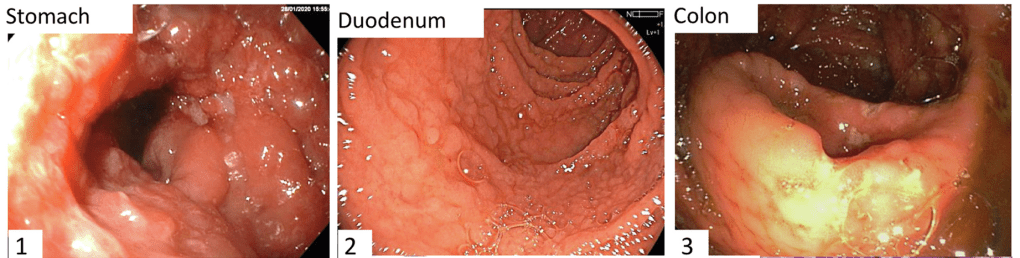
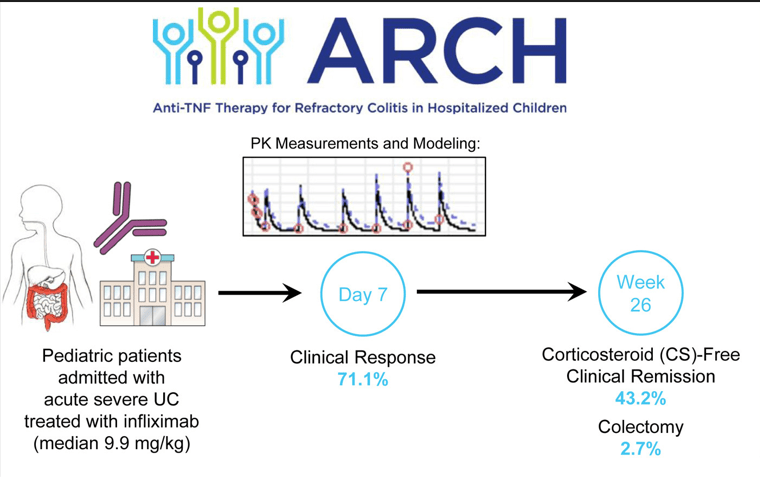
FDA 6/12/23: FDA approves first treatment for pediatric functional constipation
“FDA has approved Linzess (linaclotide) capsules to treat functional constipation in pediatric patients 6 to 17 years of age. Linzess is the first treatment for pediatric functional constipation. The recommended dosage in pediatric patients 6 to 17 years is 72 mcg orally once daily.”
“The efficacy of Linzess for the treatment of functional constipation in pediatric patients 6 to 17 years of age was established in a 12-week double-blind, placebo-controlled, randomized, multicenter clinical trial (Trial 7; NCT04026113) and supported by efficacy data from adequate and well-controlled trials in adults with chronic idiopathic constipation (constipation that persists and isn’t connected to an underlying illness).”
Safety:
- Most common adverse effect was diarrhea
- Avoid in patients with known or suspected mechanical gastrointestinal obstruction (bowel blockage)
- “Linzess contains a boxed warning that the medication should not be taken by patients less than 2 years of age”
- See full prescribing information for additional information on risks associated with Linzess.
My take: While this is good news to have the first FDA-approved treatment for pediatric functional constipation, it is worth remembering that the estimated cost for a monthly supply is between $514-$536 (in Atlanta pharmacies per GoodRx.com).
Related blog posts: