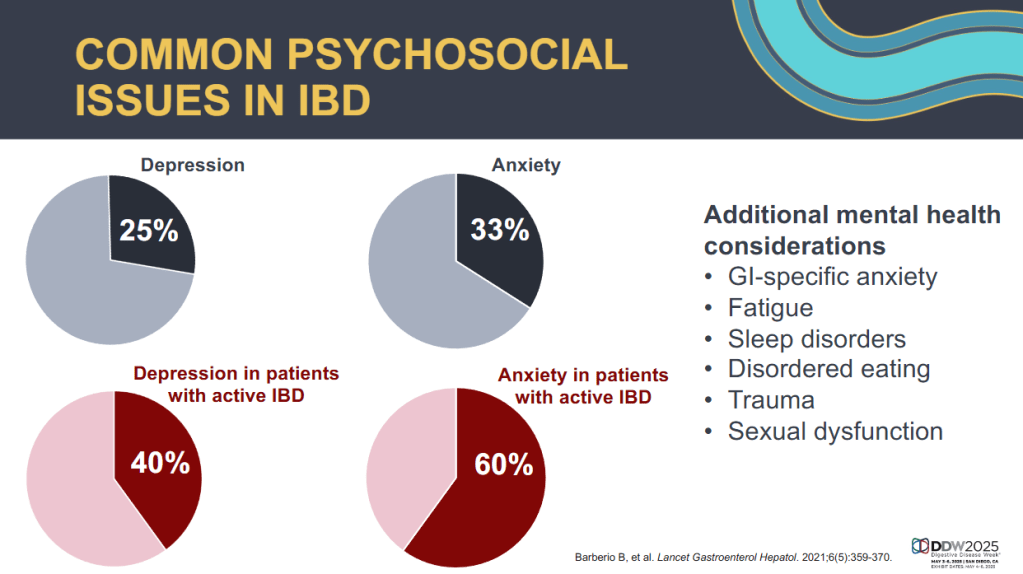
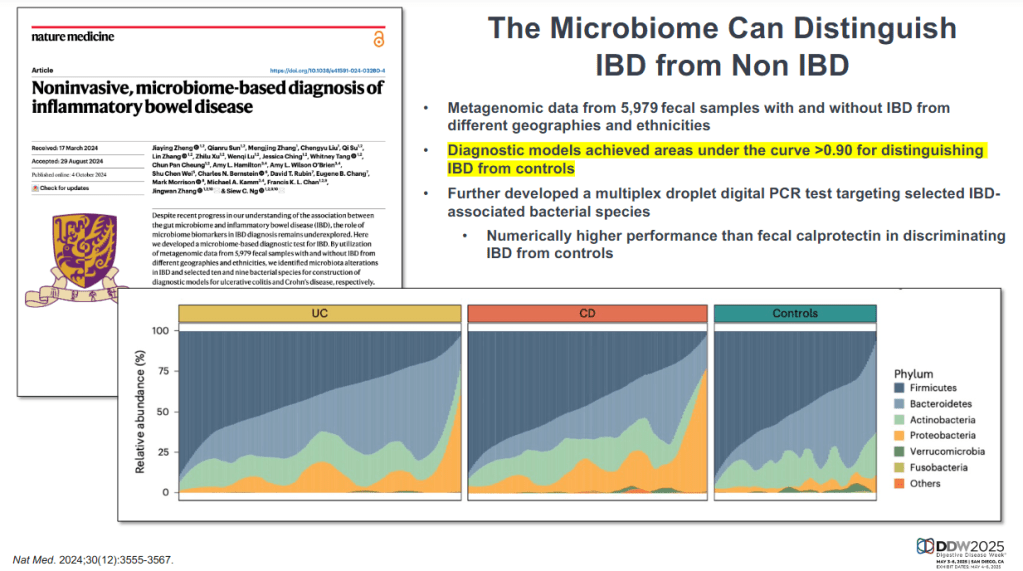
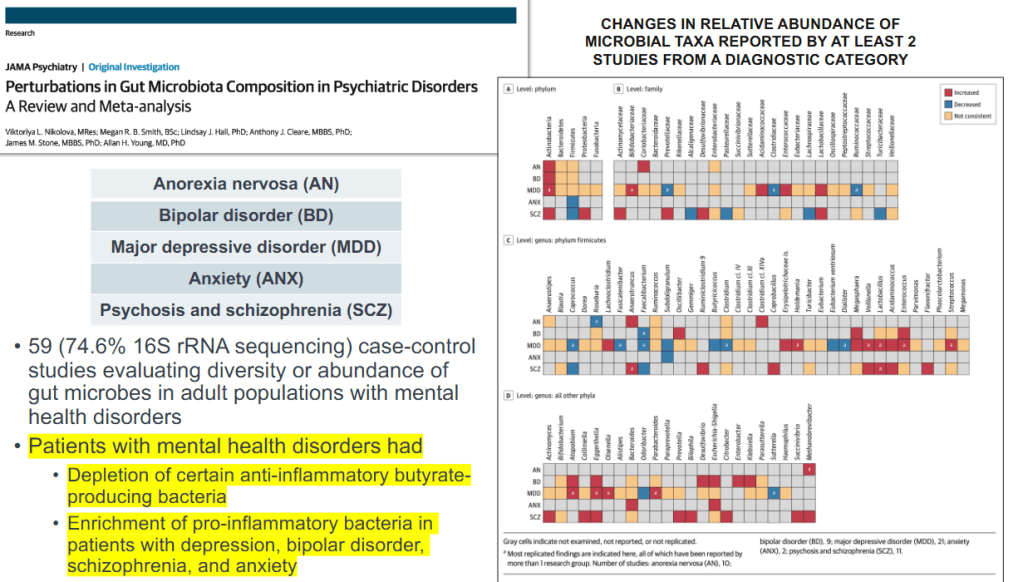
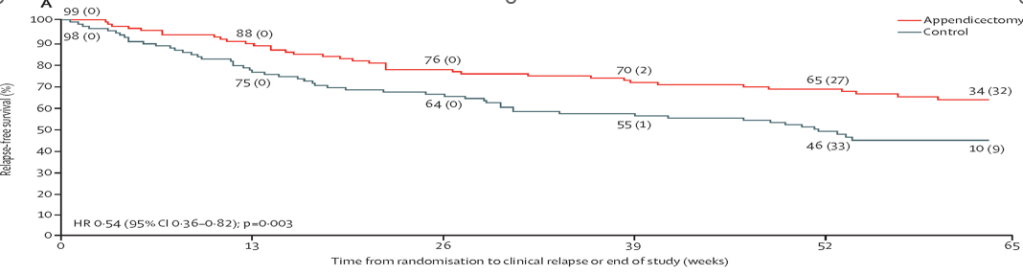
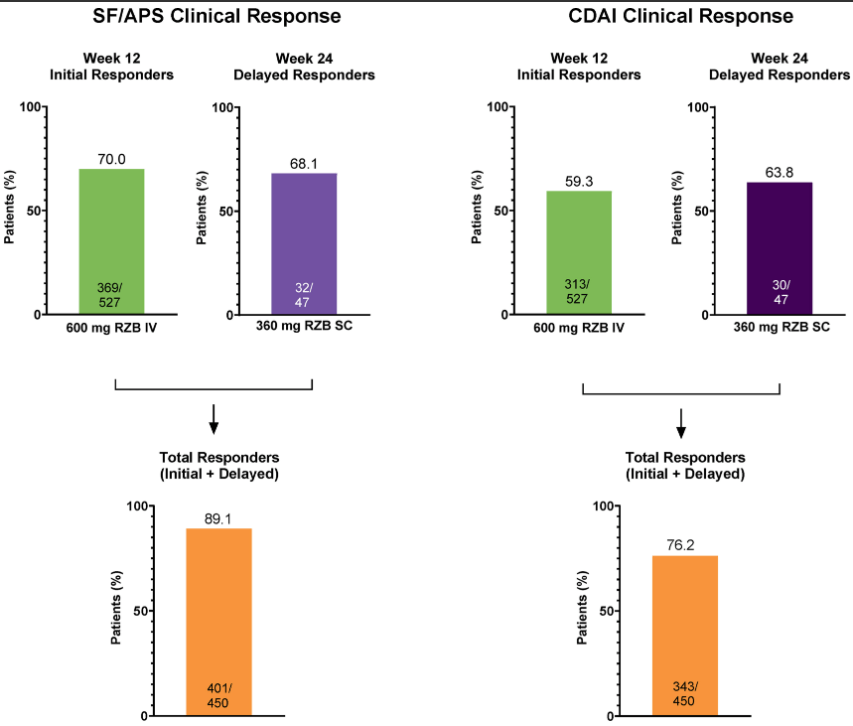
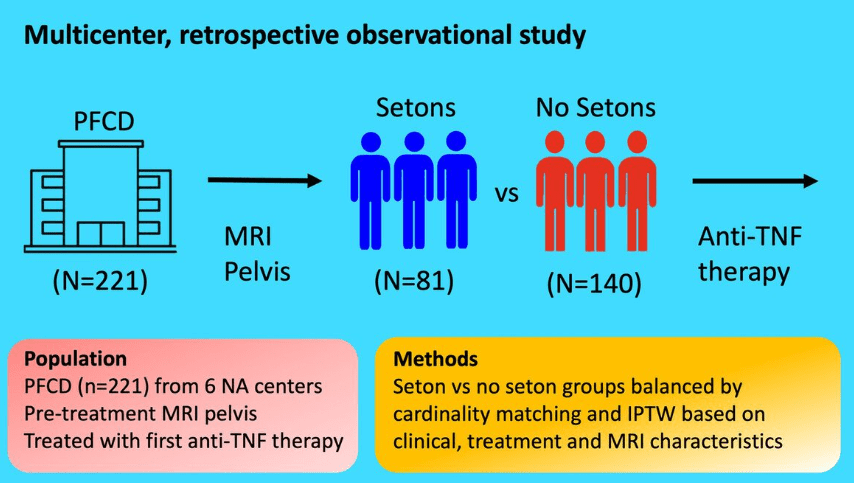
CV Almario et al. Neurogastroenterology and Motility; https://doi.org/10.1111/nmo.70020. Trends in Prevalence of Rome IV Disorders of Gut-Brain Interaction During the COVID-19 Pandemic: Results From a Nationally Representative Sample of Over 160,000 People in the US
Methods: From May 2020 to May 2022, the authors performed a series of cross-sectional online surveys among a representative sample of adults ≥ 18 years old in the US (n=160,154). We administered Rome IV gastroduodenal and bowel DGBI questionnaires.
Key findings:
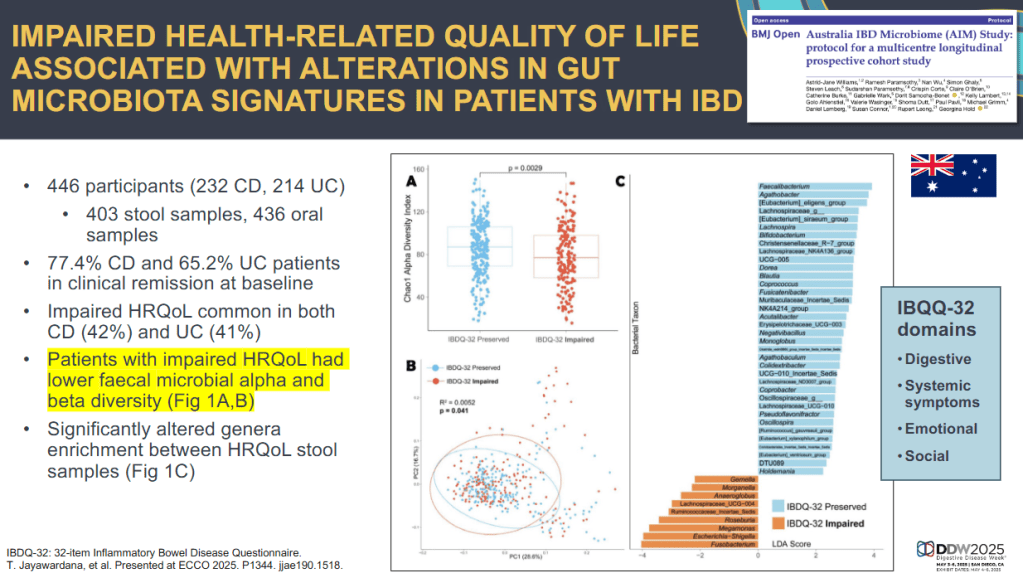
- During the COVID-19 pandemic, the prevalence of irritable bowel syndrome (IBS) increased from 6.1% [May 2020] to 11.0% [May 2022]
- In addition, the prevalence of chronic idiopathic constipation (CIC) increased mildly from 6.0% [May 2020] to 6.4% [May 2022]
- No changes in prevalence were seen for the other examined gastroduodenal and bowel disorders of gut-brain interaction (DGBI)
My take: This study identified increases in the prevalence of IBS during COVID. Increases in IBS following other enteric infections (eg. norovirus, shigella, campylobacter) has been shown previously as well.
Related blog posts:
- Irritable Bowel After Campylobacter Enteritis
- Clinically Useful Biomarkers for Irritable Bowel Syndrome?
- Mechanisms of Postinfectious Irritable Bowel Syndrome & Functional Disorders
- New Study: Leaky Gut and Irritable Bowel Syndrome
- Can We Predict Which Patients With Irritable Bowel Will Respond to Dietary Manipulation Based on Their Microbiome?
- Is There a Way to Prove Which Dietary Factors Trigger Irritable Bowel Syndrome?
- Patient Information on Irritable Bowel Syndrome From Rome Foundation
- Mechanisms of irritable bowel syndrome