Recently I saw this 1984 skit on SNL for the first time: Science Editor: Spot Bleeding (2 minutes). “Nothing is being done about this:”






Recently I saw this 1984 skit on SNL for the first time: Science Editor: Spot Bleeding (2 minutes). “Nothing is being done about this:”






A Rybak et al. JPGN 2022; 74: 681-692. Open Access: Colonic Function Investigations in Children: Review by the ESPGHAN Motility Working Group
This article reviews the following:
The article provides useful information for indications and for interpreting these studies along with some terrific figures.
For colonic transit radiomarkers:

Related blog posts:

Abbvie Press Release: SKYRIZI® (risankizumab-rzaa) Receives FDA Approval as the First and Only Specific Interleukin-23 (IL-23) to Treat Moderately to Severely Active Crohn’s Disease in Adults
– Third approved indication for SKYRIZI (risankizumab-rzaa) is supported by safety and efficacy data from two induction and one maintenance clinical trials evaluating SKYRIZI in moderately to severely active Crohn’s disease, ADVANCE, MOTIVATE and FORTIFY1-4
– As early as week 4 in the induction studies, clinical response and clinical remission were achieved by significantly more subjects treated with SKYRIZI versus placebo, as were co-primary endpoints of endoscopic response and clinical remission at week 12 and week 521-4
About SKYRIZI® (risankizumab-rzaa)
SKYRIZI is an interleukin-23 (IL-23) inhibitor that selectively blocks IL-23 by binding to its p19 subunit.9,10 IL-23, a cytokine involved in inflammatory processes, is thought to be linked to a number of chronic immune-mediated diseases, including Crohn’s disease.10 The approved dose to treat adults with moderately to severely active Crohn’s disease is 600 mg administered by intravenous infusion over at least one hour at week 0, week 4, and week 8, followed by 360 mg administered by subcutaneous injection at week 12, and every 8 weeks thereafter.4 SKYRIZI is also approved in the U.S. to treat moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy, as well as to treat active psoriatic arthritis in adults, and the recommended dosage is 150 mg administered by subcutaneous injection at week 0, week 4, and every 12 weeks thereafter.4 Phase 3 trials of SKYRIZI in psoriasis, Crohn’s disease, ulcerative colitis and psoriatic arthritis are ongoing
Related blog posts:
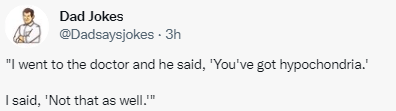
JD Nathan et al. JPGN 2022; 74: 706-719. Open Access: The Role of Surgical Management in Chronic Pancreatitis in Children: A Position Paper From the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition Pancreas Committee
This society paper provides a terrific review of potential operations and rationale in children with chronic pancreatitis. Some of the highlights from this open access article include figures detailing the anatomical considerations of the operations (eg. Frey, Modified Puestow, TPIAT, Berger, Whipple, and Berne) and an algorithm in choosing which procedure should be considered.
Key points:
My take: Fortunately, very few children need operations for chronic pancreatitis. As such, surgical expertise/judgement is particularly important.
Related blog posts:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition
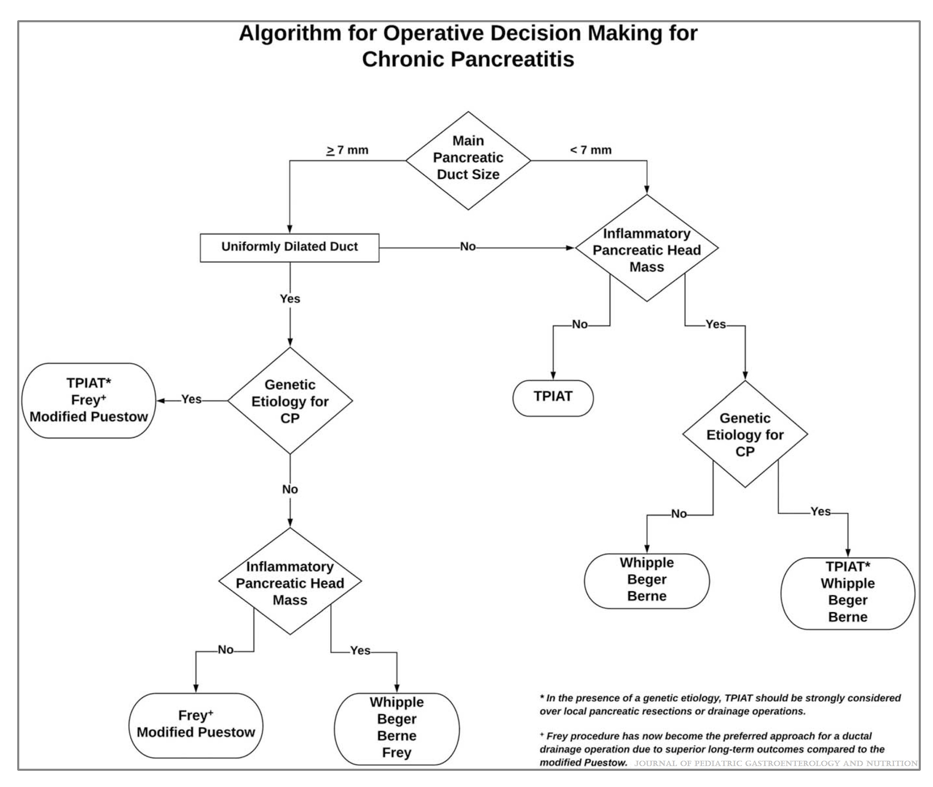
HB Murray et al. JPGN 2022; 74: 588-592. Prevalence and Characteristics of Avoidant/Restrictive Food Intake Disorder in Pediatric Neurogastroenterology Patients
Associated commentary by SB Oliveira, A Kaul: Invited Commentary Re: Prevalence and Characteristics of Avoidant/ Restrictive Food Intake Disorder in Pediatric Neurogastroenterology Patients
This was a retrospective study with 129 consecutive neurogastroenterology patients. Key findings:
The discussion notes that while elimination diets (eg. low FODMAPs diets) are frequently used for disorders of brain-gut interaction, they may increase the risk of ARFID. “Some children may develop fear of what will happen when they try foods again.”
The insightful commentary makes several useful points:
My take: ARFID can be difficult to manage and is important to consider in our patient population, and probably even more so in patients seen in neurogastroenterology programs. The exact prevalence of ARFID in these programs is uncertain and prospective studies are needed.
Related blog post:
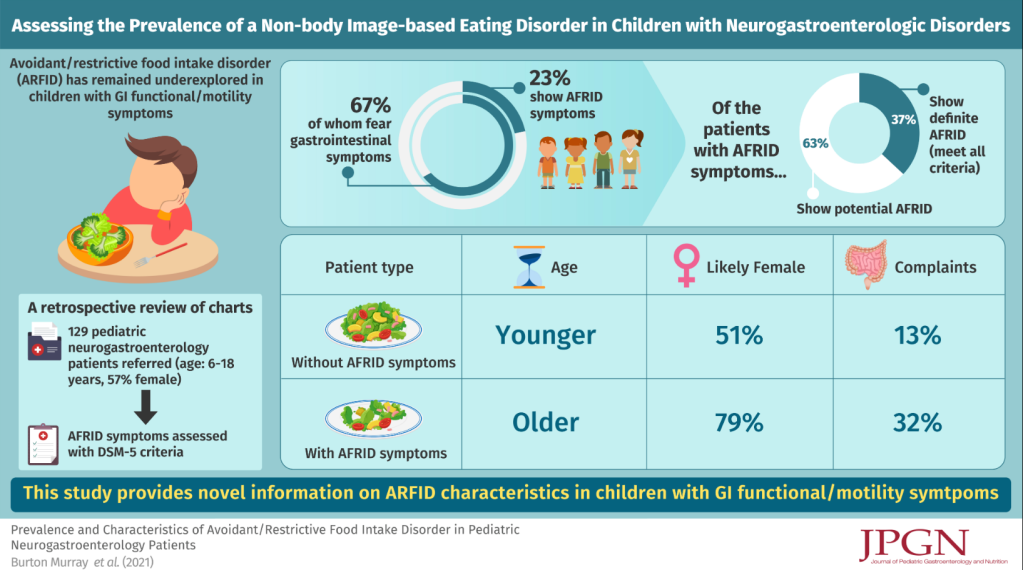
ES Dellon et al Gastroenterol 2022; DOI: https://doi.org/10.1053/j.gastro.2022.03.025 Open Access: A Clinical Severity Index for Eosinophilic Esophagitis: Development, Consensus, and Future Directions “The Index of Severity for Eosinophilic Esophagitis (I-SEE)—that can be completed at routine clinic visits to assess disease severity using a point scale of 0–6 for mild, 7–14 for moderate, and ≥15 for severe EoE.”
From AGA: Eosinophilic Esophagitis Index
“The Index of Severity for EoE (I-SEE) is now available for you to use as a tool to help assess EoE patients. Developed by a multidisciplinary team of experts, the new tool is now published in Gastroenterology.”
Details about I-SEE
Link: I-SEE Tool Scoring Table

X Roblin et al. Inflamm Bowel Dis 2022; 28: 720-727. Swapping Versus Dose Optimization in Patients Losing Response to Adalimumab With Adequate Drug Levels
Many times, treatment decisions are like on “Let’s Make a Deal.” That is, should I stick with what I’ve got or should I try for something better & sometimes wind up with a goat. In this referenced article, patients were under maintenance therapy with adalimumab (ADA) monotherapy (40 mg every 14 days) and had experienced a secondary loss of response (LOR) despite trough levels > 4.9 μg/mL. In this nonrandomized prospective study, patients were either swapped to vedolizumab (VDZ) or optimized on adalimumab (ADA) treatment.
Key findings:
In their discussion, the authors state “current guidelines recommend switching to another class of biologics in case of LOR to ADA with therapeutic drug levels.” However, the authors note that their therapeutic level cut-off of >4.9 mcg/mL is lower than the latest recommendations. In addition, in their conclusion, they note that due to limited biologic options, “ADA optimization strategy might be considered” in a subgroup.
My take: Despite better results in the patients that swapped to VDZ in this study, I think it is important to assure adequate drug levels before choosing a new drug class. For ADA, expert recommendations have suggested a level of 8-12 as therapeutic and to avoid discontinuation if ADA level is less than 10. In this study, more than 40% remained on ADA two years after LOR in those with dosing optimization.
Related blog post:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.

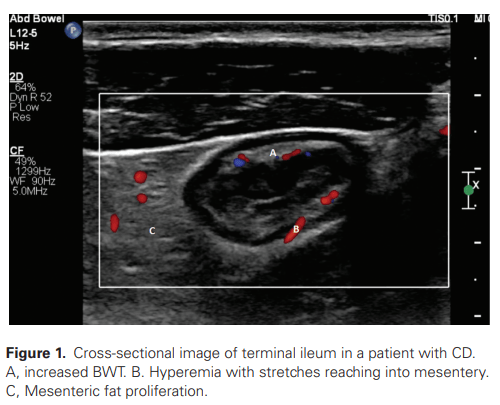
EA van Wassenaer et al. Inflamm Bowel Dis 2022; 28: 783-787. Open Access PDF: Intestinal Ultrasound in Pediatric Inflammatory Bowel Disease: Promising, but Work in Progress
Key points from this review:
My take: Due to low upfront costs, IUS would be appealing adjunct to current monitoring. However, one could envision IUS leading to more downstream studies (& costs), especially if its sensitivity and specificity are not very high.
EJ Brenner et al. Inflamm Bowel Dis 2022; 28: 728-733. Anxiety and Depressive Symptoms Are Not Associated With Future Pediatric Crohn’s Disease Activity
In this internet-based cohort of 9-17 yr olds (n=159, 96% white), the authors found no association between baseline PROMIS Pediatric anxiety score and subsequent sCDAI (change in sCDAI for 3-point change in PROMIS Pediatric −0.89; 95% CI −4.81 to 3.03). This study is in contrast to studies in adults which have shown a bidirectional relationship between anxiety/depression and IBD activity.
My take: It is difficult to know with certainty whether anxiety/depression may trigger IBD activity; more studies are needed. Treatment of mental health is important regardless of its effects on IBD activity.
Related blog posts:
NY Times Magazine, Helen Ouyang 5/22/22: Can Virtual Reality Help Ease Chronic Pain?
This lengthy article describes the emerging therapy of virtual reality to help with chronic pain. Some of the article focuses on chronic abdominal pain.
Here are some excerpts:
My take: I am looking forward to the pediatric studies which will be needed before this technology can be promoted. I would think pediatric patients with chronic pain may respond even more favorably than adults. If this technology were in our clinic, I am certain that are “no show” rate would be lower.
Related blog posts:

M Alsayid, V Kotwal. Clin Gastroenterol Hepatol 2022; DOI:https://doi.org/10.1016/j.cgh.2021.06.030. Open Access: Chronic Constipation With Fecal Stasis
A 50 year old with a history of abdominal distention for a week and chronic constipation had a CT with intravenous contrast. Ultimately he had a disimpaction under general anesthesia and a flexible sigmoidoscopy.
My view: A complete examination with digital rectal exam (if feasible) or an AXR would have been sufficient for diagnosis.
Blog note: Link for We Can’t Wait App updated in yesterday’s post.

