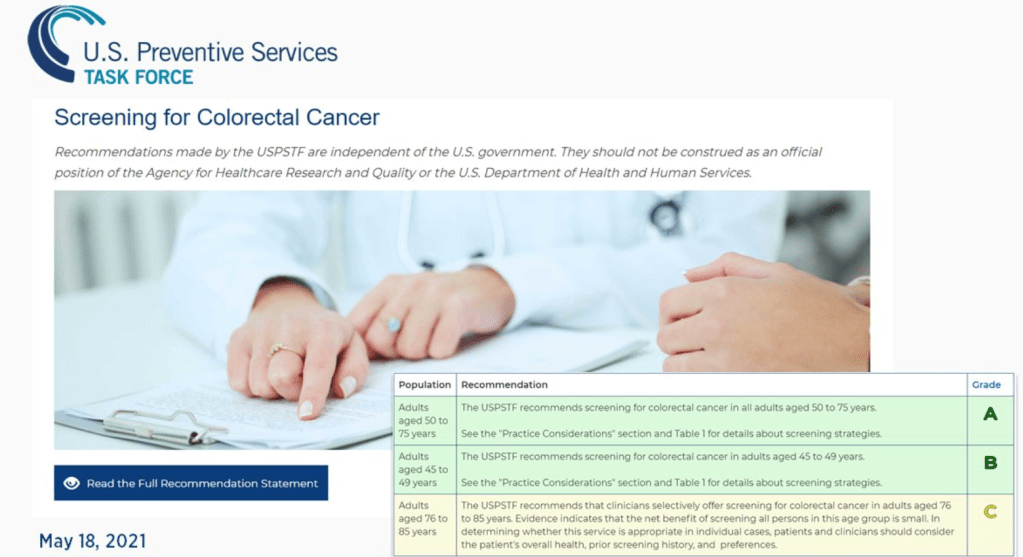
USPTF Recommends Starting CRC Screening at Age 45 in Healthy Adults


Z Wang et al. Clin Gastroenterol Hepatol 2021; 19: 788-796. Associations Between Nonalcoholic Fatty Liver Disease and Cancers in a Large Cohort in China
It is well-recognized that obesity/overweight increases the risk of cancer (related blog post: Cancer due to Overweight/Obesity). Wang et al provide data regarding cancer risk due specifically to nonalcoholic fatty liver disease (NAFLD) from a large prospective adult cohort (n=54,187). Key findings:
My take: This study shows that NAFLD increases the risk of cancer; much of this risk may be due to obesity/metabolic syndrome and associated chronic inflammation. Overall, cardiovascular disease in patients with NAFLD represents a higher risk for morbidity and mortality.
Related blog posts:

RS Boneh et al. Clin Gastroenterol Hepato 2021; 19: 752-759. Dietary Therapies Induce Rapid Response and Remission in Pediatric Patients With Active Crohn’s Disease
The authors collected data from a multicenter randomized trial of the CD exclusion diet (CDED) in children (mean age, 14.2 ± 2.7 y) with Crohn’s disease who were randomly assigned to groups given either exclusive enteral nutrition (EEN, n = 34) or the CDED with 50% (partial) enteral nutrition (PEN) (n = 39).
The CDED has been discussed previously on this blog; it aims to avoid animal and saturated fat, milk fat, gluten, specific emulsifiers, taurine, red (reduced heme) and processed meat, and certain fibers from some fruits and vegetables. In addition to excluding patients who received competing therapies (eg. steroids, immunomodulators, and biologics), the authors excluded patients with isolated large bowel disease (L2).
Key findings:
The authors note that the rapid response to dietary therapy suggests a role for a ‘trial by diet’. As such, dietary therapy could be used as monotherapy, for patients failing other therapies, or as a bridge to biological therapy. The authors note that the exact reasons for response to dietary therapy are unsettled and could be “due to both foods excluded and foods enriched in the diet.” In addition, they note that diet appears to be a trigger for inflammation and that reintroduction of foods leads to rebound in inflammation (eg. higher calprotectin) and dysbiosis.
My take: This study shows that dietary therapy works quickly. In this small study, the effectiveness of combined CDED with 50% PEN was similar to EEN.
Related blog posts:





More information from Rome Foundation:
Related blog posts:
Related humor: YouTube Link: SNL IBS Ad (4/10/21) Very funny!
Correction made on blog post on 5/12/21 Humira Dosing Guidelines. For Crohn’s induction >/= 40kg, there is not an 80mg dose on day 8, just 160mg day 1 and 80mg day 15. Blog post: Ustekinumab for Refractory Pediatric Ulcerative Colitis and Updated Adalimumab Dosing
————————————————————————
NY Times: Can We Learn to Live With Germs Again? (4/23/21)
An excerpt:
Almost everything we know about the microbiome is uncertain, including how our activities and environments influence its makeup...
Despite the now consensus recognition that air transmission, …we continue to annihilate every microbe in our midst, even though most are harmless…
Excessive hygiene practices, inappropriate antibiotic use and lifestyle changes such as distancing may weaken those [microbial] communities going forward in ways that promote sickness and imperil our immune systems. By sterilizing our bodies and spaces, they argue, we may be doing more harm than good…
Dr. Graham Rook, an emeritus professor of medical microbiology at University College London, likens the immune system to a computer. He says that the microbes we encounter in daily life — on other people and in our spaces — are the data that the immune system relies on to program and regulate its operations.
Deprived of these exposures, especially at the start of life, the immune system is prone to malfunction. The result can be allergies, asthma, autoimmune disorders, obesity, Type 2 diabetes and other chronic medical conditions...
“Even before the pandemic, we know that half of antibiotic use was inappropriate.”…
For those who aren’t yet able to mix and mingle — and right now, that’s most of us — there are other ways to support microbial health. “If you want to do something proactive right now, I would put eating a healthy diet high on your list,” says Dr. Emeran Mayer… plant foods (legumes, greens, whole fruits, a variety of vegetables), as well as fermented foods, support the richness and diversity of the gut microbiome. So, too, does limiting one’s intake of processed and fast foods, especially those that contain added sugar…
Before the pandemic, only one of the top 10 causes of death in America — influenza — was attributable to an infectious disease that someone could “catch.” Nearly all the rest, such as heart disease and stroke, cancer, brain disease and diabetes, are associated with poor microbiome health or dysfunction.
Related blog posts:

SC Shah et al. Gastroenterol 2021; 160: 1831-1841. Full text: AGA Clinical Practice Update on the Management of Refractory Helicobacter pylori Infection: Expert Review
Key recommendations:
Related blog posts:

R van Rijn et al. NEJM 2021; 384: 1391-1401. Hypothermic Machine Perfusion in Liver Transplantation — A Randomized Trial
Background: Nonanastomotic biliary strictures are a major complication after liver transplantation, and ischemia–reperfusion injury is a key mechanism in their development. Although static cold preservation provides some protection against injury, preclinical studies have shown that a short period of hypothermic oxygenated machine perfusion restores mitochondrial function and reduces damage.
Methods: In this multicenter, controlled trial, we randomly assigned patients who were undergoing transplantation of a liver obtained from a donor after circulatory death to receive that liver either after hypothermic oxygenated machine perfusion (machine-perfusion group) or after conventional static cold storage alone (control group). A total of 160 patients were enrolled, of whom 78 received a machine-perfused liver and 78 received a liver after static cold storage only (4 patients did not receive a liver in this trial).
Key points:
My take: Hypothermic oxygenated machine perfusion led to lower risk of nonanastomotic biliary strictures
Related blog posts:

As noted in previous blog posts (see below), adult guidelines for ulcerative colitis favor ustekinumab over vedolizumab for ulcerative colitis patients who have had anti-TNF therapy; recent pediatric guidelines appeared to do the opposite, possibly due to limited data with ustekinumab.
A recent study (J Dhaliwal et al. AP&T 2021; https://doi.org/10.1111/apt.16388. One‐year outcomes with ustekinumab therapy in infliximab‐refractory paediatric ulcerative colitis: a multicentre prospective study) provides prospective data on ustekinumab effectiveness when given to children with UC refractory to other biologics; n=25. Thanks to Ben Gold for this reference.
Key findings:
My take: Ustekinumab is a good option for pediatric patients with ulcerative colitis who are refractory to anti-TNF agents. More data are needed to help in positioning therapies.
Also, Humira (adalimumab) is now FDA-approved for children as young as 5 years with ulcerative colitis: FDA Approves Adalimumab as Treatment for Children With Ulcerative Colitis (2/25/21). “This approval is based on results from the phase 3, randomized, double-blind, multicenter ENVISION I (NCT02065557) study.” Abbvie has now updated their Humira dosing recommendations (Reference: https://www.rxabbvie.com/pdf/humira.pdf). Thanks to Clair Talmadge for this update.


Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
C Yzet et al. Clin Gastroenerol Hepatol 2021; 19: 668-679. Full Text: No Benefit of Concomitant Immunomodulator Therapy on Efficacy of Biologics That Are Not Tumor Necrosis Factor Antagonists in Patients With Inflammatory Bowel Diseases: A Meta-analysis
In a systematic review, key findings:
Why don’t immunomodulators seem to help? “Unlike anti-TNF, prospective studies as well as post hoc analysis of randomized controlled trial consistently reported a low immunogenicity [with ustekinumab and vedolizumab]…all the prospective studies available to date have shown no impact of immunomodulator on the trough serum level of vedolizumab or ustekinumab.”
Limitation: patients treated with combination therapy in the included studies could be more severe
My take: “This meta-analysis found that overall the use of combination therapy in patients treated with vedolizumab or ustekinumab was not associated with a clinical benefit in comparison with the use of monotherapy.”
————————————————————————————————————————–

