Smith-Bindman R, Chu PW, Azman Firdaus H, et al. JAMA Intern Med. Published online April 14, 2025. doi:10.1001/jamainternmed.2025.0505. Open Access! Projected Lifetime Cancer Risks From Current Computed Tomography Imaging
Methods: Lifetime radiation-induced cancer incidence and 90% uncertainty limits (UL) were estimated by age, sex, and CT category using National Cancer Institute software based on the National Research Council’s Biological Effects of Ionizing Radiation VII (BEIR VII) models and projected to the US population using scaled examination counts.
Key findings:
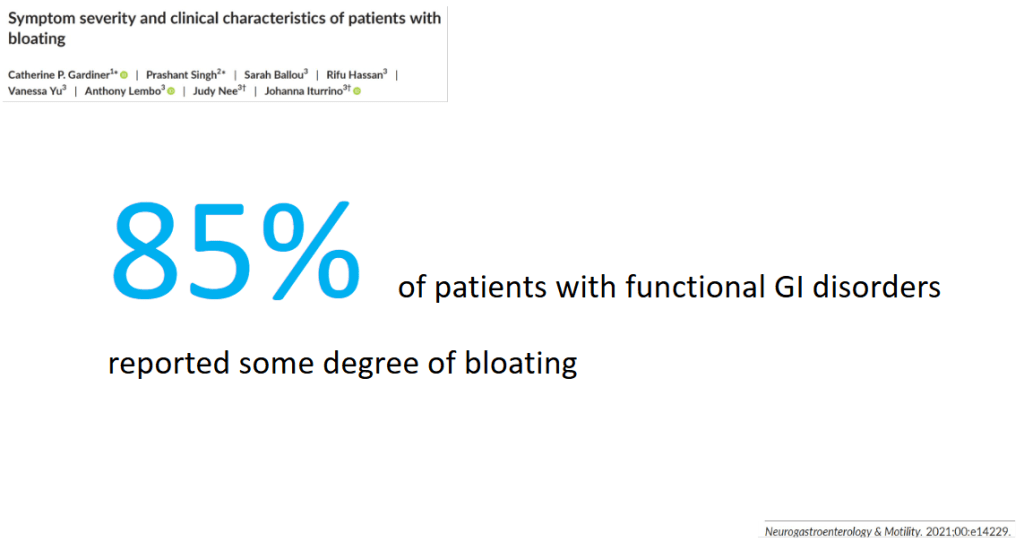
- Ninety-three million CT examinations were performed in 61 510 000 patients in the United States in 2023, including an estimated 3,069,000 CTs (3.3%) in 2,570,000 children (4.2%) and 89,931,000 CTs (96.7%) in 58,940,000 adults (95.8%)
- In this risk model, the 93 million CT examinations performed in 62 million patients in 2023 were projected to result in approximately 103,000 future cancers
- Estimated radiation-induced cancer risks were higher in children and adolescents, yet higher CT utilization in adults accounted for most (93,000) radiation-induced cancers
- “If current practices persist, CT-associated cancer could eventually account for 5% of all new cancer diagnoses annually”
Discussion: “The projected number of radiation-induced cancers in this analysis is 3 to 4 times higher than the earlier assessment of CT exposure for several reasons”
- CT use is 30% higher today than in 2007
- Dose modeling in this study accounted for multiphase scanning
- Substantially higher organ doses in this study were reconstructed using newer dosimetry methods
- More granular CT categories reflecting imaging indications that have important dose differences
- “Many of the model assumptions were conservative” and could underestimate the risk
My take (borrowed from authors): “Even very small cancer risks will lead to a significant number of future cancers given the tremendous volume of CT use in the United States…CT could be responsible for approximately 5% of cancers diagnosed each year. This would place CT on par with other significant risk factors, such as alcohol consumption (5.4%) and excess body weight (7.6%)”
Related blog posts:
- Risk from CT scans -Best Data to Date (2013)
- Ionizing Radiation Exposure in Adults with Inflammatory Bowel Disease
- Beyond the bombs; cancer risks of low-dose medical radiation