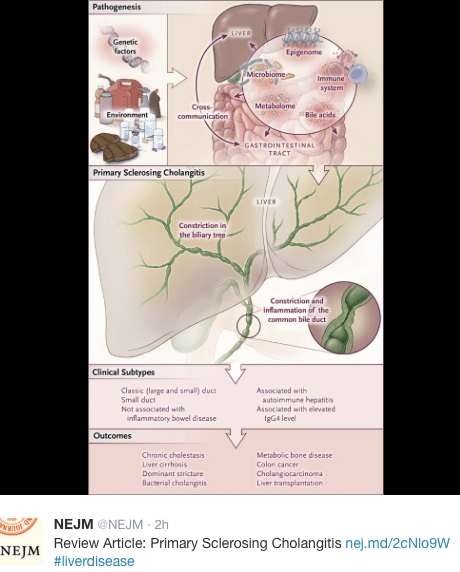
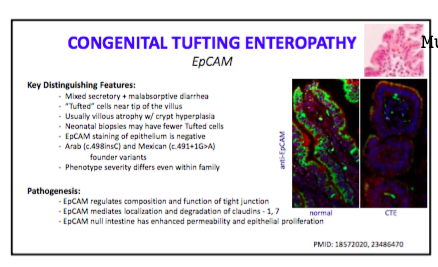
Fortunately, most mistakes do not result in long-lasting consequences. The authors’ of a recent report (JPEN J Parenter Enteral Nutr August 2016 vol. 40 no. 6 883-885) note a severe setback for a patient after accidental bolus of parenteral nutrition:
Here’s the abstract:
There is a paucity of data that exists regarding acute toxicity and management in the setting of parental nutrition (PN) overdose. We describe a case of a patient who received an accidental rapid bolus of PN and fat emulsion. She developed a seizure, metabolic acidosis, arrhythmias, myocardial ischemia, altered mental status, hypotension, and hypoxemia likely caused by elevated triglycerides, leading to a hyperviscosity syndrome. After failing standard therapy, she was successfully treated with a single-volume plasma exchange with resolution of symptoms. Fat emulsion or intravenous lipid emulsion and much of its safety have been recently described in its use as a rescue therapy in resuscitation from drug-related toxicity. Elevated serum triglyceride levels can result in a picture similar to a hyperviscosity syndrome. Plasma exchange is a known therapeutic modality for the management of hyperviscosity syndrome and a novel therapy in the treatment of hyperviscosity syndrome due to fat emulsion therapy. In a patient receiving PN with development of rapid deterioration of clinical status, without an obvious etiology, there should be consideration of PN overdose. A rapid assessment and treatment of severe electrolyte abnormalities should be undertaken immediately to prevent life-threatening cardiovascular and central nervous system collapse. If fat emulsion was rapidly coadministered and there are signs and symptoms of hyperviscosity syndrome, then consideration should be given to plasma exchange as an effective therapeutic treatment option.