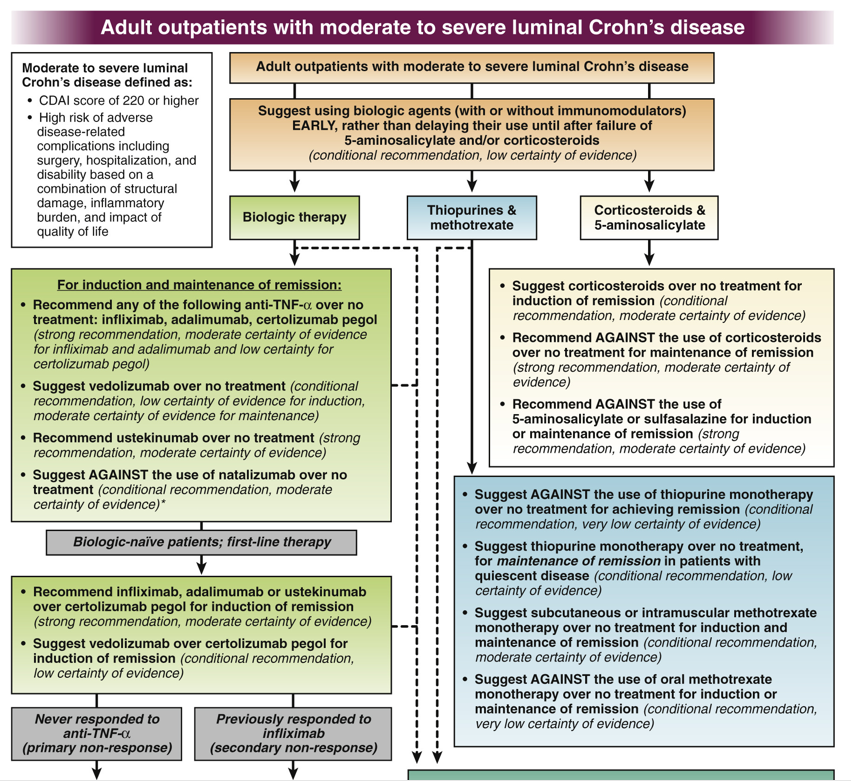
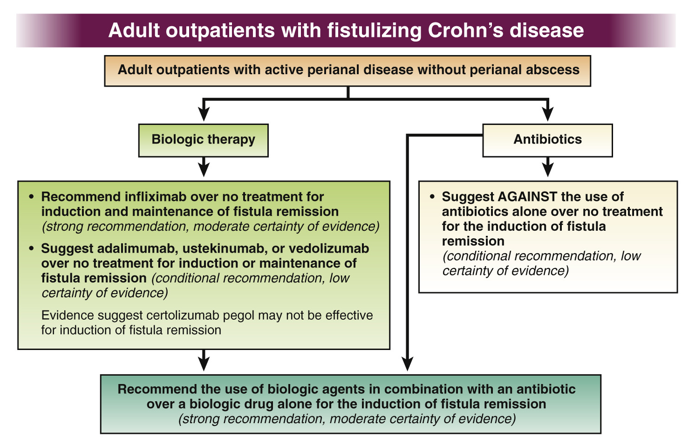
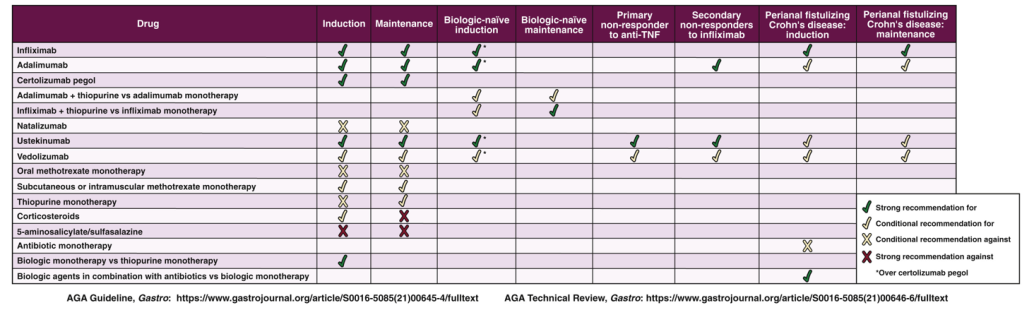
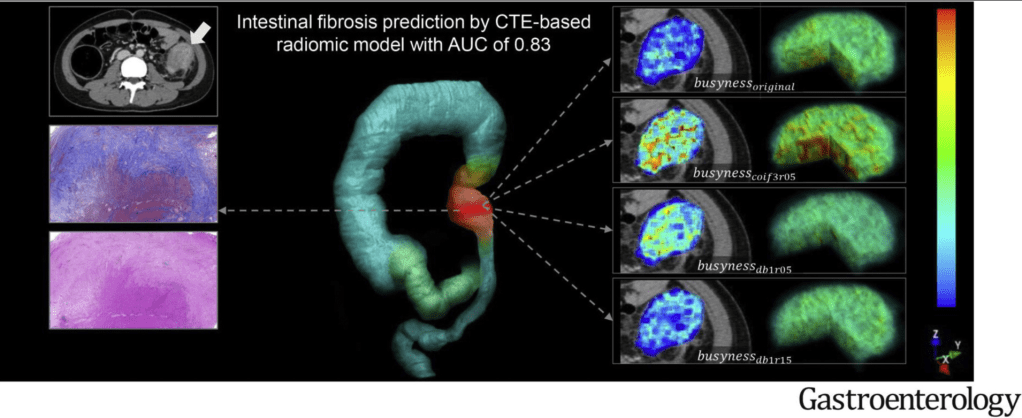
K Borovsky et al. JPGN 2021; 73: 80-85. Applying an Age-specific Definition to Better Characterize Etiologies and Outcomes in Neonatal Acute Liver Failure
This single-center retrospective study with 43 patients (over 11 year timeframe) identified etiology and outcomes for neonatal acute liver failure (NALF).
Key findings:
- Etiologies included viral infection (23%), gestational alloimmune liver disease with neonatal hemochromatosis (GALD-NH) (21%), cardiac-associated ischemia (16%), other ischemia (14%), genetic etiologies (9%), Trisomy 21-associated myelodysplasia (TAM) (7%), hemophagocytic lymphohistiocytosis (HLH) (2%), and not identified (7%)
- Infants with viral etiologies had the highest alanine aminotransferase (ALT) at presentation (1179 IU/L, interquartile range [IQR] 683–1585 IU/L) in contrast to low levels in GALD-NH (23 IU/L, IQR 18–64 IU/L)
- Across all etiologies, only 33% were alive at 1 year
- Among laboratory values at presentation, alpha-fetoprotein (AFP) was significantly higher in patients that survived with their native liver (P = 0.04)
Figure 4 provides a helpful management algorithm for NALF. Figure 4 is similar to the slide below (shared by lead author).

- -Consider empiric acyclovir in those with INR >/= 2.0 in the first 30 days of life
- -In those with normal to low elevation of aminotransferases, consider empiric IVIG while undergoing workup. Part of workup should include either MRI or lip biopsy for GALD
- -In those with moderate to severe elevation of aminotransferases, workup should include assessment for viral, HLH and genetic etiologies. Fulminant viral hepatitis or HLH likely with Ferritin levels >10,000. Hypoglycemia and hyperammonemia is suggestive of metabolic/mitochondrial disorder
- -Liver biopsy may be needed if etiologies not identified
Related blog posts:
- Changing Approach to Neonatal Acute Liver Failure
- A new GALD phenotype | gutsandgrowth
- Acute Liver Failure –Is There a Role for Steroids? | gutsandgrowth
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.