M Matcovici et al. JPGN 2023; 76: 66-71. What Makes A “Successful” Kasai Portoenterostomy “Unsuccessful”?
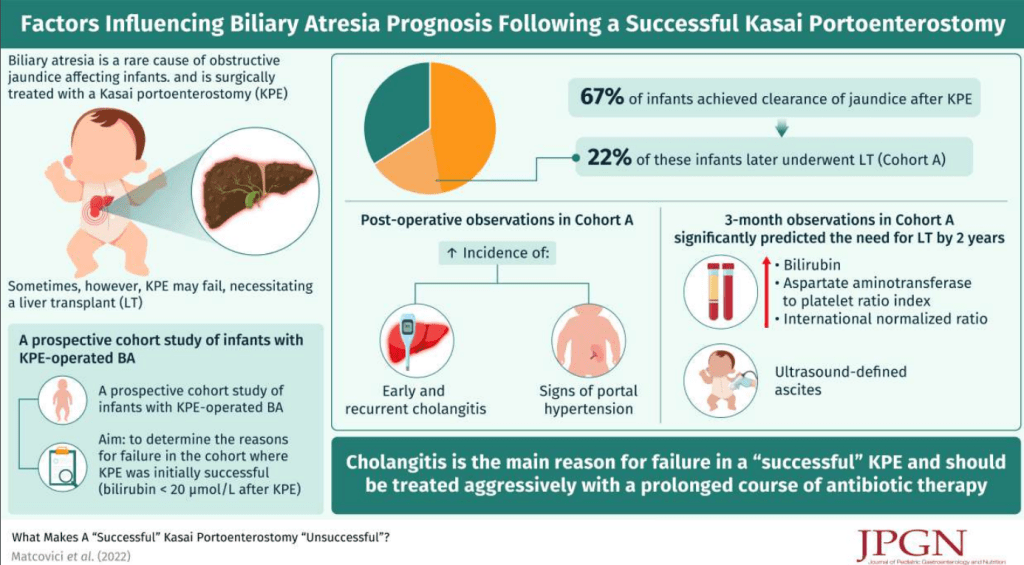
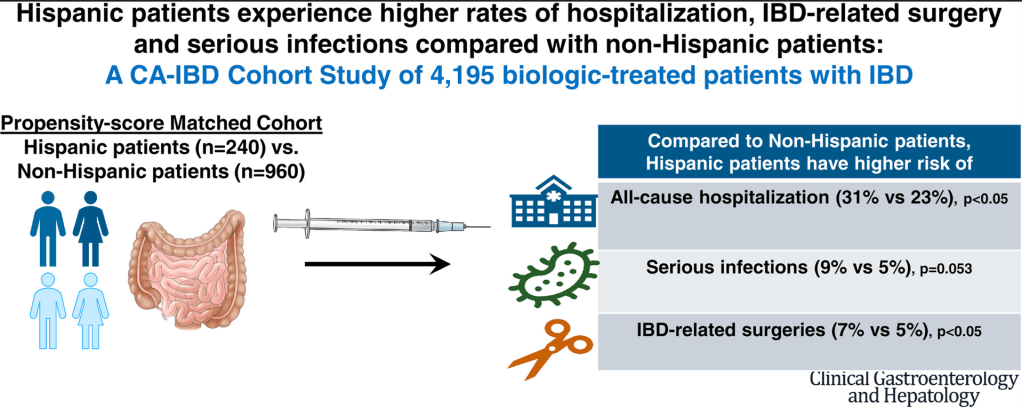
Methods: This review of a single-center prospective biliary atresia (BA) database examined which factors were associated with long-term success of a Kasai portoenterostomy (KPE). Successful KPE was defined by achieving a postoperative bilirubin of ≤20 µmol/L. Cholangitis was based on Tokyo (Adult) Guidelines (Calculator MD Calc: Tokyo Guidelines for Acute Cholangitis 2018). Explanation of Tokyo Guidelines: Tokyo Classification Cholangitis
Key findings:
- 90 (67%) achieved clearance of jaundice after KPE. From these 20 (22%) (Cohort A) underwent LT with the remainder continuing with native liver (Cohort B) (median follow-up of 4.15 years)
- Postoperatively, both cholangitis [any episode, 18/20 (90%) vs 15/70 (21%); P < 0.0001] and portal hypertension (PHT) [gastrointestinal (GI) bleed, 10/20 (50%) vs 2/70 (2.8%); P < 0.0001] were significantly more common in cohort A
My take: The authors assert that “failure is not preordained at KPE but due to recurrent cholangitis and/or symptoms of PHT.” In my view, this study shows an association but not causation of cholangitis/PHT with increased likelihood of KPE failure. It is quite possible that the cholangitis develops in those with suboptimal bile flow; thus, cholangitis (as well as PHT) may be an indicator that the KPE is not working as well, rather than the reason. Yet, it is also likely that episodes of cholangitis exacerbate any underlying problems.
Related blog posts:
- Cholangitis After Kasai Procedure for Biliary Atresia
- Online Aspen Webinar (Part 5) -Biliary Atresia Diagnosis and Screening
- Biliary Atresia Biomarkers 2020
- Useful Data on Cholangitis Following Kasai Portoenterostomy
- Impact of Kasai Portoenterostomy on Liver Transplantation
- START Study: Steroids Not Effective For Biliary Atresia (After Kasai)
- Landmark Study on Universal Screening for Biliary Atresia -It Works!