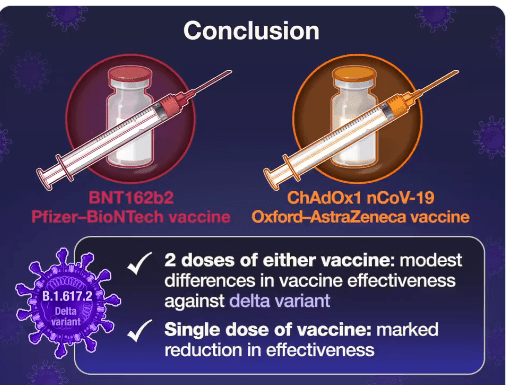
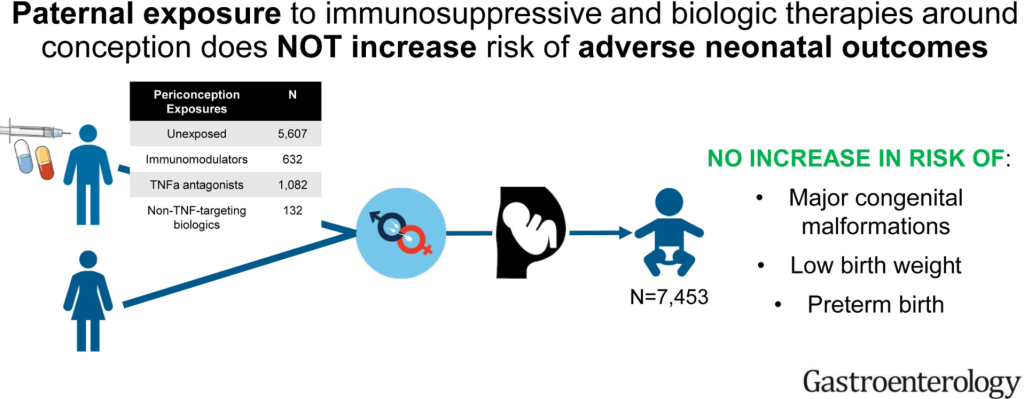
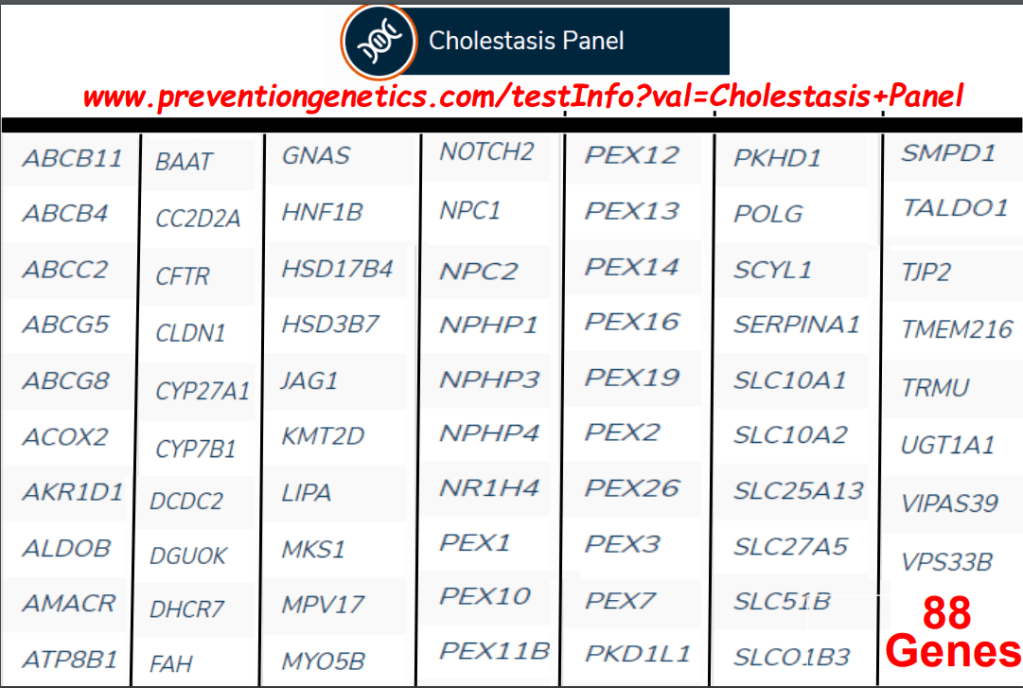
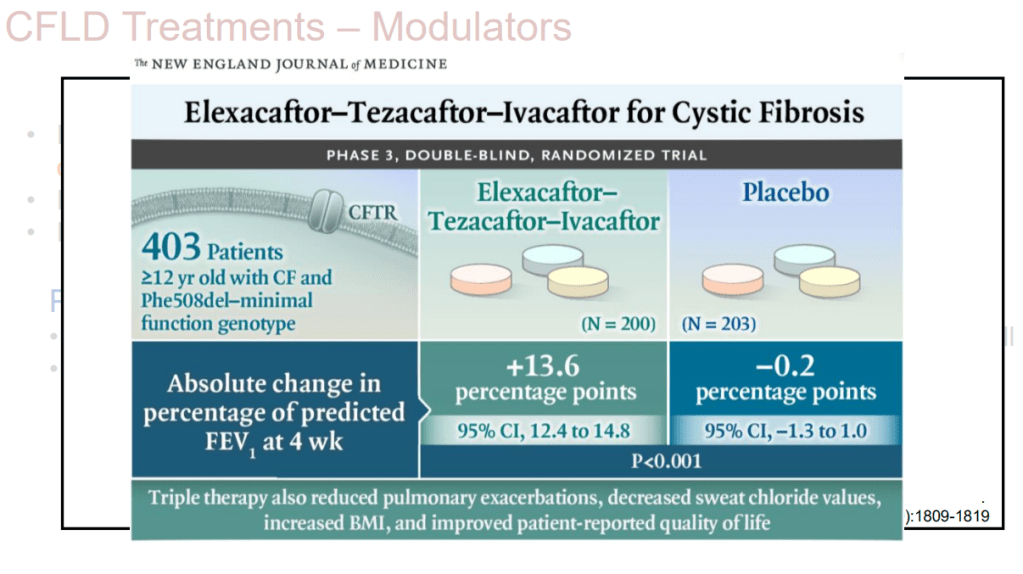
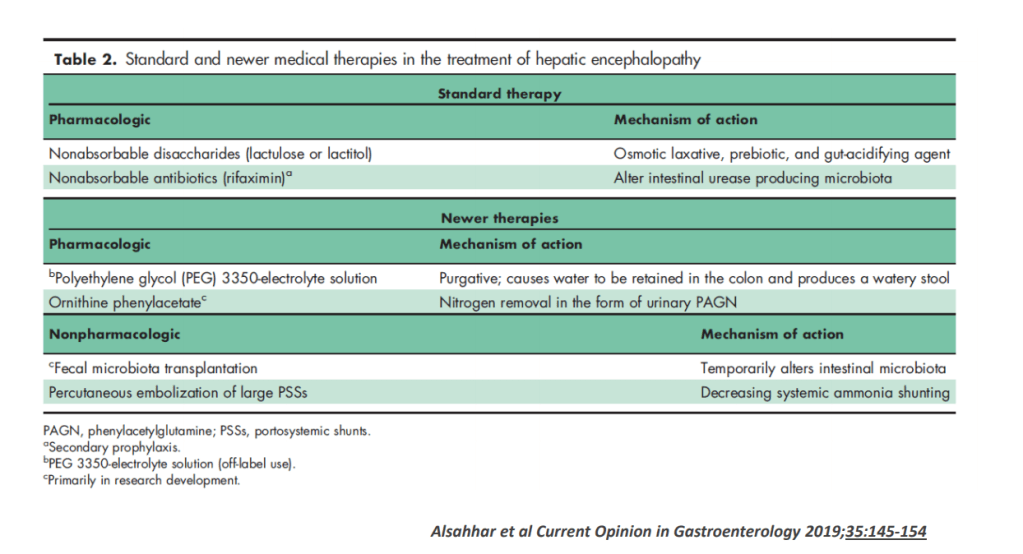
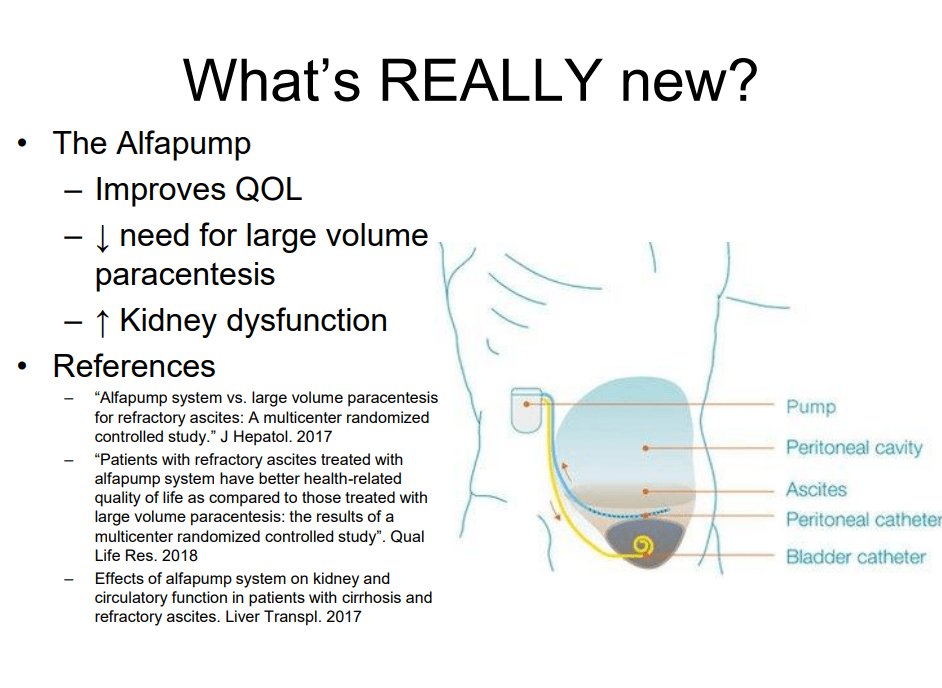
On Friday, our office started fielding questions regarding COVID-19 booster shots in our IBD population. Currently, I agree with the advice for patients as detailed by Dr. Rubin in the screenshots that follow. Key points:
- Studies have shown that IBD patients are not at increased risk of COVID-19 infections compared to the general population.
- Except for those on high-dose prednisone, it appears that our patient population with IBD does mount an adequate response to vaccination. That is, they are not considered severely immunocompromised.
- In short, it is reasonable, but not a clear recommendation, to give a booster mRNA vaccine dose to patients who are receiving anti-TNF agents and those receiving immunomodulators; this is a patient choice.

Also, from CDC 8/13/21:
- Talking with Patients Who Are Immunocompromised
- COVID-19 Vaccines for Moderately to Severely Immunocompromised People

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.